Abstract
Biological invasions represent a global threat to biodiversity. Particular attention should be made to the management of alien plants. The species from the Carpobrotus genus are well-known invasive plants native to South Africa, whose detrimental effects on native communities are widely documented. These impacts have become particularly important in terms of their invasive potential on small Mediterranean islands, where they often threaten particular ecosystems and endemic species. We report the experience from the efforts to eradicate this species from the island of Giannutri (Tuscan Archipelago, Italy)—an action included within the EU LIFE project “RESTO CON LIFE”—where the species occurred on about 14,000 m2 of coastal vegetation. The eradication employed an integrated approach with two control methods (manual removal and covering with mulching sheets) and involved mosaic rocky cliff coastal vegetation and disturbed areas with loose soil close to the cliffs. We present here the results obtained from five years of monitoring using permanent plots. Carpobrotus spp. exerted strong impacts on the invaded plant communities, with significant depletion and replacement of native species. Both of the control methods proved effective in the suppression of the mat formed by the invasive species and led to a positive response of native vegetation. Nevertheless, the recovery of native plant communities differed depending on the type of substratum, taking longer on rocky cliffs due to the harsher conditions. Improvements were driven by nitrophilous species in the plots treated with mulching sheets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As indicated by the first Global Assessment of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services (IPBES), biological invasions represent one of the most dramatic threats to biodiversity, contributing substantially to the widespread and accelerated decline in Earth’s biodiversity and associated benefits to people (Díaz et al. 2019). The situation is particularly serious given that the rate of new introductions is expected to increase in the future (Seebens et al. 2017). A critical challenge at this point is to downscale the findings of the IPBES Global assessment and act at national and local scales, where most policy and management decisions affecting biodiversity and ecosystem services are made (Ruckelshaus et al. 2020). Indeed, managing invasive alien species (IAS) is an important modern challenge for nature conservation that is also addressed by the Convention on Biological Diversity (decision VI/23), which incorporated findings of invasion science to define its guiding principles. These rank four main steps in IAS management in a so-called hierarchical approach: prevention, early detection, eradication and control. Within such context, local eradication (i.e., “The complete and permanent removal of all wild populations of an alien plant or animal species from a specific area by means of a time-limited campaign”, see Simberloff et al. 2013) is considered a very useful conservation tool in IAS management and biodiversity restoration, becoming pivotal to the prevention of extinctions and restoration of native insular communities (Ruffino et al. 2015). Different techniques such as manual, chemical, and mechanical methods (including the use of fire) can be used based on the biology of the target organism, the cost, the efficiency, the feasibility, and the presence of non-target organisms. The achievement of a successful eradication can be very difficult (practically unfeasible in some cases), highly demanding on resources and time, and often requires a commitment to long-term management and monitoring, as well as a human-assisted restoration of native plant communities (Ruffino et al. 2015). Studies indicate that island ecosystems represent the ideal target in which to achieve these results (Simberloff et al. 2018).
In the whole Mediterranean basin the species of the genus Carpobrotus N.E. Br. (Aizoaceae)—particularly the species C. acinaciformis (L.) L.Bolus and C. edulis (L.) N.E.Br., and hybrids—are among the most abundant and most investigated invasive alien plants (see Campoy et al. 2018; Lazzaro et al. 2020a, b). Indeed Carpobrotus spp. display their invasive potential in areas with Mediterranean climates, and are particularly invasive in the entire Mediterranean basin (Brundu 2013; Campoy et al. 2018), where they are widespread across small Mediterranean islands (Vilà et al. 2008). In Italy, Carpobrotus spp. are among the most threatening invasive plants on the Tyrrhenian coast (Acosta et al. 2006; Carranza et al. 2010; Santoro et al. 2011) and island ecosystems (Celesti-Grapow et al. 2016), with well-documented impacts on EU habitats of Community Interest, in accordance with Directive no. 92/43/EEC (Lazzaro et al. 2020a). In Tuscany, they are particularly invasive in the islands of the Tuscan Archipelago, including the island of Giannutri (Lazzaro et al. 2014)—the area under study in this work.
The invasion by Carpobrotus spp. is linked to strong negative impacts on the ecology of invaded ecosystems, mainly sand dunes and rocky sea cliffs (Campoy et al. 2018) with well-established deleterious effects on both plant biodiversity (Acosta et al. 2006; Carranza et al. 2010; Santoro et al. 2012; Jucker et al. 2013; Badalamenti et al. 2016) and soil conditions (Santoro et al. 2011; Badalamenti et al. 2016). The depletion of native diversity caused by Carpobrotus spp. is particularly harmful on small Mediterranean islands (Celesti-Grapow et al. 2016; Lazzaro et al. 2016; Brundu 2013; Vilà et al. 2008), where these species exhibit the largest impact on native diversity compared to other invasive alien plant species (Vilà et al. 2006), and consistently demonstrate highly successful invasion across different Mediterranean islands and environments (Traveset et al. 2008). These threats call for particular awareness, particularly considering the contribution of the complex system of archipelagos, islands and islets and their flora to the peculiar plant diversity of the Mediterranean basin, which amounts to about 24,000–25,000 species—accounting for 10% of world plant richness—with rates of endemism often exceeding 10%, and sometimes 20%, of local flora (Brundu 2013).
Across the Mediterranean basin, these species have been targeted by several projects of control, mostly fostered by local stakeholders, but also often linked to the EU LIFE program (see also Campoy et al. 2018). In the coastal dunes of the Mediterranean basin in particular, Carpobrotus spp. is the genus with the largest number of records of control actions (Brundu 2013; Brunel et al. 2013). The control/eradication methods for Carpobrotus spp. include mulching, use of herbicides, and manual removal but generally, the latter is the most used; the use of glyphosate (and chemical compounds in general) usually raises many concerns such that its use is currently restricted or banned by legislation in many European Countries (see Lazzaro et al. 2020b; Fos et al. 2021a, b).
