Abstract
Invasive aquatic macrophytes are a major threat to estuarine ecosystems globally, posing difficult control challenges for resource managers. This study examined the efficacy of a fluridone treatment program to control invasive submerged aquatic vegetation (SAV). We leveraged four datasets to examine efficacy of fluridone treatment in the Sacramento–San Joaquin Delta, a freshwater tidal estuary in California, from 2014 to 2018. Annual SAV coverage maps derived from spectroscopy data were used in conjunction with fluridone application data and water current speed predicted by a 3-D hydrodynamic model of the study system. Our objective was to determine if the probability of SAV occurrence in treated sites was significantly different from untreated sites. We examined whether efficacy was influenced by the amount of herbicide applied at a site, the local current speed, and the treatment history of the site. We found that treatment efficacy was positively associated with the amount of herbicide applied per unit area, but this effect was mediated by a negative association with current speed. The efficacy of treatment did not improve when a site was treated for multiple consecutive years. Finally, we did not detect legacy effects one year after cessation of treatment. Our results suggest the need for careful selection of treatment sites to ensure that the amount of fluridone applied is likely to have significant impact given local current speeds. Our study underscores the need for development of additional treatment methodologies for hydrologically dynamic systems such as estuaries, particularly when control is needed year-round and for multiple years.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive Submerged Aquatic Vegetation (SAV) often act as ecosystem engineers that can influence the physical environment though positive feedbacks that further their own survival and expand their niche space (Jones et al. 1996; Koch 2001). Most invasive submerged macrophytes that act as ecosystem engineers develop dense canopies in the water column (Wilson et al. 2007; Santos et al. 2012), which can reduce flow by 40–90% and cause channel incision (Fonseca and Cahalan 1992; Rybicki et al. 1997; Lacy et al. 2021). Slower water velocities reduce sediment carrying capacity causing sediment to settle out of the water column over time, reducing turbidity in the ecosystem (Yarrow et al. 2009; Hestir et al. 2016; Drexler et al. 2020). Significant increases in SAV invaded habitat within aquatic ecosystems can shrink and degrade pelagic habitat, offer cover to non-native predatory fishes, and reduce habitat favored by native fishes (Schultz and Dibble 2012). Once SAV invades, it is the costliest and most difficult form of invasive aquatic vegetation to control (Hussner et al. 2017). Given the many-pronged impact of SAV on invaded ecosystems, effective control strategies are imperative.
Some of the most aggressive invasive submerged species documented around the world are Hydrilla verticillata (L. f.) Royle (water thyme), Egeria densa Planch. (Brazilian waterweed), and Myriophyllum spicatum L. (Eurasian watermilfoil). Fluridone is one of the major herbicides that has been used to treat all three of these species (Netherland et al. 1997; Madsen et al. 2002; Getsinger et al. 2008; Parsons et al. 2009). Fluridone, 1-methyl-3-phenyl-5-[-3(trifluoromethyl)phenyl]-4(1H)-pyridinone, is a systemic herbicide that received a USEPA Section 3 (nation-wide) aquatic registration in 1986 and was the first to be registered for SAV control since the early 1960s (Arnold 1979; Getsinger et al. 2008). This herbicide is known to be effective when applied at low concentrations for a long duration of 60–120 days slowly causing chlorosis and sinking of the SAV canopy (Arnold 1979). In some systems, longer exposure times are needed to achieve control (Fox et al. 1994). This unique requirement of Concentration and Exposure Time (CET) facilitates targeting invasive species like E. densa and M. spicatum without harming native species that might be affected at higher concentrations of the herbicide (Madsen et al. 2002). However, fluridone has shown only limited success with other invasive SAV species such as fanwort (Cabomba caroliniana) and curlyleaf pondweed (Potamogeton crispus) (Madsen et al. 2002; Nelson et al. 2002).
Fluridone efficacy has been tested on various SAV species, most often in mesocosms or whole-lake experiments with a few sample lakes and at most two target species (Netherland et al. 1993; Madsen et al. 2002; Wagner et al. 2007; Parsons et al. 2009). Relative to flowing waters, lentic environments are easier to treat due to the slow-moving water with high water-residence times, which facilitate maintaining the required CET conditions. Very few studies have looked at efficacy of fluridone in flowing water environments (Fox et al. 1994), and most of these have been conducted in artificially simulated tanks with slow-moving water (Van and Steward 1986; Leslie et al. 1993). Moreover, the vast majority of herbicide treatment studies of SAV in flowing waters have focused on contact herbicides that act quickly, such as endothall (Sisneros et al. 1998) and diquat (Fox 1987). Very few studies have examined the duration of treatment effects of systemic agents like fluridone in more dynamic flow systems such as estuaries. The tidal flows in an estuary can quickly transport fluridone away from the application site, thereby decreasing its concentration in SAV. However, SAV is increasingly prevalent in coastal and estuarine systems, and continues to threaten biodiversity and ecosystem integrity (Guy‐Haim et al. 2018); posing difficult challenges to coastal and estuarine natural resource managers.
We evaluated the efficacy of a fluridone herbicide-based approach to control widespread invasive SAV in the Sacramento–San Joaquin Delta (henceforth, Delta), a large, dynamic, tidal, and heavily invaded estuary in California (Cohen and Carlton 1998). Specifically, we examined the efficacy of the fluridone application program on SAV coverage across 133 sites in the Delta and tested the following hypotheses:
-
1.
Factors affecting treatment impact: The impact of treatment at a site will be mediated by (i) amount of herbicide applied during the treatment season, (ii) local hydrology such as speed of the current and (iii) number of consecutive years of treatment at the site.
-
2.
Legacy effect of treatment: The effect of treatment will carry over to the following year even if a site is not treated that year, given that the herbicide is slow-acting and has a systemic effect on the target SAV species.
By testing these hypotheses, we seek to determine herbicide application thresholds necessary to observe significantly reduced SAV occurrence, whether local conditions predetermine effectiveness, whether repeated treatment over multiple years is associated with increased efficacy, and whether treatment can have legacy effects that last multiple years following the cessation of treatment.
