Abstract
Anthropogenic habitat modification and fragmentation is considered one of the most serious threats to biodiversity. To develop effective conservation strategies towards such pressures we need to improve our understanding of how species richness and community composition are shaped by species’ responses to landscape patterns. In this study we tested relationships between patch size and isolation, species richness and species traits in a fragmented landscape of calcareous grasslands—a diversity hotspot in Southern Norway. We recorded a total of 381 vascular plant species, of which 50 are considered habitat specialists, distributed among 86 habitat patches (50–9,475 m2) in 22 of 50 randomly sampled 500 × 500 m-plots. We found that large habitat patches held more species of both generalists and specialists than small habitat patches and that well-connected patches held more specialist species than isolated patches. About 1/3 of the habitat specialists in this study system showed vulnerability to isolation, i.e. lower probability of occurrence in isolated patches. Traits related both to persistence (short lifespan) and colonization ability (low seed production per plant) were predictors of vulnerability to isolation. Our results indicate that both colonization and extinction processes affect species composition and richness, and that the rescue-effect—mitigation of local, area-dependent extinctions through colonization—is reduced in isolated patches. These findings suggest that conservation strategies should place greater emphasis on the spatial configuration of the habitat network, and on the preservation of colonization processes to ensure regional persistence of species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthropogenic habitat modification represents the most important cause of habitat loss and fragmentation (Fahrig 2003), and such landscape changes represent one of the most serious threats to biodiversity (Foley et al. 2005; Rockstrom et al. 2009). Habitat protection is an established conservation measure that can alleviate the negative effects of land use changes. Lately, the spatial patterns and functional connectivity within a network of protected areas have received increased attention, both in ecology and in international policy initiatives. The international Aichi Biodiversity Targets state that the areas of particular importance for biodiversity and ecosystem services are to be conserved through “ecologically representative and well-connected systems of protected areas and other effective area-based conservation measures” (Convention on Biological Diversity Strategic plan for biodiversity 2011). To fulfill this target we need to improve our understanding of how species and communities respond to spatial patterns in the landscape, based on empirical research.
Island biogeography and metapopulation dynamics predict that species persistence in fragmented habitats depend on local colonization and extinction processes (MacArthur and Wilson 1967; Levins 1969). As habitat loss leads to decrease in size of fragments and increased fragment isolation, it reduces local population sizes and rates of colonization and enhances the risk of local extinctions (Hanski 1998; Fahrig 2003; Rybicki and Hanski 2013). Consequently local species richness can be expected to decrease as habitat patch size is reduced and distance between habitat patches is increased (MacArthur and Wilson 1967; Losos and Ricklefs 2009). A more relevant question for conservation management than species richness per se is species composition, i.e. which species are likely to persist or disappear when habitats are lost (Hobbs and Yates 2003). Classical metapopulation models (Levins 1969; Hanski 1994) take the perspective of individual species but do not consider in detail how species differ beyond local persistence and colonization abilities. Recent literature reviews emphasize that species respond differently to habitat loss and fragmentation depending on their life history strategies (Hobbs and Yates 2003; Henle et al. 2004; Ewers and Didham 2006), and several characteristics are predicted to predispose species to vulnerability to fragmentation (Table 1).
Firstly, plant species with a high habitat specificity—habitat specialists—will experience their habitat as more fragmented compared to generalist species, which have a higher probability of occurring also in the surrounding landscape (Henle et al. 2004; Ewers and Didham 2006). Consequently, species with narrow habitat requirements are predicted to be more strongly affected by habitat fragmentation than generalist species (Andrén et al. 1997). Furthermore Adriaens et al. (2006) found the species richness of habitat specialists, but not of generalists, to be negatively affected by isolation in fragmented calcareous grasslands in Belgium. Secondly, the capacity of plants to colonize suitable habitat patches depends on their reproductive potential (Turnbull et al. 2000) and their dispersal range (Johst et al. 2002; Nathan 2006). With a low number of offspring, colonization rates are reduced, making species with a low reproductive potential more prone to negative effects of habitat fragmentation (Higgins et al. 2003; Henle et al. 2004). Low reproductive potential is also frequently associated with low population growth, which reduces the probability of successful establishment of a population (Henle et al. 2004). A high dispersal capacity increases the probability of recolonizing habitat patches in the landscape and may increase the long-term persistence of species in a fragmented landscape (Johst et al. 2002; Verheyen et al. 2003). Finally, traits that positively affect plant population persistence, such as long lifespan, iteroparity (the ability to flower more than once), clonal reproduction and long-lived seed banks, may allow the formation of remnant populations that persist over long periods of unfavourable environmental conditions and low recruitment rates (Eriksson 1996; Lindborg et al. 2005). Traits enhancing population persistence may thus decrease the species’ vulnerability to fragmentation on a short-term scale. If environmental perturbations result in permanently unfavourable conditions, this can lead to delayed extinctions, also called extinction debt (Fahrig 2002; Kuussaari et al. 2009; Cousins and Vanhoenacker 2011; Hylander and Ehrlén 2013). Short-lived species respond more rapidly to changes in environmental conditions than long-lived (Morris et al. 2008) and have higher extinction rates (Ehrlén and van Groenendael 1998).