Within this work, we aimed to assess the intervention of control of Carpobrotus spp. conducted within the EU LIFE project LIFE13 NAT/IT/000471 “Island conservation in Tuscany, restoring habitat not only for birds”—RESTO CON LIFE, on the island of Giannutri. The control has been implemented through the integration of two techniques of removal: manual removal and covering with mulching sheets. Accordingly, we aimed to (i) verify the response of Carpobrotus spp. and the re-growth of the native vegetation after the interventions, testing whether the native species richness and diversity increased after the intervention and (ii) evaluate how the native communities recovered after the eradication of Carpobrotus spp. in terms of species composition. Toward these aims we monitored a series of vegetation plots for five years, starting from a first survey (in 2015) prior to the main intervention and for the following four years (2016–2019).
Methods
Study area
The island of Giannutri (Lat 42.252801° N, Lon 11.100882° E) has an area of 232 hectares, a coastal development of 13.7 km, and is the southernmost and easternmost island of the Tuscan Archipelago. It is fully contained within the Tuscan Archipelago National Park and is framed within the Natura 2000 Network as a Special Area of Conservation (SAC) according to Directive 92/43/EEC "Habitat" (site code IT51A0024).
The island is totally calcareous (dolomite of Monte Argentario) and has a typical Mediterranean mesothermal, semi-arid climate with a water deficit during the late spring–summer period (Foggi et al. 2011). The vegetation of the island is mainly represented by Mediterranean shrublands (ranging from high to low and sparse formations which cover more than 80% of the total surface area) and coastal vegetation of rocky cliffs (covering about 15% of the total surface area); woodlands are poorly represented (less than 1% of the total surface area), as are anthropized habitats (Foggi et al. 2011). The rocky cliff coastal vegetation is of particular interest for this study, being the main target for invasion by Carpobrotus spp. It hosts a mosaic of habitats of conservation interest according to Directive 92/43/EEC "Habitat" including the habitat of vegetated sea cliffs of the Mediterranean coasts with endemic Limonium spp. (habitat code 1240 according to Dir. 92/43/EEC), and including the important endemic Limonium sommerianum Fiori, see also Foggi et al. (2015), the habitat of Salicornia and other annuals colonizing mud and sand (habitat code 1310, particularly in the subtypes of Mediterranean halo-nitrophilous pioneer communities of the Frankenion pulverulentae) and elements of the habitat of Low formations of Euphorbia close to cliffs (hab. code 5320). In particular, monitoring has been carried out in the promontory of Punta San Francesco on the East side of the island, where Carpobrotus spp. was invading two main types of habitats: the rocky cliff coastal mosaic and some disturbed areas with loose soil close to the cliffs that were once used as an airport runway.
The eradication of Carpobrotus spp.
The present study analyzed the changes in Carpobrotus spp. and native species presence and abundance during the action of eradication of this invasive species conducted within the EU LIFE project LIFE13 NAT/IT/000471 “Island conservation in Tuscany, restoring habitat not only for birds”—RESTO CON LIFE, on the island of Giannutri. We conducted a detailed mapping of the spread of Carpobrotus spp. on Giannutri at the beginning of the project through the interpretation of aerial photos and surveys on the island, recording an initial distribution of this species on about 14,000 m2 of the island. The eradication of Carpobrotus spp. has been implemented through the integration of two techniques of removal: manual removal and covering with mulching sheets. Manual removal consisted of the removal of all the plants, including their root systems, and was adopted in areas where the invasive alien species cover was not reaching high values (generally up to 75%) and thus there was also a significant cover of native species. The use of mulching sheets consisted of covering the entire ground area that was covered by a thick mat of Carpobrotus spp. with mulch sheets, made of woven plastic fiber (Landscape fabric, 105 g/m2) that favor evaporation and consequently the drying of covered plants. This technique is also known as tarping or solarization. It was adopted in areas where the cover of this species exceeded 75% (these areas were, in fact, represented almost entirely by a cover close to 100%). It should be noted that approximately 90% of the Carpobrotus spp. on the island was treated with mulching sheets, while manual removal was adopted only in a limited number of areas. The main intervention was carried out in 2016, with a first phase in May (laying of mulching sheets and implementing the manual removal) and a second phase in September to remove the mulching sheets. During the first years following the main intervention (from 2017 to 2019) eventual seedlings and resprouts were manually removed and, at the time of writing, continue to be removed yearly. Overall, the intervention had a cost of 140 man-days. Further technical details on the methods adopted are available in the executive project for the eradication (https://www.restoconlife.eu/wordpress/wp-content/uploads/2015/11/Interventi-di-riqualificazione-della-vegetazione-nell%E2%80%99Isola-di-Giannutri.pdf).
Data collection
The survey of the vegetation in the area of Punta San Francesco took into consideration two different factors in a stratified random design. We differentiated two different substrate types hosting two different plant communities (i.e., rocky cliffs and loose soils) and three different treatments according to Carpobrotus spp. cover, following the plan of the interventions described above. The treatment levels surveyed were (1) covering with mulching sheets in areas with a cover of Carpobrotus spp. close to 100%, both in rocky cliffs and loose soils, (2) manual control of Carpobrotus spp. in areas with a lower cover of Carpobrotus spp. (i.e., between 30 and 75%) and with a relevant presence of native species (a situation present only on rocky cliffs), and (3) control with no presence of Carpobrotus spp. This resulted in a total of 42 plots of 2 × 2 m, 18 on rocky cliffs (6 replicates × 3 treatments) and 24 on loose soils (12 replicates × 2 treatments). The higher number of replicates in loose soils is linked to the greater area and higher variability of this type of surface in the region of study. Each plot was georeferenced, and a vegetation sampling was carried out during the vegetative period (May–June) for five years: before the eradication (2015) and for the four subsequent years (2016 to 2019) to observe the evolution of the vegetation. In each plot we collected information on the cover of Carpobrotus spp. and of each native species using a percentage scale. The repeated sampling of these 42 areas over 5 years resulted in the survey of 210 plots.