Methods
Study area
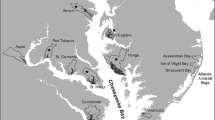
The Sacramento–San Joaquin Delta is a tidal freshwater system in California that covers an area of about 2220 km2 (Fig. 1) and contains diverse habitats from slow-moving lake-like environments to channelized flows with steep bathymetry (Khanna et al. 2012). The SAV community in the Delta is diverse with multiple non-native and native species. During the recent California drought (2012–2016), invasive SAV occurrence in the Delta expanded from 1740 hectares in 2008 (7% of waterways) to 5590 hectares in 2021 (25% of waterways) and some of the changes in niche space appear to be long-lasting (Ustin et al. 2021; Kimmerer et al. 2019).
The Delta, in its current state, is a highly modified landscape. Before the mid-nineteenth century, the tidal marsh area was extensive with few open-water lake-like environments (Atwater et al. 1979). Since then, over 90% of the estuary’s tidal marshes have been diked (Atwater et al. 1979), and myriad water control infrastructure have been built, which reroute the water into straighter paths. Consequently, the modern Delta contains tidally connected marshes, meandering channels, and inundated islands that arose from levee failure over time. This has created a diverse system of channels and large expanses of water with varying bathymetry, water velocity and quality. The area invaded by SAV has been increasing in recent years, making it hard to engineer a cost-effective treatment program (Ta et al. 2017; Ustin et al. 2021). The SAV community in the Delta consists of seven native and four major non-native species (Table 1), but is dominated by the invasive E. densa (Santos et al. 2010, 2012; Ta et al. 2017).
Long-term control program for SAV in the delta
Egeria densa was first reported in the Delta in 1946 (Light et al. 2005) as a release from aquaria, and rapidly expanded its distribution in the 1980s (Jassby and Cloern 2000). Control and management of E. densa and other SAV species (non-native Potamogeton crispus, Myriophyllum spicatum, Cabomba caroliniana, and the native Ceratophyllum demersum) in the Delta is under the jurisdiction of the California State Parks and Recreation, Division of Boating and Waterways (original legislation in 1996, Assembly Bill 2193, amendment to Harbors and Navigation Code, Section 64). The program has always relied on herbicides as the control method (Carruthers et al. 2013; USFWS 2019). Between 2006 and 2019, the program used fluridone almost exclusively because other agents, such as diquat, are limited or prohibited because of toxicity concerns to invertebrate prey and early life stages of the endangered Delta smelt Hypomesus transpacificus, which is endemic to the Delta (Hartless and Lin 2010; DBW 2018, 2019). The Division of Boating and Waterways (DBW) SAV control program has experienced increasing regulatory complexity over the last two decades, and must comply with the National Pollutant Discharge Elimination System (NPDES) to meet Clean Water Act requirements, as well as with mandates of both California and federal Endangered Species Act for both aquatic and terrestrial species (Caudill et al. 2021). This control program is among the most extensive and complex large-scale aquatic invasive species projects in the United States. Until recently, many of the restrictions on the program have prevented it from experimenting with alternative herbicides or physical control methods, and only recently has a permitting structure been developed to allow DBW to investigate the potential of other herbicides such as endothall as alternatives to fluridone (Caudill et al. 2021; Conrad et al. 2022). Furthermore, regulatory compliance has restricted not only the agents that can be used, but also the timing of application. For example, fluridone use was not permitted before June until 2007, but its effectiveness improved in Franks Tract once regulations changed and treatment could begin in April to combat early season growth for E. densa (Santos et al. 2009).
Because fluridone requires a long and sustained CET, the DBW protocol is to apply fluridone on a weekly basis for 8–16 consecutive weeks, with the number of weeks depending on specific site conditions. As per permits acquired by DBW, treatment can be conducted between the months of March and November, but the earlier months are targeted in order to ensure that fluridone is taken up by the plants during the spring growth period. Fluridone is applied at rates consistent with targets of 5–30 ppb in the Delta to minimize direct or indirect effects to protected species (Carruthers et al. 2013; USFWS 2019). Only pelleted, rather than liquid, formulations of fluridone (Sonar® Q, Sonar® One, and Sonar® PR, https://www.sepro.com/aquatics/sonar) are used in the Delta because these formulations drop to the substrate below and release fluridone over a longer period, thus increasing the likelihood of maintaining the required CET in the flowing and tidal dynamics of the Delta. The DBW control program aims to maintain a fluridone concentration of just 2–5 ppb in the water column (DBW 2014) post application, and consistently used this approach throughout the study period. As SAV coverage has continued to expand, treatment has also expanded, and DBW has treated between 1000 and 1800 hectares annually since 2016.
Source datasets
Data were integrated from four distinct datasets to test the efficacy of herbicide treatment for submerged macrophytes in the Delta. More details on data used are presented in supplementary materials and cited references but a short description is included here.
Herbicide application data
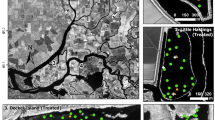
Herbicide application data were computed from two independent datasets provided by DBW—boat path vectors and herbicide application log tables. Boat paths describe where the treatment boats travelled while the log tables record date, time, and amount of herbicide applied at the reach scale. We combined these two datasets to determine if sites embedded within DBW defined reaches were treated or untreated (reference) in a particular year (see Supplementary Material and Fig. 2).
Once the treatment and reference sites were defined, boat path data from 2003 to 2018 were used to determine Consecutive Treatment Years (CTY) for each treatment site from 2014 to 2018. For example, if a site in 2014 had already been treated two years in a row (in 2014 and 2013), its CTY value was 2. A CTY value of 1 indicates that the site was treated in the current year but not in the previous year. Table 2 summarizes number of treatment and reference sites available for analysis from each year.