In this study, we use dry calcareous grasslands to study the importance of plant traits for vulnerability to fragmentation among vascular plants with high habitat specificity (habitat specialists). Calcareous grasslands are among the most species-rich habitats in northern and central Europe. Several calcareous grassland types are listed as priority habitats for conservation under the EU Habitats Directive (Wolkinger and Plank 1981; LIFE 2008). They face severe threats both from habitat loss and habitat degradation (e.g. shrub invasion, afforestation, and agricultural intensification). In order to safeguard the calcareous grasslands we need a better understanding of the interplay between patch size and patch isolation on species richness and composition in these habitats.
We use a study system of dry calcareous grasslands in the Oslo area, Southern Norway, where land use pressure has been, and still is, substantial. The distribution of dry calcareous grassland in the study area is naturally fragmented with several of the habitat patches located on islands in the Oslo fjord. Anthropogenic impact, through habitat conversion and degradation, has resulted in a further reduction in the mean habitat patch size and an increase in the mean patch isolation. A critical threshold of fragmentation can be reached where the distribution of habitat patches no longer coincides with the migration patterns of the species and metapopulation dynamics are disrupted (Hanski et al. 1995; With and Crist 1995). At this threshold level the probability of population extinction increases sharply (Fahrig 2001; Swift and Hannon 2010). However, no common threshold value exists across species and habitats (Fahrig 2001). Our aim is to investigate how the current spatial pattern of dry calcareous grasslands within this area affects the species richness of plants. Using information on species characteristics, we investigate the relationship between plant traits and response to spatial patterns. Specifically, we address the following questions: (1) How does the richness of generalist and specialist plant species respond to size and isolation of remaining patches of dry calcareous grasslands? (2) Can the response of individual habitat specialist species to habitat fragmentation be explained by species characteristics such as longevity and reproductive capacity, and if so, does it match the theoretical predictions of traits predisposing for vulnerability to fragmentation? And finally (3) How can our results improve management decisions on which patches to prioritize for conservation, in an area under a major land use pressure?
Materials and methods
Study area
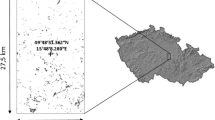
The study was carried out in the inner Oslo fjord, in the counties of Oslo and Akershus, Norway (Fig. 1). The habitat dry calcareous grasslands in Norway is restricted to bedrock sites dating to the period from Cambrian to Silurian, and mainly found in a narrow zone between the sea and the forested inland areas. The habitat is frequently exposed to drought due to a combination of high soil drainage and exposure to wind and sun. This, combined with effects of land rise and former grazing, creates the characteristic vegetation type. Dry calcareous grasslands in the inner Oslo fjord have a high occurrence of nationally red-listed species, including vascular plants, lichens, fungi and invertebrates.
Due to the dependency on bedrock qualities, exposure and a warm and dry climate, dry calcareous grasslands are naturally fragmented in the study area, and many of the habitat patches occur on islands in the Oslo fjord (Fig. 1). The Oslo fjord area is also the most densely populated area of Norway. Human impact, primarily through housing and infrastructure, has led to habitat loss with a reduction in habitat patch sizes and increased distances between them. Other important threats to the dry calcareous grasslands in the study area include recreational use (trampling, soil erosion), invasive species, and natural succession (transition to forest) as a result of the cessation of livestock grazing in the Oslo fjord area.
Habitat patch data
The mapping of dry calcareous grasslands in the study area is ongoing. Therefore, information on the occurrence of dry calcareous grasslands is not spatially comprehensive yet. We mapped a sample of dry calcareous grassland habitat patches by superimposing a grid system of 500 × 500 m plots on the study area giving a total of 238 plots (islands less than 0.25 km2 were treated as one sampling unit even if they were intersected by the sampling grid). The plots were numbered from 1−238, and using a random number generator we selected 50 plots for field inventory. In 22 of the plots we found dry calcareous grassland, and a total of 93 habitat patches were surveyed and digitized.