Statistical analyses
The effect of the interventions on Carpobrotus spp. and on the recovery of the native vegetation was evaluated using a series of Repeated Measurement ANOVA type models via Permutational Analysis of Variance (PERMANOVA) using 999 permutations. In the univariate case, the PERMANOVA F-value is calculated from a Euclidean distance matrix and corresponds to the classical F-value, whereas the P value evaluation via a permutation approach is free from many of the assumptions of parametric statistics (Anderson 2001). In all the models, permutations are restricted within groups represented by plot identity, thus accounting for the correlation of the observations. We studied the changes in the cover of Carpobrotus spp. and of all the other native species, using the cover of the species as the response variable, while time, treatment, and species (Carpobrotus spp. versus all the native species pooled together) were used as explanatory variables in a full factorial design. Similarly, we tested whether the native species richness and diversity, expressed as the Shannon H’ diversity index, changed after the interventions using time and treatment as fixed effect explanatory variables and the plot identity as a random effect factor. Moreover, we studied the variation of the relative cover of nitrophilous species to assess their role in the plant community recovery, using the same framework described above. Nitrophilous species were defined as those with an Ellenberg's ecological indicator value for eutrophication (Pignatti 2005; Ellenberg 2009) above 6. Due to the differences in the two types of plant communities, we conducted all the analyses separately for loose soils and rocky cliffs communities.
We studied the changes in the species composition of plots using multivariate analyses aiming to verify the effects of the different treatments. The analyses included plots in all soil types and treatments between 2015 and 2019 but considered only plots with at least one species (total of 178 plots: 21 plots had no species in 2016 [the year of the main interventions], eight had no species in 2017, while three had none in 2018). Plot species composition dynamics over time were analyzed using a non-metric multidimensional scaling (NMDS) analysis based on Bray–Curtis dissimilarities calculated on abundance data (expressed as percentages). Moreover, we evaluated the extent and divergence of species composition variations according to time and treatment in a Principal Response Curve (PRC) analysis (ter Braak and Smilauer 2012). In this analysis, time was treated as a categorical variable and was used as a covariate. The significance of the effect of the treatments on the species composition was assessed with a permutation analysis using 9999 permutations due to the hierarchical structure of the data and allowing freely exchangeable permutations on the whole-plot level, and no permutation at the split-plot level. Given the differences in the two types of plant communities, we conducted the PRC analyses separately for loose soils and rocky cliffs communities.
The PERMANOVAs and the NMDS analysis were conducted in R environment (R version 4.1.0) using the vegan package version 2.5-7 (Oksanen et al. 2020); plots were drawn using ggplot2 package version 3.3.3 (Wickham 2016). PRC analyses were performed using Canoco 5 for Windows vers. 5.12 (ter Braak and Smilauer 2012).
Results
The sampling from 2015 to 2019 identified 44 species other than the invasive alien species Carpobrotus spp.; Papaver somniferum L. was the only other alien species (but it is considered an archaeophyte), while Portulaca oleracea L. is considered cryptogenic (i.e., with uncertain native status). The communities growing on rocky cliffs and loose soils were quite different, both in terms of composition and the total number of species retrieved (i.e., the γ-diversity); they shared only 11 species out of 44 recorded, while nine were exclusive to rocky cliffs (from a total of 20 species) and 24 were exclusive to loose soils (from a total of 35 species) (see Appendix S1).
The control of Carpobrotus spp. led to substantial suppression of the invasive species, subsequently resulting in a rapid positive response of native vegetation. Treatments induced significant variation over the five years of the survey on both rocky cliffs and loose soils (interaction terms Treatment:Year:Species were significant in both cases, with P value < 0.001, see Table 1). Particularly, as shown in Fig. 1A and B, Carpobrotus spp. cover dropped to zero after 2015 in both treatments (manual removal and mulching) in both substrata, while the recovery of native species cover proceeded differentially in loose soils and rocky cliffs and according to the control method. In loose soils, the recovery of native species after the removal of mulching sheets (in autumn 2016) was considerably faster than in rocky cliffs, in which the cover of native plants took until 2019 to reach values comparable with the controls. In case of manual removal on rocky cliffs the recovery of cover values was faster.
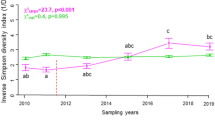
Native species richness and diversity (H’) displayed similar trends and were both significantly affected by treatments over the five years of survey (with a significant interaction term Treatment:Year P value < 0.001, see Table 1). In treatment plots, native richness and diversity were close to zero in 2015 and increased after Carpobrotus spp. removal in 2016. The recovery of native communities up to the levels of control plots was faster in plots treated with mulching sheets on loose soil and with manual removal on rocky cliffs, while in the plots treated with mulching sheets on rocky cliffs, recovery occurred only in the last two years of the survey (Fig. 2A–D).
Variation of native species richness in A rocky cliff and B loose soil plots, native species diversity (Shannon H’) in C rocky cliff and D loose soil plots, and relative cover of nitrophilous species (i.e., species with Ellenberg N value > 6) in E rocky cliff and F loose soil plots, according to time (years 2015–2019) and treatment (solid line = control, dashed line = manual removal (only on rocky cliffs) and dotted line = covering with mulching sheets)
The relative cover of nitrophilous species also varied significantly due to treatment and time (interaction term Treatment:Year significant with a P value < 0.05 on rocky cliffs and a P value < 0.001 on loose soils, see Table 1). Nitrophilous species’ contribution to the local communities increased dramatically after the interventions, particularly in plots treated with mulching sheets, but also to a lesser extent in those treated with manual removal on rocky cliffs.