Class maps based on image data (described below and in Supplementary Material) integrated per DBW reach provided us with the total SAV area within each DBW reach. Thus, we could calculate the amount of herbicide applied per unit area of SAV for all five years of the study by dividing the SAV area treated within each DBW reach by the amount of herbicide sprayed within that reach for that year. Each treated site present within a DBW reach was assigned the application per unit area calculated for that reach.
We did not consider the size of the SAV patch as one of the covariates in our analyses even though the class maps used to determine total SAV area at a site could potentially have been used to determine the area, width or length of the patch within the site. It is possible that smaller patches are more effectively treated by the same fluridone application rate compared to larger patches, or that larger patches reduce water speed more effectively in the center of the patch increasing the residence time of the herbicide and hence, its efficacy. However, we did not have enough sites to justify dividing up our data into more subsets to tease apart these effects.
Image data
Coverage data for SAV came from annual maps produced from hyperspectral imagery acquired in September to November of 2014–2018 (for specific dates, see Table S1). The data along with a detailed report has been published on a public data repository (Khanna et al. 2022). The legal Delta boundary was acquired in 2014 and 2015 (Fig. 1) and in 2016–2018, data were acquired over just the northwest and central Delta (see blue boxes in Fig. 1). The overall workflow and the datasets used for this analysis are shown in Fig. 3. The images were preprocessed to reflectance, geocorrected, transformed and classified using a random forest classifier to produce a location map of the major vegetation communities in the Delta such as SAV, floating aquatic vegetation (FAV), and emergent marsh. For a complete description of data and methods, please refer to Khanna et al. (2018).
Workflow for the study showing the pre-processing steps for image analysis, the four datasets integrated for each treated and reference site, and analyzed for treatment impact and legacy effects. Rectangle stacks represent multiple years of data, rectangles represent a single dataset, diamonds represent a transforming/analyses of data and the grey box represents results of statistical modeling
For each treatment and reference site in the Delta for each year, we determined the number of pixels from our imagery-derived maps with SAV presence and absence. The imagery for all five years was collected in Fall (Table S1). The treatment season generally begins in March, application is at its peak in April to September and starts to taper off in October–November. Thus, our imagery acquisition date is typically at the end of the treatment season and allows us to determine SAV cover after months of herbicide applications.
Predicted current speed
We used the UnTRIM hydrodynamic model (Casulli and Walters 2000; Casulli and Zanolli 2002) to simulate current speed. This model has been widely applied in the San Francisco Estuary and calibrated to extensive hydrodynamic data including water level, flow and salinity measurements (Andrews et al. 2017; Kimmerer et al. 2019). The current speed represents an idealized summer-fall tidal current speed averaged over two semi-diurnal tidal cycles for each site. Since summer-fall current speeds are dominated by tidal flows, as opposed to net flows from freshwater inputs, these current speeds are highly consistent among years. We used it as one of the predictor variables for all five years of the study. Figure 2 shows the UnTRIM grid overlaid on the sites and DBW reaches in both panels.
Statistical assessment of fluridone efficacy
We evaluated herbicide efficacy and legacy effects using a hypothesis-driven approach. We tested whether the probability of SAV presence was a function of the amount of herbicide applied, speed, the interaction between herbicide amount and speed (i.e., allowing the potential herbicide effect to vary across speed), and the consecutive years of herbicide treatment. Specifically, we took a longitudinal, three-level mixed effects logistic (binomial) regression approach with the random effects (random intercepts) of site nested within reach as well as site nested within year. Similarly, we tested legacy effects by evaluating whether the probability of SAV presence during non-treatment years was a function of speed and CTY of the previous year, before treatment stopped (i.e., at time t−1). That is, we evaluated whether multiple years of fluridone treatment would result in reduced probability of SAV presence upon cessation of fluridone treatment using a longitudinal, three-level mixed effects logistic (binomial) regression approach with the non-nested random effects (random intercepts) of site within reach and year. Prior to analysis, continuous variables were transformed to meet the assumption of normality; collinearity was evaluated via correlations analyses among all predictor variables using Kendall’s \(\tau\). The absolute value of Kendall’s \(\tau\) among covariates in all analyses was < 0.02.
Statistical analyses were performed using R CRAN Statistical software (R Core Team 2020; version 4.0.2) using package ‘lme4’ (version 1.1–2.6) following procedures described in Zuur et al. (2009) and Gelman and Hill (2006). Model residuals were used to evaluate model fit and the ‘effects’ package (version 4.2–0) was used to estimate 95% confidence intervals (CI).
Results
Factors affecting treatment impact
Amount of fluridone applied was the strongest factor influencing probability of SAV presence. However, this effect was mediated by current speed. The treatment history of a site (CTY) was the third factor influencing SAV probability but in unexpected ways.
The probability of SAV presence for the average untreated site in the average year decreased with increases in speed (Fig. 4). Specifically, the probability of SAV presence was 0.735 (95% CI of 0. 558–0.859 at the lowest observed speed of 0.001 \(m\bullet {s}^{-1}\), 0.441 (95% CI of 0.345–0.540) at the mean observed speed of 0.053 \(m\bullet {s}^{-1}\) (the mean was calculated on loge-transformed speed), and 0.294 (95% CI of 0. 193–0.420) at the highest observed speed of 0.366 \(m\bullet {s}^{-1}\) for the average untreated site in the average year (Fig. 4).
Grand mean model predicted probability of SAV presence in the average study site during the average study year across the observed range of speed \(\left(m\bullet {s}^{-1}\right)\) for non-treated reference sites (no fluridone application; gray line) and for the mean (pink line) and 95th percentile (green line) of fluridone \(\left(kg\bullet {m}^{-2}\right)\) application for sites treated for one consecutive treatment year (CTY = 1). The polygons represent 95% confidence intervals. See Eq. S1 for model structure and Table S1 for parameter estimates
We also saw a reduction in the probability of SAV presence associated with amount of fluridone applied (Figs. 4 and 5). However, the magnitude of this change decreased with increasing speed. The greatest reduction in the predicted probability of SAV presence due to fluridone occurred at the lowest speeds (< 0.1 \(m\bullet {s}^{-1}\)). The observed pattern was similar for all CTY but the magnitude of change in the probability of SAV presence across fluridone application amounts and speed was diminished for sites treated for three or four consecutive years (Fig. 5e–h) relative to sites treated once or for two consecutive years (Fig. 5a–d). A visual inspection of residuals revealed our model fit was unbiased across covariates, indicating appropriate model fit (Fig. S1).