In order to fill the gaps for the analysis of connectivity, we merged data mapped in the field with a predictive habitat distribution model developed by Wollan et al. (2011). After a validation of the model based on presence/absence information from the mapped 500 × 500 m plots, areas with a prediction value above 0.62 outside the mapped 500 × 500 m plots were classified as dry calcareous grassland habitat. Above the chosen value, the area of correctly predicted presences increased significantly, while the amount of (wrongly predicted) absence decreased respectively. However, through the validation we also found an overestimation of occurrence of dry calcareous grasslands in the areas delineated from the model of about four times the mapped area of occurrence. Therefore, we downscaled the area of habitat polygons delineated from the prediction model by a factor of 0.25.
Connectivity
We calculated the connectivity S i of the 93 surveyed habitat patches to all patches j within a radius of 1 km of the patch centroid as:
where A j is the area of patch j (ha), d ij is the distance between the centroids of patches i and j (km), and α is the parameter of the exponential distribution setting the influence of distance on connectivity. Since we calculate the connectivity for a set of species differing in dispersal capacity, we set α = 1 (cf. Hanski 1994), which also makes our results comparable to previous studies in grasslands (e.g. Cousins et al. 2007; Lindborg 2007). The 1 km radius was chosen as it is commonly used in grassland studies (e.g. Cousins et al. 2007; Lindborg 2007), and represents the area that may affect species richness at the patch scale. However, the rank order of patch connectivity was similar if all patches within the study area were included, as opposed to including only patches within the 1 km-radius.
Vascular plant species data
In each habitat patch we recorded the occurrence of all vascular plants present. Seven of the habitat patches were incompletely surveyed and were excluded from further analyses. Fieldwork was carried out in June–July 2009 and 2010.
We defined the species to be either habitat specialists, that is, species which are strongly confined to the habitat dry calcareous grasslands within the study area, or generalists (occurring in a range of habitats) based on information in the flora (Lid and Lid 2005) and own knowledge of the species (Table 2). To characterize the habitat specialist species we selected six plant traits important for plant reproduction, dispersal and persistence (cf. Weiher et al. 1999), which represent analogs to the characteristics hypothesized to be important predictors of vulnerability to fragmentation (Henle et al. 2004; Table 1). Reproductive potential was represented by seed number, defined as the number of seeds per ramet/plant. Dispersal capacity was represented by dispersal agent (wind-dispersed or non-wind dispersed) and seed mass. To characterize persistence we used lifespan (annual and biannual species classified as short-lived, perennial species classified as long-lived), clonal reproduction (three categories; (1) little or no vegetative spread, (2) shortly creeping, (3) far creeping, cf. Fitter and Peat 1994), and seed bank longevity, using the seed bank longevity index (Bekker et al. 1998). Trait values were collected from trait databases (Kleyer et al. 2008; Kühn et al. 2004; Fitter and Peat 1994) and the local flora (Lid and Lid 2005). When several records were available in the databases, we used median values for each combination of trait and species.
Statistical analyses
First we assessed whether plant species richness was related to patch size and connectivity. Due to the spatially structured sampling of habitat patches, with patches nested within 500 × 500 m plots, we used linear mixed effect models, with habitat patch nested within plot as random factor (cf. Zuur et al. 2009). We used maximum likelihood estimation (ML), model simplification and comparison of models using the AIC criterion, and fitted the optimal model using restricted maximum likelihood estimation (REML), as recommended by Zuur et al. (2009). Patch size was log2-transformed to ensure homogeneity of variance and normally distributed residuals. Separate models were run for total plant species richness and for habitat specialist species richness.
Secondly, we investigated the importance of patch size and patch connectivity on the probability of occurrence of habitat specialists. We constructed two separate models for each habitat specialist species occurring in ≥10 habitat patches. We used generalized linear mixed effect models (GLMMs), with binomial error structure and patch nested within plot as random factor. Presence/absence of the species was used as the response variable. The first model used patch size as predictor variable, and the second model used patch connectivity as predictor variable. Inspection of parameter estimates and p-values were used to assess whether probability of occurrence was related to patch size or patch connectivity. We did not correct for multiple testing. p-values for each model are included in Table 2.
We then classified species with a probability of occurrence significantly related to patch size or connectivity as vulnerable to area or isolation and species without such a relationship as being neutral to area or isolation (Table 2). Then we used generalized linear models (GLMs) to investigate the association between vulnerability to area/isolation and plant traits. Vulnerability to area and isolation were used as response variables in two separate model runs and seed number, dispersal agent, seed mass, lifespan, clonality and seed bank longevity were used as predictor variables. The models were run with a binomial error structure. We used backward model simplification with comparison of AIC-values to guide model selection.