The NMDS analysis (stress = 0.090, non-metric fit R2 = 0.992, linear fit R2 = 0.969, see Fig. 3) highlighted a well-defined differentiation (as expected) between plant communities growing on loose soils and those growing on rocky slopes (laying on axis NMDS1). Moreover, the ordination highlighted the strong changes in plant composition after the Carpobrotus spp. removal (moving on both NMDS2 and NMDS1). The plot monitored before the interventions (year 2015) lay on the left upper part of the graph, with plots on loose soils and rocky cliffs presenting high Carpobrotus spp. cover (that will be treated with mulching sheets) almost overlapping and plots on rocky cliffs with low Carpobrotus spp. cover positioned closer to the controls. After the interventions, the plots on loose soils migrate to the right part of the ordination close to the control plots on loose soils, while those on rocky cliffs move close to the respective control in the bottom left part of the graph.
Non-metric multidimensional scaling (NMDS) analysis based on Bray–Curtis dissimilarities and run on the compositional data on a total of 178 plots between 2015 and 2019. Full squares = control plots on rocky cliffs; full triangles = manual removal plots on rocky cliffs; full circles = mulching sheets plots on rocky cliffs; empty squares = control plots on loose soils; empty circles = mulching sheets plots on loose soils. Plant species names were shortened to enhance readability; please see Appendix S1 for plant full names
The PRC analysis of the composition of survey plots during the treatments are consistent with the above-mentioned trend of recovery of the communities towards the communities of the control plots. Indeed, the analyses highlighted significant effects of treatment over time in both rocky cliffs (pseudo-F = 3.3, P value < 0.001) and loose soils (pseudo-F = 12.5, P value < 0.001) communities. In both cases it is possible to note a trend of convergence of the treated plots with the control ones over time (Fig. 4A, B).
Principal response curves (PRC) showing the effect of the different treatments on the plant species composition of the plots (only the 10 best fitting species are shown) in A rocky cliff and B loose soil plots. Species weights indicate the strength of the trend for each species and indicate a negative association or inverse of the trends depicted. The product of species weight and the canonical coefficient (y-axis) for a given treatment and time equals the natural log change in density of that species relative to the control. The PRC analysis over all dates was significant (P < 0.001) based on an F-type permutation test. Solid line = control treatment (used as reference to assess variation in other treatments), dotted line = manual removal (only on rocky cliffs), dashed line = covering with mulching sheets
Discussion
Within this work, we aimed to test the effectiveness of the control techniques adopted within the EU LIFE project "RESTO CON LIFE" for the eradication of Carpobrotus spp. on the island of Giannutri and to monitor the recovery of native species after the removal of this invasive species. Our results provide important evidence of the effectiveness of both manual removal and covering with mulching sheets in the suppression of the invasive species, subsequently leading to the recovery of native plant communities.
The first pivotal result of the monitoring is represented by the evidence of the strong impacts exerted by Carpobrotus spp. on the invaded plant communities, as showed by the difference in native species richness and diversity among the control plots and those with a high cover of Carpobrotus spp. Our results are consistent with those coming from several other case studies where Carpobrotus spp. invasion resulted in a loss in native species diversity (Vilà et al. 2006; Santoro et al. 2012; Fried et al. 2014; Andreu et al. 2010). These trends in native species depletion and replacement recorded at Giannutri are comparable to those found in the nearby Giglio Island, where a similar action is currently ongoing. Here, the first monitoring data showed that Carpobrotus spp. induces changes in the composition and diversity of invaded rocky coastal communities, with negative impacts on species richness, diversity and abundance of native species (see Mugnai et al. 2022). These data confirm that Carpobrotus spp. invasion may act throughout the process of replacement and exclusion of native species, rather than coexistence (as already pointed out in Andreu et al. 2010). These impacts are particularly noteworthy since the typical flora of rocky habitats both at Giglio and Giannutri islands host high conservation value species such as the endemic Limonium sommerianum. In fact, small Mediterranean islands like Giglio and Giannutri may constitute key ecological systems and current “refugia” to ensure the long-term preservation of coastal plant biodiversity. However, as demonstrated here, these are threatened by drivers such as climate warming and aridification, sea-level rise, and biological invasions (Medail 2017). The interaction between biological invasions and climate change is particularly worrying in the case of Carpobrotus spp., because these species, as well as other succulents, are considered to number among those that are likely to expand the most as a result of global warming (Campoy et al. 2021), further threatening Mediterranean island ecosystems. Indeed, Campoy et al. (2021) found increased growth and physiological performances in Carpobrotus edulis under experimental conditions of increased temperatures and reduced rainfall levels. Similarly, increased potential distribution of Opuntia ficus-indica has been predicted due to increases in air temperature and reduction in rainfall at Caatinga Biome, Northeast Brazil (Cavalcante et al. 2020). This phenomenon also appears to be acting in our study area, although further studies are needed to explore the evidence of this particular link.
Fortunately, the control of Carpobrotus spp. is not an unknown territory. These species have been targeted by several projects of control, mostly fostered by local stakeholders, but also often linked to the EU LIFE program (see also Campoy et al. 2018). Particularly in Italy, several actions similar to those described here have been carried out or are still in progress in other LIFE projects in small Italian islands (e.g., Pelagic Islands in Sicily within LIFE11 + NAT/IT/000093 “Pelagic Birds” (Badalamenti et al. 2016); Pontine Islands in Lazio within LIFE 14 NAT/IT/000544 “PonDerat” (Celesti-Grapow et al. 2017); Tavolara Island in Sardinia within LIFE19 NAT/MT/000982 “PanPuffinus” and Giglio Island in Tuscany within LIFE18 NAT/IT/000828 “LETSGO GIGLIO” (Mugnai et al. 2022)). In addition, several studies in recent years dealt with monitoring the intervention of local control or eradication of this invasive species, focusing on the effectiveness of control techniques or the recovery of native species (or on both these aspects). Nevertheless, our data appear particularly interesting, considering that while most of the previous studies dealt with invasion in sand dune ecosystems (Magnoli et al. 2013; Lazzaro et al. 2020b; Souza-Alonso et al. 2019; Fos et al. 2021a, b) controlled by using chemical treatment (Lazzaro et al. 2020b; Fos et al. 2021a, b) or manual removal (Magnoli et al. 2013; Buisson et al. 2021), few dealt with rocky habitats (Buisson et al. 2021), and we could only find one of study that included a robust evaluation on the use of mulching sheets as an effective control technique (Fos et al. 2021b).