Grand mean model predicted probability of SAV presence among consecutive treatment years (CTY; one treatment year: a, b; two treatment years: c, d; three treatment years: e, f; and four treatment years: g, h). The left column of panels (a, c, e, g) illustrate the predicted probability of SAV presence in reference sites at the maximum and minimum speed given no fluridone application as faint teal and brown points, respectively, with 95% confidence intervals (faint teal and brown error bars, respectively). The faint horizonal polygons show the non-fluridone treated 95% confidence intervals for comparison to treated sites. The dark dashed lines (in a, c, e, g) are the grand mean model predicted probability of SAV presence in treated sites at the maximum and minimum observed speeds (teal and brown, respectively) across the observed range of fluridone application with the dark colored polygons representing the 95% confidence intervals. The right column of panels (b, d, f, h) illustrate the grand mean model predicted probability of SAV presence in treated sites across the observed ranges of fluridone application and speed with the color corresponding to the scale on the right figure margin. The dashed teal and brown lines correspond to the to the dashed teal and brown lines in the left column of panels (a, c, e, g). Points indicate observed fluridone application and speed values. The solid magenta and green lines in (b) correspond to Fig. 4. All x-axes are cube-root scaled with back-transformed values shown on the top axes; y-axes in (b), (d), (f), and (h) are loge-scaled with back-transformed values shown on the right axes. See Eq. S1 for model structure and Table S1 for parameter estimates
Model simulations for management relevance
We took a simulation approach to evaluate the speed-specific fluridone application values associated with two potential management targets: (1) the amount of fluridone it takes to get a significant reduction in predicted probability of SAV presence in treated sites compared to the predicted probability of SAV presence in reference sites, and (2) the amount of fluridone it takes to reduce the predicted probability of SAV presence to ≤ 0.2.
In general, the amount of fluridone required for a significantly lower predicted probability of SAV presence (i.e., non-overlapping 95% confidence intervals) increased with speed and was higher for sites treated for three or four consecutive years (Fig. 6a). For the average site treated for one year during the average year (Fig. 6a, red line), 0.004 \(kg\bullet {m}^{-2}\) of fluridone was required to achieve a significantly lower predicted probability of SAV presence at the lowest observed speed of 0.008 \(m\bullet {s}^{-1}\) and as high as 0.037 \(kg\bullet {m}^{-2}\) at the highest observed speed of 0.366 \(m\bullet {s}^{-1}\).
Consecutive treatment year-specific (CTY) quantity of fluridone (\(kg\bullet {m}^{-2}\)) associated with the grand mean model predicted probability of SAV presence across current speed (\(m\bullet {s}^{-1}\)) (a) being significantly lower than the predicted probability of SAV presence when not treated and (b) associated with predicted probability of SAV presence ≤ 0.2. (b) the lower prediction limit (dashed lines), mean model prediction (solid lines), and upper prediction limit (dotted lines) can be interpreted as 5%, 50%, and 95% (i.e., significant) chance, respectively, of the predicted probability of SAV presence being ≤ 0.2. A lack of a line on the figure indicates the predicted probability of SAV presence was never ≤ 0.2 (i.e., the upper prediction limits for CTY ≥ 3). See Eq. S1 for model structure and Table S1 for parameter estimates
Using a predicted probability of SAV presence of ≤ 0.2 as a target, we found, on average, fluridone applications of 0.015 \(kg\bullet {m}^{-2}\) were required at the minimum observed speed to meet the target, and fluridone applications of 0.042 \(kg\bullet {m}^{-2}\) were required at the maximum observed speed (Fig. 6b, red solid line). However, to achieve the same result in 95% of the treated sites, the fluridone application to meet the target was 0.026 \(kg\bullet {m}^{-2}\) at the minimum observed speed and this target was never reached at velocities ≥ 0.139, even given the maximum application of 0.054 \(kg\bullet {m}^{-2}\) (Fig. 6b, red dotted line). The pattern was very similar for sites treated for two consecutive years (Fig. 6b, orange dotted line). The predicted probability of SAV presence was never significantly ≤ 0.2 in sites treated for three or four consecutive years given the maximum fluridone applications of 0.023 \(kg\bullet {m}^{-2}\) and 0.031 \(kg\bullet {m}^{-2}\), respectively.
Legacy effect of treatment
Our herbicide legacy effect analysis indicated that the probability of SAV presence was no different between treated and reference sites, regardless of the CTY value for the previous year, as indicated by overlapping 95% confidence intervals in Fig. 7. This lack of a legacy effect was apparent across the observed range of speeds. Furthermore, the grand mean predicted probability of SAV presence did not exhibit a trend of reduced probability of SAV presence with increased years of consecutive treatment. A visual inspection of residuals revealed our model fit was unbiased across covariates, indicating appropriate model fit (Fig. S2).