All statistical analyses were carried out in R version 3.0.1 (R Development Core Team 2013), with packages lme4 (Bates et al. 2013) and nlme (Pinheiro et al. 2013).
Results
A total of 381 vascular plant species were recorded in the 86 habitat patches, of which 50 species were characterized as habitat specialists. There were 26–108 (mean ± SD: 66.9 ± 21.3) species and the number of habitat specialists varied from 2–30 (14.7 ± 6.0) species per patch. There was a large size range of habitat patches (50–9475 m2, 901.3 ± 1317.2 m2), and patch connectivity varied from 0.02 to 4.04 (1.30 ± 1.27). Patch size and patch connectivity were positively correlated across the total study area as a whole (Spearman’s rank correlation r s = 0.318, p = 0.003, n = 86), but when taking account of the spatial structure of the data (habitat patches nested within plots), the relationship between habitat patch size and connectivity was weakly negative (β =−0.037, SE = 0.012, p = 0.003). This means that for a given patch size, a large variation in connectivity was found across the habitat patches, and vice versa.
The total species richness was positively related to patch size only (β = 5.594, SE = 1.403, p < 0.001), i.e. large habitat patches held higher species richness than small patches. The species richness of habitat specialists was positively related to both patch size (β = 1.146, SE = 0.382, p = 0.004) and patch connectivity (β = 2.153, SE = 0.920, p = 0.022), i.e. large habitat patches held higher habitat specialist species richness than small patches, and a well-connected habitat patch of a given size was on average more species rich than an isolated patch of the same size.
Of the 50 habitat specialists found in the sample sites, 34 occurred in ≥10 habitat patches. Of these only six species had a probability of occurrence significantly related to patch size (Table 2). No plant traits could significantly contribute to explaining species vulnerability to habitat patch size (results not shown).
A total of 11 habitat specialist species could be characterized as vulnerable to isolation (Table 2). After model simplification, the best model explaining vulnerability to patch isolation included lifespan (β = −2.416, SE = 1.197, p = 0.044) and seed number (β = −0.797, SE = 0.336, p = 0.018), with a higher probability for vulnerability in short-lived species and species with low seed number (Fig. 2).
Discussion
The dry calcareous grasslands in the Oslo fjord area represent a naturally fragmented system of small habitat patches with high conservation value. Human impact, through habitat conversion and degradation, has led to loss of habitat and increased fragmentation in this densely populated area. As expected, the large habitat patches have higher vascular plant species richness, both totally and of habitat specialists, than small patches, reflecting the general species-area effect, i.e. an increase in the number of species with the area sampled (Arrhenius 1921; Rosenzweig 1995). Focusing only on habitat specialists we find an additional positive effect of patch connectivity, implying that for a habitat patch of a given size, a well-connected patch will hold more habitat specialists than an isolated patch, in line with findings in calcareous grasslands (Adriaens et al. 2006) and heathlands (Piessens et al. 2004) in Belgium. However, several studies fail to find an effect of present-day patch size or isolation on the richness of habitat specialists in semi-natural grasslands (Krauss et al. 2004; Lindborg and Eriksson 2004; Helm et al. 2006; Cousins et al. 2007). As many plant species are long-lived with the capacity to form remnant populations (Eriksson 1996), a slow response to changes in environmental conditions is expected, at least partly explaining this lack of relationship. This is further supported by a significant relationship between historical landscape configurations and present-day species richness in several studies (Lindborg and Eriksson 2004; Helm et al. 2006; Krauss et al. 2010), which suggests that there may be an extinction debt in these systems. The size of extinction debts can be expected to vary at different spatial scales (Cousins and Vanhoenacker 2011) and will increase with increasing habitat change (Hylander and Ehrlén 2013). It is thus likely that differences in the studied landscapes could contribute to explaining the lack of a consistent relationship between species richness and habitat size and isolation in fragmented grassland (Cousins 2009). Based on a literature review Cousins (2009) suggests that the amount of target habitat remaining in the landscape is a key factor. In landscapes with >10 % of the target habitat remaining the existence of extinction debts is to be expected, and plant species richness should be more strongly related to the historical landscape configuration. In landscapes with <10 % of the target habitat remaining, however, a stronger relationship with present landscape configuration could be expected. The dry calcareous grassland patches in the Oslo fjord area are small (0.005–0.95 ha) compared to other grassland studies (Helm et al. 2006; Cousins et al. 2007; Lindborg 2007; Lindborg et al. 2011) and naturally fragmented, which could explain why we find significant effects of present-day isolation on habitat specialist species richness. In small-scale patches, the populations will be smaller and thus more sensitive to both genetic and stochastic extinctions (Groom 1998). If the inter-patch distance is increased by human land use changes beyond the dispersal capacity of the specialist species, a further response to increased isolation is to be expected.