Indeed, even if covering with mulching sheets is a technique already known for the control of Carpobrotus spp., in the past it was generally reported with negative experiences (Campoy et al. 2018). However, Fos et al. (2021b) has recently confirmed that tarping was an effective method for Carpobrotus spp. eradication, but in light of concerns raised regarding its costs, they considered this technique suitable in only a restricted set of situations, such as where invasion is still in its early stages or in small-scale interventions. Our data offer further confirmation that this technique has the potential to achieve totally effective removal of this invasive species. As reported by Fos et al. (2021b), covering time is quite an important factor in determining the effectiveness of the methodology and, in our case, we retained the covering for four months. As to its suitability for important invaded surfaces, it should be noted that Fos et al. (2021b) reported that solarization is an unrealistic option for eradicating invasive plant species over large areas, which is further supported by evidence from areas invaded by grasses and forbs (Holl et al. 2014; Orr et al. 2019) on sand dunes. In our case, covering with mulching sheets was used as the main technique on about 90% of the area invaded by Carpobrotus spp. (corresponding to approximately 12,500 m2 of the 14,000 m2 total), and on surfaces with a significant slope inclination (see Fig. 5). A cost evaluation by Holl et al. (2014) compared the cost of herbiciding (US$1440/ha) versus tarping (US$14,040/ha). However, these costs are not applicable to situations such as the one presented here, in which costs are increased by the terrain asperity—which also increases costs for manual removal or use of herbicides. We estimate a cost for the whole intervention of about 7 € per square meter, which appears comparable to previous experiences in the Tuscan Archipelago at the island of Pianosa, where the removal of Carpobrotus spp. has been achieved with the use of chemical treatments that were estimated to cost 9.4 € per m2 (Campoy et al. 2018). The whole cost of the intervention on Giannutri was approximately 100,000 € for the removal, which appears affordable with the use of specific funds such as those provided under the EU LIFE program. Moreover, tarping with mulching sheets removes the necessity of using glyphosate and other chemical treatments. In fact, even though chemical control has been shown to be efficient—particularly using glyphosate—and with no significant long-term negative effects on the recovery of native plant communities (Lazzaro et al. 2020b; Fos et al. 2021b), the approach still raises important concerns linked to the risks for human health and the ecosystem, as well as ethical concerns on its use in protected areas. Not least, the use of herbicides meets considerable difficulties in obtaining the necessary authorizations. For example, in the European Union the Dir. 2009/128/EC on the sustainable use of pesticides prescribes the reduction of pesticide use or risks in specific areas, including protected areas and Natura 2000 sites, and Italian laws adopting the Directive have imposed a ban on the use of these products in Natura 2000 sites unless specific exemptions are obtained, which is not always possible.
Some images of the interventions of Carpobrotus spp. removal at Giannutri Island. Mulching sheets on A flat and B steep rocky cliffs; C Carpobrotus spp. under the sheets a few weeks after the covering; D images of the mats at the moment of the mulching sheets removal; E colonization of the loose soils area in 2017 (note individuals of Ecballium elaterium (L.) A. Rich. in the foreground and Carduus cephalanthus Viv. in the background)
Besides the effectiveness of the method for the removal of the invasive species per se, another pivotal issue is the recovery of native vegetation (Buisson et al. 2021). Indeed, the optimal result would be a rapid convergence of the vegetation composition of treated areas toward that of its immediate surroundings. According to Tu et al. (2001), covering the soil with plastic sheets is not a recommended method to control major infestations, since it may cause significant physical, chemical, and biological changes in the soil that can last for several years. Nevertheless, in our case we observed a clear recovery of native vegetation, with several native species able to colonize the treated areas in the medium period (2–3 years), with colonization occurring faster on loose soils. Following the removal of the mulching sheets from their experimental plots, Fos et al. (2021b) registered a rapid colonization from six different native species and three invasive species (including Carpobrotus spp.), which also highlights that tarping did not kill all the seeds present in the sand underneath the Carpobrotus spp. cover. As noted by Fos et al. (2021b), seedling emergence after the removal of the mulching sheets is common and was often recorded across the whole intervention area in our study. Indeed, specific actions of monitoring and seedling removal are foreseen for several years. One important limitation in our data is the lack of a specific quantification of the seedling emergence during the years after the interventions. While Carpobrotus spp. seedlings were recorded in the surveyed plots after the interventions, their abundance was evaluated by assessing their percentage cover. It is likely that an ad-hoc seedling sampling design would have been very useful, particularly for the evaluation of the effectiveness of mulching sheets on the depletion of the Carpobrotus spp. seed bank. We note here that, initially, the seedlings were mainly recorded on the rocky cliffs subjected to manual removal (in one single plot in 2016, 2018 and 2019, and in two plots in 2017), while they were also recorded on rocky cliffs treated with mulching sheets only in the latter years and to a lesser extent (two plots in 2018). No seedlings were recorded on loose soils during the monitoring, probably due to the fact that these were removed by the workers at least once a year.
We also note that a contribution to the spreading of Carpobrotus spp. seeds on Giannutri may be attributed to the presence of the alien species of European rabbit (Oryctolagus cuniculus) on the island. The facilitation among these IAS, resulting in invasional meltdown processes, has been already reported on offshore islands in southeast France (Bourgeois et al. 2005). Moreover, the establishment of C. edulis, in terms of germination and early growth, is increased by scarification of seeds following passage through the European rabbit intestines, suggesting that the rabbits may have potential implications for plant establishment (Novoa et al. 2012). It should also be noted that the European rabbit population on Giannutri island was severely contracted in the years following the interventions described here, and an appropriate study could shed light on the real characteristics of its population and the eventual interaction with Carpobrotus spp.