Grand mean model predicted probability of SAV presence across current speed \(\left(m\bullet {s}^{-1}\right)\) given a one, b two, c three, or d four consecutive treatment years (CTY) during the previous year (t−1; polygons representing the 95% confidence interval). Shown with the grand mean model predicted probability of SAV presence given no treatment in the previous year (i.e., CTYt−1 = 0; gray line; gray polygons representing the 95% confidence interval). See Eq. S2 for model structure and Table S2 for parameter estimates
Discussion
Control of non-native SAV is a major challenge yet critical for maintaining and restoring the ecosystem integrity of the Sacramento and San-Joaquin River Delta, a dynamic, tidal, and heavily invaded estuary in California (Cohen and Carlton 1998). Broadly, estuarine and coastal systems are increasingly subject to aquatic macrophyte invaders (Guy‐Haim et al. 2018), and innovative approaches for their control are urgently needed. Many existing methodologies were developed for hydrologically distinct systems such as lakes as opposed to tidal or estuarine environments (Getsinger et al. 2008). In this study, we evaluated a fluridone-based approach to control widespread invasive SAV in the Delta using five years of chemical treatment observations across its extent. We determined the efficacy of consecutive years of fluridone application on SAV as well as legacy effects of treatment following cessation. We found that treatment efficacy was (i) positively associated with the amount of herbicide applied, however, this effect was mediated by (ii) a negative association with current speed at the application site and (iii) related to the history of treatment at the site (CTY). Additionally, we did not detect legacy effects of treatment regardless of CTY, suggesting SAV recovers within a year following the cessation of treatment.
Challenges of herbicide application in a tidal estuary
While our study confirms that fluridone application is associated with reduced probability of SAV occurrence (Fig. 5), similar to the findings of previous studies (Madsen et al. 2002; Wagner et al. 2007; Parsons et al. 2009), we did not observe the magnitude or longevity of treatment effects reported by these studies. Fluridone treatments were successful in reducing frequency of occurrence of target SAV species by more than 80% in all three of these previous studies, which were conducted in lentic systems. In one of the four lakes treated, Madsen et al. (2002) had partial success showing a reduction in frequency of occurrence by only 27% because the recommended CET conditions could not be maintained. Our findings differ from previous studies likely because the mixed semidiurnal tidal pattern in the Delta makes it extremely difficult to maintain the required CET for fluridone efficacy. Our study indicates that the magnitude of herbicide application required for reducing the probability of SAV occurrence increases with higher current speed (Figs. 4 and 5). Moreover, successful SAV treatment, when defined as a reduction in the probability of SAV occurrence to 20% or less, appears to be infeasible at high speeds given the maximum application of 0.054 \(kg\bullet {m}^{-2}\) observed in the Delta (Fig. 6b).
Other studies have provided evidence that lower CET may be responsible for the low observed efficacy in the Delta. A recent study of SAV treatment impacts in two treated and two reference sites in the Delta showed that even though the targeted post-treatment water concentration of fluridone in the Delta is 2–5 ppb, observed concentrations in water samples were below the minimum 2 ppb threshold, 88% of the time (Rasmussen et al. 2022). They found that the concentration varied with the tidal cycle, with almost 75% of the variability in concentration explained by tidal stage alone (Rasmussen et al. 2022). Indeed, one early study investigating herbicidal approaches to E. densa treatment in the Delta used rhodamine dye to estimate approximate contact time of liquid herbicides with target plants and noted that desired concentrations may last less than 6 h in some locations (Anderson 1999). While the treatment program continues application over an 8–16 week period, the insufficient concentration of herbicide in the water column indicates that the CET requirements are not being met (Rasmussen et al. 2022).
The recommended CET guidelines for fluridone are more than 8 weeks of treatment targeting a sustained concentration of 4–5 ppb for M. spicatum and 8–12 ppb for E. densa (Getsinger et al. 2008; Parsons et al. 2009). Moreover, the non-native C. caroliniana requires a higher dosage of herbicide compared to M. spicatum and E. densa to be effectively managed (Nelson et al. 2002). Based on their current protocols, DBW only targets 5 ppb in the water column. Water samples are tested for residue level and when the target of 5 ppb is detected, DBW decreases the amount of fluridone applied the following week (DBW 2018). Thus, the CET conditions in the Delta are never sufficient to effectively treat E. densa and C. caroliniana and are most often insufficient even for treating M. spicatum.
Our management-focused simulations can inform future herbicide application to achieve a significant reduction in probability of SAV presence or a targeted probability of occurrence (Fig. 6). These simulations could also be used for site selection as we determined a probability of 20% SAV occurrence may be impossible to achieve for some sites even at the highest observed application amounts in our data of 0.054 \(kg\bullet {m}^{-2}\) (Fig. 6b). Moreover, applying a sufficient amount of herbicide to achieve significant reductions may not be permitted across the entire Delta due to restrictions under the federal and California Endangered Species Act and other mandates (USFWS 2019; Conrad et al. 2020, 2022). Beyond these restrictions, Rasmussen et al. (2022) discovered that fluridone tends to accumulate in the sediment at concentrations that are many orders of magnitude higher than in the water column and may persist for months. The effects of fluridone accumulation in sediment on the benthic community in the Delta are unknown but urgently need to be studied.
In addition to the magnitude of herbicide application and current speed, we also evaluated the relationship between consecutive treatment years (CTY) and SAV occurrence. Sites that were treated for just one year or two consecutive years showed a similar relationship between the magnitude of herbicide application and speed of the current. However, sites with significantly lower SAV occurrence probability after being treated for three consecutive years were associated with higher herbicide applications and sites treated for four consecutive years required an even higher application amount to achieve the same SAV occurrence probability (Fig. 6a). We noted that all sites treated for three or four consecutive years were in channel environments (except for one site, the largest marina in the Delta) where current speed is generally faster and the plume of herbicide dilutes over a larger volume of water due to a larger tidal flux. These patterns suggest that channel environments require more intensive herbicide applications. Another possibility is that the previous two years of treatment reduced the SAV cover in the site likely increasing the water speed. Hence, the same site now needs a higher application of herbicide for effective treatment. Since we are using the same water speed value for all years of analysis, this change within site may not be reflected in our data. Given that our dataset for three and four CTYs was limited to channel environments, further research investigating habitat-specific fluridone efficacy is needed.
Legacy effect of treatment
Few field studies have evaluated how long effects can last following cessation of herbicide treatment, and the few studies that exist have been conducted in lake systems. These studies report that treatment effects last at least one year with near total reduction in biomass and frequency of occurrence of the target species (Madsen et al. 2002; Valley et al. 2006; Parsons et al. 2009). Wagner et al. (2007) followed progress in four lakes for 4–7 years and treated regrowth patches with 2,4-D, a contact herbicide, and sometimes with fluridone again. Even with this re-treatment strategy, they found that the frequency of occurrence of M. spicatum was reduced for 1–4 years, but once treatment ceased, M. spicatum returned at a similar or higher frequency compared to pre-treatment levels.