About 1/3 of the habitat specialists in our study system show vulnerability to isolation, defined as having lower probability of occurrence in isolated habitat patches. Traits related both to persistence and recolonization ability are predictors of species’ vulnerability. Firstly, we find that short-lived species show higher vulnerability to isolation than long-lived species, in line with the theory of remnant population formation (Eriksson 1996). Short-lived plants commonly have large fluctuations in the number of individuals (Matthies et al. 2004) and a high rate of local extinctions (Fischer and Stöcklin 1997), and small, isolated populations can suffer from reproductive failure (Groom 1998). Variability in survival and reproduction has a stronger negative impact on the population growth and stability of short-lived species than long-lived (García et al. 2008; Morris et al. 2008). The empirical evidence of the importance of lifespan in grassland fragmentation studies is inconclusive. Some studies find no vulnerability of short-lived species, suggesting that short-lived species are more dependent on local habitat characteristics (such as rocky outcrops) than of habitat patch size or isolation per se (Adriaens et al. 2006; Lindborg et al. 2011). In our study system the results indicate that the rate of local extinctions is higher for short-lived species in isolated than well-connected patches (Bruun 2000; Lindborg 2007). As we did not assess single-trait relationships with vulnerability to patch isolation and size, but rather assessed the relative importance of the different traits to vulnerability in a multiple regression framework, no effect of the other persistence related traits (clonality, seed bank longevity) was found. Short-lived species are however often non-clonal (Piqueray et al. 2011; Fischer and Stöcklin 1997). A larger sample of species would be necessary to assess the importance of clonality for perennial species.
Secondly, we find that the number of seeds produced per plant is negatively related to vulnerability to isolation, i.e. species with a low seed production have a lower probability of occurrence in isolated habitat patches. Immigration or recolonization can reduce the risk of local extinctions (the rescue-effect; Brown and Kodric-Brown 1977), and modeling studies show that species with low reproductive rate require large amounts of habitat for persistence (Fahrig 2001) and that only species with a high fecundity and long-distance dispersal should be insensitive to habitat loss (Higgins et al. 2003). Seed production has also been found to be an important predictor of vulnerability to fragmentation in urban ruderal habitats (Schleicher et al. 2011) and forest understory herbs (Dupré and Ehrlén 2002). Our results suggest that the increased rates of local extinction (vulnerability of short-lived species) and reduced rates of local recolonization (vulnerability of species with low reproductive output) in combination affects the species richness of habitat specialists, and that mitigation of local, area-dependent extinctions through colonization is reduced in isolated patches.
No traits representing dispersal ability were related to vulnerability to isolation in our study system. Theoretical and empirical studies underline the importance of both mean dispersal range and the number of potential dispersers (Higgins et al. 2003; Schleicher et al. 2011). Terminal velocity is suggested as a relevant and easily measured trait for dispersal range (Schleicher et al. 2011), but due to several missing values in our dataset we did not include terminal velocity in the analyses. Instead we included seed mass and dispersal mode (wind- versus not wind-dispersed), which might have been too crude measures of interspecific differences in mean dispersal range. Dispersal mode was also found to be a poor predictor of vulnerability to isolation in Danish grasslands (Bruun 2000). A more detailed investigation of dispersal distances and how dispersal vectors interact with habitat fragmentation is warranted in order to understand how species are limited by migration in a fragmented landscape (Higgins et al. 2003).
Conclusions
Species predicted to show the fastest response to environmental change also have lowest persistence in the fragmented landscape of dry calcareous grasslands in the Oslo fjord area, suggesting that landscape structure indeed affects species richness in our study system. Being more species rich, conservation of the large habitat patches is an important prerequisite for protection of vascular plant biodiversity. The additional effect of connectivity, i.e. that isolated patches hold lower habitat specialist species richness than non-isolated patches of a given size, implies that maintaining well-connected, large habitat patches should be a management priority in this region.
Our findings support the recent international efforts within “connectivity conservation management” (cf. Worboys et al. 2010), which focus on the maintenance and restoration of connectivity of core areas of habitat. In order to maintain biodiversity patterns and ecological processes in the landscape, more attention should be given to the spatial configuration of habitat networks (Franklin and Lindenmayer 2009), and on the preservation of colonization processes to ensure regional persistence of species.