As to native species recovery, the NMDS and PRC analyses highlighted that plant communities in the treated plots converged quite rapidly towards the relative controls, but with some differences among the three different conditions: loose soils treated with mulching sheets, rocky cliffs with total Carpobrotus spp. cover treated with mulching sheets and rocky cliffs with low Carpobrotus spp. cover treated with manual removal. This latter case was, in a sense, the simplest; here, native species were already present and could easily expand after the removal of Carpobrotus spp. mats, such that the plots could rapidly become very similar to control plots on rocky cliffs. In the other two cases, the analysis of the recovery of the vegetation should take into account three main factors: the substantial absence of native species before the interventions, the use of mulching sheets, and the important layer of litter left in place after the removal of the mulching sheets. As to this latter factor, unfortunately, we did not collect precise data on the litter of Carpobrotus spp. present in the plots after the eradication. However, it should be noted that, while no litter was present in control plots and in plots subjected to manual removal, a thick layer of litter was persistent (albeit slowly reducing during the years of monitoring) in most of the plots subjected to covering with mulching sheets. Indeed, only in plots on the rocky cliffs—and, significantly, only in 2019—did the layer started to disappear (locally removed by the wind), allowing native species to colonize these areas. On loose soil plots, the colonization of litter and soils had already started in 2017 and took place over the litter layer. It should be noted that in all the intervention plots we observed a sharp increase in the relative cover of nitrophilous species. Indeed, one of the well-known effects of litter accumulation by Carpobrotus spp. is the significant modification of soil parameters, with a net increase in nitrogen and organic matter content and a decrease in soil pH (Santoro et al. 2011). Consequently, it has been found that Carpobrotus spp. may favor the replacement of typical native dune plants by ruderal nitrophilous species through soil nutrient enrichment (Novoa et al. 2013; Fried et al. 2014; Malavasi et al. 2016). In sand dune communities (as evidenced in Novoa et al. 2013), the effect of litter accumulation is particularly important, as it may severely restrict the colonization of treated areas by less competitive species, actually preventing or inhibiting a return to the pre-invasion situation. Indeed, several previous eradication experiences showed that the effects of invasive plants on community composition or ecosystem properties may continue after their removal, thus exerting a “legacy” that persists over an ensuing time period, and that to achieve truly successfully restoration goals eradication may need to be accompanied by strategies to overcome these legacies (Corbin and D’Antonio 2012). Accordingly, Novoa et al. (2012, 2013) advise against leaving fresh or dry Carpobrotus spp. litter on restored areas, although it should be noted that the complete removal of Carpobrotus spp. and its litter may lead to massive soil erosion, as reported by Chenot et al (2018), requiring solutions to mitigate the issue. We also note that on large areas, this would mean a further and substantial increase in the costs of eradication and/or control interventions. As to the role of litter in our case, we observed that nitrophilous species do indeed appear to drive vegetation recovery, particularly in the plots treated with mulching sheets. For instance, looking at the NMDs ordination plots, plots treated with mulching sheets on rocky cliffs in 2018 are almost closer to those on loose soils than to the respective controls on rocky cliffs, and only in 2019 moved towards the latter ones. Nevertheless, “legacy effects” on rocky areas seem to be rapidly decreasing due to the rapid reduction of the litter layer, and the return to a more natural condition of bare rock substratum on which natural communities usually grow. Nonetheless, these effects may be longer-lasting on loose soil areas and delay the full recovery of native plant communities for a considerable period.
In conclusion, we report the experience from the eradication of Carpobrotus spp. from the Island of Giannutri (Tuscan Archipelago, Italy), an action within the EU LIFE project "RESTO CON LIFE". The monitoring data, spanning from a year before to four years after the interventions, showed a rapid recovery of the native vegetation, but mainly driven in these first stages by nitrophilous species. As already expressed by Buisson et al. (2021), it is very important to collect and provide data for a long timespan after eradication interventions. In our case, we are providing a medium time (four years after the interventions), but it will be very important to continue to survey the evolution of native vegetation for a long time, even with a lower frequency of sampling (i.e., once every few years), to provide insight into the extent of further colonization by native species and natural vegetation recovery phases. Our data are also important as they provide information on significant extent once invaded by Carpobrotus spp.