Although varying in long-term efficacy, these studies showed at least one year of sustained reduction in target species cover. In the Delta, for SAV control efforts to provide benefits for native fishes, SAV cover must be reduced for more than a year because native fishes require open water habitat throughout the year. In addition, the prevalence of SAV contributes to increased water clarity throughout the year (Hestir et al. 2016) to the detriment of a critically endangered and endemic fish, Delta smelt (Hypomesus transpacificus), which relies on turbid water (Sommer and Mejia 2013). The control program must maintain SAV reductions on an interannual basis to achieve human and ecological benefits. Hence, we tested if treatment impact could last beyond the year of treatment.
This study, among the first to look at legacy effects of fluridone treatment in a tidal freshwater estuarine ecosystem, did not observe any differences among treatment and reference sites one year post-treatment. This pattern was consistent regardless of current speed or treatment history at each location (Fig. 7). One possible reason for lack of long-term reduction in SAV presence is that the temporary reduction in SAV occurrence creates more niche space for SAV to grow. Treated areas may catch up to the SAV cover in reference sites, indicating that the SAV is likely released from competition and can grow even faster to fill up the niche space made available (Odum 1953). This phenomenon has been observed previously: Madsen et al. (2002) found an increase in M. spicatum biomass and frequency of occurrence beyond pre-treatment levels after one year in one of the lakes where CET guidelines had not been met.
Another possible reason for the springing back of SAV presence is that many of the SAV species in the Delta have overwintering storage organs such as turions (Madsen et al. 2002; Wilson et al. 2007; DiTomaso and Kyser 2013) and tubers (Wersal et al. 2006), which are produced in early spring before the start of treatment. Additionally, the lotic and tidal nature of the Delta can facilitate rapid recolonization of previously treated sites with plant propagules released from patches elsewhere and transported to the treated areas. Even if the roots and shoots of SAV are killed by the herbicide, the plants have already dropped their overwintering organs, which survive to the following year as viable sources of plant regrowth. For example, P. crispus requires treatment early in spring to prevent the plant from producing turions that can sprout and spread even after treatment has killed the plants (Madsen et al. 2002).
Two of the studies that showed a more lasting impact of treatment were in eutrophic lakes (Valley et al. 2006; Parsons et al. 2009). When SAV reduction in biomass was effective, it led to a proliferation of planktonic algae and an increase in turbidity making it harder for SAV to reestablish (Valley et al. 2006; Parsons et al. 2009). In fact, Valley et al. (2006) observed SAV biomass reduction in one lake but not in the other because there was no increase in turbidity in the second lake. This suggests that after the initial impact of the herbicide in reducing SAV biomass, a positive feedback of increasing turbidity due to proliferation of algae is necessary for continued absence of SAV. Whole lake experiments have previously shown the toggling potential of lakes between SAV-dominated, phytoplankton-dominated and floating vegetation dominated systems (Scheffer and Jeppesen 2007; Scheffer et al. 2007). The Sacramento–San Joaquin Delta, however, is typically a low-productivity system (Alpine and Cloern 1992) with respect to phytoplankton and hence the positive feedback loop for maintaining low levels of SAV via phytoplankton dominance, is missing in this system.
Finally, the lack of a legacy effect of treatment might also be because the SAV community adapts to treatment. As invasive SAV cover decreases, other species that are more resistant to fluridone might be released from competition and fill that niche space. This has been previously observed in the Delta when invasive species were replaced by native species over several years of treatment (Caudill et al. 2019). In contrast, Rasmussen et al. (2022) showed that there was no significant change in SAV species assemblage in treated sites compared with reference sites over an 18-month period; native SAV species peaked and declined seasonally, regardless of treatment. A rigorous study focused on SAV species composition in treated and reference sites across the ecosystem for a longer period of time would go a long way in identifying patterns of community change in response to treatment, if they exist.
Management implications
As estuaries are increasingly affected by invasive plants (Williams and Grosholz 2008; Guy‐Haim et al. 2018; Nunes et al. 2020), managers are in critical need of studies that inform best practices for use of existing control tools or explore new tools, as most methodologies have not been well tested in these dynamic, tidal environments.
Our analyses show that fluridone will have a stronger effect in areas with very low current speeds (Fig. 5) and based on this finding, we recommend that resources be focused on sites with lower speeds where there is more likely to be an effect of treatment.
However, we also show that there are limitations to the effectiveness of fluridone in tidal systems even at low current speeds. Alternative control tools, such as other herbicides, mixed herbicides, or physical controls, are needed in tidal systems. Identifying these tools will require experimentation and comparison with existing control methods and untreated reference sites, and critically, a framework for conducting this pilot work may be needed in permitting documents where SAV control is restricted for water quality or endangered species control. Without this framework, it is often impossible to conduct experimental work with new tools. In the Delta, the mechanism for this experimental work was recently adopted in permitting documents for DBW, and the agency is now permitted to experiment with benthic mats, booms and floating barriers, among other tools in specified areas (USFWS 2019).
This study also illustrates the importance of a monitoring program for SAV coverage. Notably, the availability of a system-wide time series of SAV coverage maps enabled the analyses we present in this paper, even though these analyses were not necessarily an articulated goal when the maps were originally developed. Landscape-scale monitoring that includes untreated reference sites, complemented by ground-level data collection at a subset of both treated and untreated sites that includes monitoring of SAV species and herbicide concentrations, is critical to achieving a robust statistical approach to efficacy evaluation (Walters 1986; Allen et al. 2011; Hansen et al. 2015). A consistent monitoring program, together with experimentation and evaluation of control methods, are essential ingredients to a proactive and adaptive approach to SAV management in estuarine systems.