This is especially important for the most vulnerable species, like specialist species (several included on the national red list) associated with rare habitats in high-pressure areas. The dry calcareous grasslands in the Oslo fjord, along with scores of other areas of high biodiversity value around the world, occur in an area of several conflicting interests, including urban development and human recreation. Careful spatial considerations—combined with integrated cross-sector collaboration between conservation managers and urban planners—can improve the chance of long-time persistence of the biodiversity values also in high-pressure areas.
References
Adriaens D, Honnay O, Hermy M (2006) No evidence of a plant extinction debt in highly fragmented calcareous grasslands in Belgium. Biol Conserv 133:212–224
Andrén H, Delin A, Seiler A (1997) Population response to landscape changes depends on specialization to different landscape elements. Oikos 80:193–196. doi:10.2307/3546534
Arrhenius O (1921) Species and area. J Ecol 9:95–99
Bates D, Maechler M, Bolker B (2013) Lme4: Linear mixed-effect models using s4 classes. R package version 0.999999-2. http://CRAN.R-project.org/package=lme4. Accessed 15 Jan 2014
Bekker RM, Bakker JP, Grandin U, Kalamees R, Milberg P, Poschlod P, Thompson K, Willems JH (1998) Seed size, shape and vertical distribution in the soil: indicators of seed longevity. Funct Ecol 12:834–842
Brown JH, Kodric-Brown A (1977) Turnover rates in insular biogeography: effect of immigration on extinction. Ecology 58:445–449
Bruun HH (2000) Patterns of species richness in dry grassland patches in an agricultural landscape. Ecography 23:641–650. doi:10.1034/j.1600-0587.2000.230601.x
Convention on Biological Diversity Strategic plan for biodiversity 2011–2020. The Aichi Targets .
Cousins SAO (2009) Extinction debt in fragmented grasslands: paid or not? J Veg Sci 20:3–7. doi:10.1111/j.1654-1103.2009.05647.x
Cousins SAO, Vanhoenacker D (2011) Detection of extinction debt depends on scale and specialisation. Biol Conserv 144:782–787. doi:10.1016/j.biocon.2010.11.009
Cousins SAO, Ohlson H, Eriksson O (2007) Effects of historical and present fragmentation on plant species diversity in semi-natural grasslands in Swedish rural landscapes. Landsc Ecol 22:723–730. doi:10.1007/s10980-006-9067-1
Dupré C, Ehrlén J (2002) Habitat configuration, species traits and plant distributions. J Ecol 90:796–805
Ehrlén J, van Groenendael JM (1998) The trade-off between dispersability and longevity—an important aspect of plant species diversity. Appl Veg Sci 1:29–36
Eriksson O (1996) Regional dynamics of plants: a review of evidence for remnant, source-sink and metapopulations. Oikos 77:248–258
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–143
Fahrig L (2001) How much habitat is enough? Biol Conserv 100:65–74. doi:10.1016/s0006-3207(00)00208-1
Fahrig L (2002) Effect of habitat fragmentation on the extinction threshold: a synthesis. Ecol Appl 12:346–353. doi:10.2307/3060946
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. doi:10.1146/annurev.ecolsys.34.011802.132419
Fischer M, Stöcklin J (1997) Local extinctions of plants in remnants of extensively used calcareous grasslands 1950–1985. Conserv Biol 11:727–737. doi:10.1046/j.1523-1739.1997.96082.x
Fitter AH, Peat HJ (1994) The ecological flora database. J Ecol 82:415–425
Foley JA, DeFries R, Asner GP, Barford C, Bonan G, Carpenter SR, Chapin FS, Coe MT, Daily GC, Gibbs HK, Helkowski JH, Holloway T, Howard EA, Kucharik CJ, Monfreda C, Patz JA, Prentice IC, Ramankutty N, Snyder PK (2005) Global consequences of land use. Science 309:570–574. doi:10.1126/science.1111772