References
Acosta A, Izzi CF, Stanisci A (2006) Comparison of native and alien plant traits in Mediterranean coastal dunes. Community Ecol 7:35–41. https://doi.org/10.1556/comec.7.2006.1.4
Anderson MJ (2001) Permutation tests for univariate or multivariate analysis of variance and regression. Can J Fish Aquat Sci 58:626–639. https://doi.org/10.1139/f01-004
Andreu J, Manzano-Piedras E, Bartomeus I et al (2010) Vegetation response after removal of the invasive carpobrotus hybrid complex in Andalucia, Spain. Ecol Restor 28:440–448. https://doi.org/10.3368/er.28.4.440
Badalamenti E, Gristina L, Laudicina VA et al (2016) The impact of Carpobrotus cfr. acinaciformis (L.) L. Bolus on soil nutrients, microbial communities structure and native plant communities in Mediterranean ecosystems. Plant Soil 409:19–34. https://doi.org/10.1007/s11104-016-2924-z
Bourgeois K, Suehs CM, Vidal E, Médail F (2005) Invasional meltdown potential: facilitation between introduced plants and mammals on French Mediterranean islands. Ecoscience 12:248–256
Brundu G (2013) Invasive alien plants in protected areas in mediterranean islands: knowledge gaps and main threats. In: Foxcroft LC, Pyšek P, Richardson DM, Genovesi P (eds) Plant Invasions in Protected Areas: Patterns, Problems and Challenges. Springer, Netherlands, Dordrecht, pp 395–422
Brunel S, Brundu G, Fried G (2013) Eradication and control of invasive alien plants in the Mediterranean Basin: towards better coordination to enhance existing initiatives. EPPO Bull 43:290–308. https://doi.org/10.1111/epp.12041
Buisson E, Braschi J, Chenot-Lescure J et al (2021) Native plant community recovery after carpobrotus (ice plant) removal on an island—results of a 10-year project. Appl Veg Sci 24:e12524
Campoy JG, Acosta ATR, Affre L et al (2018) Monographs of invasive plants in Europe: carpobrotus. Bot Lett 165:440–475. https://doi.org/10.1080/23818107.2018.1487884
Campoy GJ, Lema M, Fenollosa E et al (2021) Functional responses to climate change may increase invasive potential of Carpobrotus edulis. Am J Bot 108:1902–1916. https://doi.org/10.1002/ajb2.1745
Carranza ML, Carboni M, Feola S, Acosta ATR (2010) Landscape-scale patterns of alien plant species on coastal dunes: the case of iceplant in central Italy. Appl Veg Sci 13:135–145
Celesti-Grapow L, Bassi L, Brundu G et al (2016) Plant invasions on small Mediterranean islands: an overview. Plant Biosyst - Int J Deal Asp Plant Biol 150:1119–1133. https://doi.org/10.1080/11263504.2016.1218974
Celesti-Grapow L, Abbate G, Baccetti N et al (2017) Control of invasive species for the conservation of biodiversity in Mediterranean islands. The LIFE PonDerat project in the Pontine Archipelago, Italy. Plant Biosyst - Int J Deal Asp Plant Biol 151:795–799. https://doi.org/10.1080/11263504.2017.1353553
Chenot J, Affre L, Gros R, Dubois L, Malecki S, Passetti A, Aboucaya A, Buisson E (2018) Eradication of invasive Carpobrotus sp.: effects on soil and vegetation. Restor Ecol 26:106–113. https://doi.org/10.1111/rec.12538
Corbin JD, D’Antonio CM (2012) Gone but not forgotten? Invasive plants’ legacies on community and ecosystem properties. Invasive Plant Sci Manag 5:117–124. https://doi.org/10.1614/IPSM-D-11-00005.1
de Cavalcante AMB, Fernandes PHC, da Silva EM (2020) Opuntia ficus-indica (L.) Mill. and climate change: an analysis in the light of modeling potential distribution of species in the caatinga biome. Rev Bras Meteorol 35:375–385. https://doi.org/10.1590/0102-7786353001
Díaz SM, Settele J, Brondízio E, et al (2019) The global assessment report on biodiversity and ecosystem services: Summary for policy makers. Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services
Ellenberg HH (2009) Vegetation ecology of central Europe, 4th edn. Cambridge University Press, Cambridge
Foggi B, Cioffi V, Ferretti G (2011) La vegetazione dell’Isola di Giannutri (Arcipelago Toscano, Grosseto). Fitosociologia 48(2):23–44
Foggi B, Viciani D, Baldini RM et al (2015) Conservation assessment of the endemic plants of the Tuscan Archipelago, Italy. Oryx 49:118–126. https://doi.org/10.1017/S0030605313000288
Fos M, Sanz B, Sanchis E (2021a) The use of glyphosate for Carpobrotus eradication in sand dune ecosystems: evaluation of the potential effects on the reintroduction of native plants. Plant Biosyst- Int J Deal Asp Plant Biol 156:1–10
Fos M, Sanz B, Sanchis E (2021b) Carpobrotus management in a Mediterranean Sand Dune ecosystem: minimum effective glyphosate dose and an evaluation of tarping. J Ecol Eng 22:57–66. https://doi.org/10.12911/22998993/138871
Fried G, Laitung B, Pierre C et al (2014) Impact of invasive plants in Mediterranean habitats: disentangling the effects of characteristics of invaders and recipient communities. Biol Invasions 16:1639–1658. https://doi.org/10.1007/s10530-013-0597-6
Holl KD, Howard EA, Brown TM et al (2014) Efficacy of exotic control strategies for restoring coastal prairie grasses. Invasive Plant Sci Manag 7:590–598. https://doi.org/10.1614/IPSM-D-14-00031.1
Jucker T, Carboni M, Acosta ATR (2013) Going beyond taxonomic diversity: deconstructing biodiversity patterns reveals the true cost of iceplant invasion. Divers Distrib 19:1566–1577. https://doi.org/10.1111/ddi.12124
Lazzaro L, Ferretti G, Giuliani C, Foggi B (2014) A checklist of the alien flora of the Tuscan Archipelago (Italy). Webbia 69:157–176. https://doi.org/10.1080/00837792.2014.907981
Lazzaro L, Foggi B, Ferretti G, Brundu G (2016) Priority invasive alien plants in the Tuscan Archipelago (Italy): comparing the EPPO prioritization scheme with the Australian WRA. Biol Invasions 18:1317–1333. https://doi.org/10.1007/s10530-016-1069-6
Lazzaro L, Bolpagni R, Buffa G et al (2020a) Impact of invasive alien plants on native plant communities and Natura 2000 habitats: state of the art, gap analysis and perspectives in Italy. J Environ Manage 274:111140. https://doi.org/10.1016/j.jenvman.2020.111140
Lazzaro L, Tondini E, Lombardi L, Giunti M (2020b) The eradication of Carpobrotus spp. in the sand-dune ecosystem at Sterpaia (Italy, Tuscany): indications from a successful experience. Biologia (Bratisl) 75(2):199–208. https://doi.org/10.2478/s11756-019-00391-z