Conclusion
Our study offers new insight into the efficacy of a systemic herbicide in estuarine and tidal ecosystems to control the spread of invasive SAV. The limitations of fluridone treatment we report highlight the need to reexamine CET guidelines for successful herbicides in these dynamic tidal ecosystems. Estuaries are some of the most modified and threatened ecosystems in the world and are particularly vulnerable to invasive species (Williams and Grosholz 2008). Successful management, therefore, requires nimble, multi-faceted, and adaptively managed invasive SAV mitigation programs supported by consistent monitoring.
Data availability
The datasets used in this study were derived from other datasets and will be made available on the Environmental Data Initiative as an open data repository (before the publication of this paper).
Code availability
All R scripts used to conduct this study will be made available on a public GitHub repository.
References
Allen CR, Cumming GS, Garmestani AS, Taylor PD, Walker BH (2011) Managing for resilience. Wildl Biol 17:337–349. https://doi.org/10.2981/10-084
Alpine AE, Cloern JE (1992) Trophic interactions and direct physical effects control phytoplankton biomass and production in an estuary. Limnol Oceanogr 37:946–955. https://doi.org/10.4319/LO.1992.37.5.0946
Anderson LWJ (1999) Egeria invades the Sacramento–San Joaquin Delta. Aquat Nuis Species Dig 3:37–40
Andrews SW, Gross ES, Hutton PH (2017) Modeling salt intrusion in the San Francisco Estuary prior to anthropogenic influence. Cont Shelf Res 146:58–81. https://doi.org/10.1016/j.csr.2017.07.010
Arnold WR (1979) Fluridone—a new aquatic herbicide. J Aquat Plant Manag 17:30–33
Atwater BF, Conrad SG, Dowden JN, Hedel CW, MacDonald RL, Savage W (1979) History, landforms, and vegetation of the estuary’s tidal marshes. In: Conomos TJ (ed) San Francisco Bay: the urbanized estuary: investigations into the Natural History of San Francisco Bay and Delta with reference to the influence of man. AAAS Pacific Division, San Francisco, CA, pp 347–386
Carruthers R, Anderson LWJ, Becerra L et al. (2013) Egeria densa Control Program: a program for effective control of Egeria densa in the Sacramento-San Joaquin Delta and its tributaries (Biological Assessment). United States Department of Agriculture, California Department of Boating and Waterways
Casulli V, Walters RA (2000) An unstructured grid, three-dimensional model based on the shallow water equations. Int J Numer Meth Fluids 32:331–348. https://doi.org/10.1002/(SICI)1097-0363(20000215)32:3%3c331::AID-FLD941%3e3.0.CO;2-C
Casulli V, Zanolli P (2002) Semi-implicit numerical modeling of nonhydrostatic free-surface flows for environmental problems. Math Comput Model 36:1131–1149. https://doi.org/10.1016/S0895-7177(02)00264-9
Caudill J, Jones AR, Anderson L, Madsen JD, Gilbert P, Shuler S, Heilman MA (2019) Aquatic plant community restoration following the long-term management of invasive Egeria densa with fluridone treatments. Manag Biol Invasions 10:473–485. https://doi.org/10.3391/mbi.2019.10.3.05
Caudill J, Madsen JD, Pratt W, Llp C (2021) Operational aquatic weed management in the California Sacramento-San Joaquin River Delta methods for quantifying aquatic macrophyte distribution and abundance view project managing invasive aquatic plants in the California Delta view project. J Aquat Plant Manag 59s:112–122
Cohen AN, Carlton JT (1998) Accelerating invasion rate in a highly invaded estuary. Science 279:555–558. https://doi.org/10.1126/science.279.5350.555
Conrad JL, Chapple D, Bush E et al (2020) Critical Needs for Control of Invasive Aquatic Vegetation in the Sacramento–San Joaquin Delta. Sacramento, CA, USA
Conrad JL, Thomas M, Jetter K et al (2022) Invasive aquatic vegetation in the Sacramento–San Joaquin Delta and Suisun Marsh: The history and science of control efforts and recommendations for the path forward. San Francisco Estuary Watershed Sci 20(4):4. https://doi.org/10.15447/sfews.2023v20iss4art4
DBW (2018) Submersed Aquatic Vegetation Control Program [Egeria densa, Curlyleaf Pondweed, Eurasian Watermilfoil, Fanwort, and Coontail]. Sacramento, CA, USA
DBW (2019) Aquatic Invasive Plant Control Program. 2019 Annual Report. Sacramento, California, USA
DBW (2014) Egeria densa control program. Sacramento, CA, USA
DiTomaso JM, Kyser GB (2013) Coontail. In: Center WR and I (ed) Weed control in natural areas in the western United States. University of California, p 544
Drexler JZZ, Khanna S, Lacy JRR (2020) Carbon storage and sediment trapping by Egeria densa Planch., a globally invasive, freshwater macrophyte. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.142602
Fonseca MS, Cahalan JA (1992) A preliminary evaluation of wave attenuation by four species of seagrass. Estuar Coast Shelf Sci 35:565–576. https://doi.org/10.1016/S0272-7714(05)80039-3
Fox AM (1987) The efficacy and ecological impacts of the management of submerged vegetation in flowing water. ProQuest Dissertations & Theses
Fox AM, Haller WT, Shilling DG (1994) Use of fluridone for hydrilla management in the Withlacoochee River, Florida. J Aquat Plant Manag 32:47–55
Gelman A, Hill J (2006) Data analysis using regression and multilevel/hierarchical models. Cambridge University Press, New York, NY, USA
Getsinger KD, Netherland MD, Grue CE, Koschnick TJ (2008) Improvements in the use of aquatic herbicides and establishment of future research directions. J Aquat Plant Manag 46:32–41