Franklin JF, Lindenmayer DB (2009) Importance of matrix habitats in maintaining biological diversity. PNAS 106:349–350. doi:10.1073/pnas.0812016105
García MB, Picó FX, Ehrlén J (2008) Life span correlates with population dynamics in perennial herbaceous plants. Am J Bot 95:258–262
Groom MJ (1998) Allee effects limit population viability of an annual plant. Am Natl 151:487–496. doi:10.1086/286135
Hanski I (1994) A practical model of metapopulation dynamics. J Anim Ecol 63:151–162. doi:10.2307/5591
Hanski I (1998) Metapopulation dynamics. Nature 396:41–49. doi:10.1038/23876
Hanski I, Pakkala T, Kuussaari M, Lei G (1995) Metapopulation persistence of an endangered butterfly in a fragmented landscape. Oikos 72:21–28
Helm A, Hanski I, Pärtel M (2006) Slow response of plant species richness to habitat loss and fragmentation. Ecol Lett 9:72–77. doi:10.1111/j.1461-0248.2005.00841.x
Henle K, Davies KF, Kleyer M, Margules C, Settele J (2004) Predictors of species sensitivity to fragmentation. Biodiv Cons 13:207–251
Higgins SI, Lavorel S, Revilla E (2003) Estimating plant migration rates under habitat loss and fragmentation. Oikos 101:354–366. doi:10.1034/j.1600-0706.2003.12141.x
Hobbs RJ, Yates CJ (2003) Impacts of ecosystem fragmentation on plant populations: generalising the idiosyncratic. Aust J Bot 51:471–488. doi:10.1071/bt03037
Hylander K, Ehrlén J (2013) The mechanisms causing extinction debts. Trends Ecol Evol 28:341–346
Johst K, Brandl R, Eber S (2002) Metapopulation persistence in dynamic landscapes: the role of dispersal distance. Oikos 98:263–270. doi:10.1034/j.1600-0706.2002.980208.x
Kleyer M, Bekker RM, Knevel IC, Bakker JP, Thompson K, Sonnenschein M, Poschlod P, van Groenendael JM, Klimes L, Klimesova J, Klotz S, Rusch GM, Hermy M, Adriaens DGB, Bossuyt B, Endels P, Götzenberger L, Hodgson JG, Jackel AK, Dannemann A, Kühn I, Kunzmann D, Ozinga WA, Römermann C, Stadler M, Schlegelmilch J, Steendam HJ, Tackenberg O, Willmann B, Cornelissen JHC, Eriksson O, Garnier E, Fitter A, Peco B (2008) The LEDA traitbase: a database of plant life-history traits of north west europe. J Ecol 96:1266–1274
Krauss J, Klein AM, Steffan-Dewenter I, Tscharntke T (2004) Effects of habitat area, isolation, and landscape diversity on plant species richness of calcareous grasslands. Biodiv Conserv 13:1427–1439. doi:10.1023/b:bioc.0000021323.18165.58
Krauss J, Bommarco R, Guardiola M, Heikkinen RK, Helm A, Kuussaari M, Lindborg R, Ockinger E, Partel M, Pino J, Poyry J, Raatikainen KM, Sang A, Stefanescu C, Teder T, Zobel M, Steffan-Dewenter I (2010) Habitat fragmentation causes immediate and time-delayed biodiversity loss at different trophic levels. Ecol Lett 13:597–605. doi:10.1111/j.1461-0248.2010.01457.x
Kühn I, Durka W, Klotz S (2004) Biolflor—a new plant-trait database as a tool for plant invasion ecology. Div Distr 10:363–365
Kuussaari M, Bommarco R, Heikkinen RK, Helm A, Krauss J, Lindborg R, Öckinger E, Pärtel M, Pino J, Rodà F, Stefanescu C, Teder T, Zobel M, Steffan-Dewenter I (2009) Extinction debt: a challenge for biodiversity conservation. Trends Ecol Evol 24:564–571
Levins R (1969) Some demographic and genetic consequences of environmental heterogeneity for biological control. Bull Am Entomol Soc 15:237–240
Lid J, Lid DT (2005) Norsk flora, 7th edn. Det Norske Samlaget, Oslo
LIFE (2008) Life and europe’s grasslands: Restoring a forgotten habitat. http://ec.Europa.Eu/environment/life/publications/lifepublications/lifefocus/documents/grassland.Pdf.