Magnoli SM, Kleinhesselink AR, Cushman JH (2013) Responses to invasion and invader removal differ between native and exotic plant groups in a coastal dune. Oecologia 173:1521–1530. https://doi.org/10.1007/s00442-013-2725-5
Malavasi M, Santoro R, Cutini M et al (2016) The impact of human pressure on landscape patterns and plant species richness in Mediterranean coastal dunes. Plant Biosyst Int J Deal Asp Plant Biol 150:73–82. https://doi.org/10.1080/11263504.2014.913730
Médail F (2017) The specific vulnerability of plant biodiversity and vegetation on Mediterranean islands in the face of global change. Reg Environ Change 17:1775–1790. https://doi.org/10.1007/s10113-017-1123-7
Mugnai M, Benesperi R, Viciani D et al (2022) Impacts of the Invasive Alien Carpobrotus spp. on Coastal Habitats on a Mediterranean Island (Giglio Island, Central Italy). Plants 11:2802. https://doi.org/10.3390/plants11202802
Novoa A, González L, Moravcová L, Pyšek P (2012) Effects of soil characteristics, allelopathy and frugivory on establishment of the invasive plant carpobrotus edulis and a co-occuring native. Malcolmia Littorea. PLOS One 7:e53166. https://doi.org/10.1371/journal.pone.0053166
Novoa A, González L, Moravcová L, Pyšek P (2013) Constraints to native plant species establishment in coastal dune communities invaded by carpobrotus edulis: implications for restoration. Biol Conserv 164:1–9. https://doi.org/10.1016/j.biocon.2013.04.008
Oksanen J, Blanchet FG, Friendly M, et al (2020) vegan: Community Ecology Package
Orr MR, Reuter RJ, Murphy SJ (2019) Solarization to control downy brome (Bromus tectorum) for small-scale ecological restoration. Invasive Plant Sci Manag 12:112–119. https://doi.org/10.1017/inp.2019.8
Pignatti S (2005) Valori di bioindicazione delle piante vascolari della flora d’Italia. Braun-Blanquetia 39:1–97
Ruckelshaus MH, Jackson ST, Mooney HA et al (2020) The IPBES global assessment: pathways to action. Trends Ecol Evol 35:407–414. https://doi.org/10.1016/j.tree.2020.01.009
Ruffino L, Krebs E, Passetti A et al (2015) Eradications as scientific experiments: progress in simultaneous eradications of two major invasive taxa from a Mediterranean island. Pest Manag Sci 71:189–198. https://doi.org/10.1002/ps.3786
Santoro R, Jucker T, Carranza M, Acosta A (2011) Assessing the effects of Carpobrotus invasion on coastal dune soils. Does the nature of the invaded habitat matter? Community Ecol 12:234–240. https://doi.org/10.1556/comec.12.2011.2.12
Santoro R, Carboni M, Carranza ML, Acosta ATR (2012) Focal species diversity patterns can provide diagnostic information on plant invasions. J Nat Conserv 20:85–91. https://doi.org/10.1016/j.jnc.2011.08.003
Seebens H, Blackburn TM, Dyer EE et al (2017) No saturation in the accumulation of alien species worldwide. Nat Commun 8:14435. https://doi.org/10.1038/ncomms14435
Simberloff D, Martin J-L, Genovesi P et al (2013) Impacts of biological invasions: What’s what and the way forward. Trends Ecol Evol 28:58–66. https://doi.org/10.1016/j.tree.2012.07.013
Simberloff D, Keitt B, Will D et al (2018) Yes We Can! exciting progress and prospects for controlling invasives on Islands and Beyond. West North Am Nat 78:942. https://doi.org/10.3398/064.078.0431
Souza-Alonso P, Guisande-Collazo A, Lechuga-Lago Y, González L (2019) The necessity of surveillance: medium-term viability of Carpobrotus edulis propagules after plant fragmentation. Plant Biosyst- Int J Deal Asp Plant Biol 153:736–739
ter Braak CJ, Smilauer P (2012) Canoco reference manual and user’s guide: software for ordination, version 5.0
Traveset A, Brundu G, Carta L et al (2008) Consistent performance of invasive plant species within and among islands of the Mediterranean basin. Biol Invasions 10:847–858. https://doi.org/10.1007/s10530-008-9245-y
Tu M, Hurd C, Randall JM (2001) Weed control methods handbook: tools & techniques for use in natural areas
Vilà M, Tessier M, Suehs CM et al (2006) Local and regional assessments of the impacts of plant invaders on vegetation structure and soil properties of Mediterranean islands. J Biogeogr 33:853–861. https://doi.org/10.1111/j.1365-2699.2005.01430.x
Vilà M, Siamantziouras A-SD, Brundu G et al (2008) Widespread resistance of Mediterranean island ecosystems to the establishment of three alien species. Divers Distrib 14:839–851. https://doi.org/10.1111/j.1472-4642.2008.00503.x
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York
Acknowledgements
We thank the personnel of the Laboratorio di Fitogeografia of the Department of Biology (University of Florence) and all the students who work there for the support in the fieldwork. The authors acknowledge the support of NBFC to University of Florence, funded by the Italian Ministry of University and Research, PNRR, Missione 4 Componente 2, “Dalla ricerca all’impresa”, Investimento 1.4, Project CN00000033.
Funding
Open access funding provided by Università degli Studi di Firenze within the CRUI-CARE Agreement. The present work was realized thanks to funding from the EU LIFE project RESTO CON LIFE, LIFE13 NAT/IT/000471 “Island Conservation in Tuscany, Restoring Habitat not Only for Birds”. The authors acknowledge the support of NBFC to the University of Florence, Department of Biology, funded by the Italian Ministry of University and Research, PNRR, Missione 4 Componente 2, “Dalla ricerca all’impresa”, Investimento 1.4, Project CN00000033.
Author information
Authors and Affiliations
Contributions
LL and MM contributed to the study conception and design. Field investigations were carried out by GF. LL and MM while data curation and analysis were performed by LL and MM. The first draft of the manuscript was written by LL and MM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lazzaro, L., Mugnai, M., Ferretti, G. et al. (Not) sweeping invasive alien plants under the carpet: results from the use of mulching sheets for the control of invasive Carpobrotus spp.. Biol Invasions 25, 2583–2597 (2023). https://doi.org/10.1007/s10530-023-03059-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-023-03059-7