Lindborg R (2007) Evaluating the distribution of plant life-history traits in relation to current and historical landscape configurations. J Ecol 95:555–564. doi:10.1111/j.1365-2745.2007.01232.x
Lindborg R, Eriksson O (2004) Historical landscape connectivity affects present plant species diversity. Ecology 85:1840–1845. doi:10.1890/04-0367
Lindborg R, Cousins SAO, Eriksson O (2005) Plant species response to land use change—Campanula rotundifolia, Primula veris and Rhinanthus minor. Ecography 28:29–36
Lindborg R, Helm A, Bommarco R, Heikkinen RK, Kühn I, Pykälä J, Pärtel M (2011) Effect of habitat area and isolation on plant trait distribution in european forests and grasslands. Ecography 34:001–008. doi:10.1111/j.1600-0587.2011.07286.x
Losos JB, Ricklefs RE (eds) (2009) The theory of island biogeography revisited. Princeton University Press, Princeton
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
Matthies D, Brauer I, Maibom W, Tscharntke T (2004) Population size and the risk of local extinction: empirical evidence from rare plants. Oikos 105:481–488
Morris WF, Pfister CA, Tuljapurkar S, Haridas CV, Boggs CL, Boyce MS, Bruna EM, Church DR, Coulson T, Doak DF, Forsyth S, Gaillard JM, Horvitz CC, Kalisz S, Kendall BE, Knight TM, Lee CT, Menges ES (2008) Longevity can buffer plant and animal populations against changing climatic variability. Ecology 89:19–25
Nathan R (2006) Long-distance dispersal of plants. Science 313:786–788
Piessens K, Honnay O, Nackaerts K, Hermy M (2004) Plant species richness and composition of heathland relics in north-western belgium: evidence for a rescue-effect? J Biogeogr 31:1683–1692. doi:10.1111/j.1365-2699.2004.01056.x
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Development Core Team (2013) Nlme: linear and nonlinear mixed effect models. R packag version 3:1–109
Piqueray J, Bisteau E, Cristofoli S, Palm R, Poschlod P, Mahy G (2011) Plant species extinction debt in a temperate biodiversity hotspot: community, species and functional traits approaches. Biol Conserv 144:1619–1629. doi:10.1016/j.biocon.2011.02.013
R Development Core Team (2013) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. doi:http://www.R-project.org.
Rockstrom J, Steffen W, Noone K, Persson A, Chapin FS, Lambin EF, Lenton TM, Scheffer M, Folke C, Schellnhuber HJ, Nykvist B, de Wit CA, Hughes T, van der Leeuw S, Rodhe H, Sorlin S, Snyder PK, Costanza R, Svedin U, Falkenmark M, Karlberg L, Corell RW, Fabry VJ, Hansen J, Walker B, Liverman D, Richardson K, Crutzen P, Foley JA (2009) A safe operating space for humanity. Nature 461:472–475. doi:10.1038/461472a
Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, Cambridge
Rybicki J, Hanski I (2013) Species-area relationships and extinctions caused by habitat loss and fragmentation. Ecol Lett 16:27–38. doi:10.1111/ele.12065
Schleicher A, Biedermann R, Kleyer M (2011) Dispersal traits determine plant response to habitat connectivity in an urban landscape. Landsc Ecol 26:529–540. doi:10.1007/s10980-011-9579-1
Swift TL, Hannon SJ (2010) Critical thresholds associated with habitat loss: a review of the concepts, evidence, and applications. Biol Rev 85:35–53
Turnbull LA, Crawley MJ, Rees M (2000) Are plant populations seed-limited? A review of seed sowing experiments. Oikos 88:225–238. doi:10.1034/j.1600-0706.2000.880201.x
Verheyen K, Honnay O, Motzkin G, Hermy M, Foster DR (2003) Response of forest plant species to land-use change: a life-history trait-based approach. J Ecol 91:563–577
Weiher E, van der Werf A, Thompson K, Roderick M, Garnier E, Eriksson O (1999) Challenging Theophrastus: a common core list of plant traits for functional ecology. J Veg Sci 10:609–620
With KA, Crist TO (1995) Critical thresholds in species responses to landscape structure. Ecology 76:2446–2459. doi:10.2307/2265819
Wolkinger F, Plank S (1981) Dry grasslands of Europe. Nature and environment series 21. Council of Europe, Strasbourg.
Wollan AK, Bakkestuen V, Halvorsen R (2011) Spatial predictive modelling of dry calcareous grasslands in the Oslofjord area. In: Halvorsen R (ed) The scientific basis for habitat monitoring in Norway—baseline surveys. University of Oslo, Natural History Museum, pp 176–196
Worboys GL, Francis WL, Lockwood M (eds) (2010) Connectivity conservation management: A global guide. Earthscan, London
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effect models and extensions in ecology with R. Springer, New York
Acknowledgments
This study was carried out under the projects “Survey and monitoring of red-listed species” (ARKO, funded by the Norwegian Environment Agency), and “Management of biodiversity and ecosystem services in spatially structured landscapes” (funded by the Norwegian Research Council, grant 208434/F40). We are grateful to Vegar Bakkestuen and Anders Wollan for making available the habitat distribution model, Kristina Bjureke for contributing to fieldwork, and two anonymous reviewers for valuable comments on an earlier version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Dirk Sven Schmeller.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Evju, M., Blumentrath, S., Skarpaas, O. et al. Plant species occurrence in a fragmented grassland landscape: the importance of species traits. Biodivers Conserv 24, 547–561 (2015). https://doi.org/10.1007/s10531-014-0835-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-014-0835-y