Guy-Haim T, Lyons DA, Kotta J et al (2018) Diverse effects of invasive ecosystem engineers on marine biodiversity and ecosystem functions: a global review and meta-analysis. Glob Change Biol 24:906–924. https://doi.org/10.1111/gcb.14007
Hansen GJA, Gaeta JW, Hansen JF, Carpenter SR (2015) Learning to manage and managing to learn: sustaining freshwater recreational fisheries in a changing environment. Changed publisher: Wiley 40:56–64. https://doi.org/10.1080/03632415.2014.996804
Hartless C, Lin J (2010) Risks of diquat dibromide use to the federally threatened delta smelt (hypomesus transpacificus). Washington D. C., USA
Hestir EL, Schoellhamer DH, Greenberg JA, Morgan-King T, Ustin SL (2016) The effect of submerged aquatic vegetation expansion on a declining turbidity trend in the Sacramento-San Joaquin River Delta. Estuaries Coasts 39:1100–1112. https://doi.org/10.1007/s12237-015-0055-z
Hussner A, Stiers I, Verhofstad MJJM et al (2017) Management and control methods of invasive alien freshwater aquatic plants: A review. Aquat Bot 136:112–137. https://doi.org/10.1016/j.aquabot.2016.08.002
Jassby AD, Cloern JE (2000) Organic matter sources and rehabilitation of the Sacramento–San Joaquin Delta (California, USA). Aquat Conserv-Mar Freshw Ecosyst 10:323–352
Jones CG, Lawton JH, Shachak M (1996) Organisms as ecosystem engineers. In: Samson FB, Knopf FL (eds) Ecosystem management: selected readings. Springer, New York, NY, pp 130–147
Khanna S, Santos MJ, Boyer JD, Shapiro KD, Bellvert J, Ustin SL (2018) Water primrose invasion changes successional pathways in an estuarine ecosystem. Ecosphere 9:e02418. https://doi.org/10.1002/ecs2.2418
Khanna S, Santos MJ, Hestir EL, Ustin SL (2012) Plant community dynamics relative to the changing distribution of a highly invasive species, Eichhornia crassipes: a remote sensing perspective. Biol Invasions 14:717–733. https://doi.org/10.1007/s10530-011-0112-x
Khanna S, Ustin SL, Hestir EL, Santos MJ, Andrew ME, Lay M, Tuil J, Bellvert J, Greenberg JA, Shapiro KD, Rajapakse SS (2022) The Sacramento–San Joaquin Delta genus and community level classification maps derived from airborne spectroscopy data. Knowl Netw Biocomplex. https://doi.org/10.5063/F1N014ZX
Kimmerer W, Wilkerson F, Downing B, et al. (2019) Effects of drought and the emergency drought barrier on the ecosystem of the California Delta. San Francisco Estuary and Watershed Science 17:Article 2. https://doi.org/10.15447/sfews.2019v17iss3art1
Koch E (2001) Beyond light: physical, geological, and geochemical parameters as possible submersed aquatic vegetation habitat requirements. Estuaries Coasts 24:1–17. https://doi.org/10.2307/1352808
Lacy JR, Foster-Martinez MR, Allen RM, Drexler JZ (2021) Influence of invasive submerged aquatic vegetation (E. densa) on currents and sediment transport in a freshwater tidal system. Water Resour Res 57:e2020WR028789. https://doi.org/10.1029/2020WR028789
Leslie AJ, Schmitz DC, Kipker RL, Girardin DL (1993) Movement of fluridone in the upper St. Johns River, Florida. Fla Sci 56:168–177
Light T, Grosholz T, Moyle PB (2005) Delta Ecological Survey (Phase I): nonindigenous aquatic species in the Sacramento–San Joaquin Delta, a Literature Review. U. S. Fish and Wildlife Service
Madsen JD, Getsinger KD, Stewart RM, Owens CS (2002) Whole lake fluridone treatments for selective control of eurasian watermilfoil: II. impacts on submersed plant communities. Lake Reserv Manag 18:191–200. https://doi.org/10.1080/07438140209354148
Nelson LS, Stewart AB, Getsinger KD (2002) Fluridone effects on fanwort and water marigold. J Aquat Plant Manag 40:58–63
Netherland MD (2015) Laboratory and greenhouse response of monoecious hydrilla to fluridone. J Aquat Plant Manag 53:178–184
Netherland MD, Getsinger KD, Skogerboe JD (1997) Mesocosm evaluation of the species-selective potential of fluridone. J Aquat Plant Manag 35:41–50
Netherland MD, Getsinger KD, Turner EG (1993) Fluridone concentration and exposure tune requirements for control of eurasian watermilfoil and hydrilla. J Aquat Plant Manag 31:189–194
Nunes M, Adams JB, van Niekerk L (2020) Changes in invasive alien aquatic plants in a small closed estuary. S Afr J Bot 135:317–329. https://doi.org/10.1016/j.sajb.2020.09.016
Odum EP (1953) Fundamentals of ecology. W. B. Saunders Co., Philadelphia, Pennsylvania, and London, England
Parsons JK, Couto A, Hamel KS, Marx GE (2009) Effect of fluridone on macrophytes and fish in a coastal Washington Lake. J Aquat Plant Manag 47:31–40
R Core Team (2020) R: A Language and Environment for Statistical Computing
Rasmussen N, Conrad JL, Green H et al (2022) Efficacy and fate of fluridone applications for control of invasive submerged aquatic vegetation in an estuarine environment. Estuaries Coasts. https://doi.org/10.1007/s12237-022-01079-5
Rybicki NB, Jenter HL, Carter V, Baltzer RA, Turtora M (1997) Observations of tidal flux between a submersed aquatic plant stand and the adjacent channel in the Potomac River near Washington, D.C. Limnol Oceanogr 42:307–317. https://doi.org/10.4319/lo.1997.42.2.0307
Santos MJ, Anderson LWJ, Ustin SL (2010) Effects of invasive species on plant communities: an example using submersed aquatic plants at the regional scale. Biol Invasions 13:443–457. https://doi.org/10.1007/s10530-010-9840-6
Santos MJ, Hestir EL, Khanna S, Ustin SL (2012) Image spectroscopy and stable isotopes elucidate functional dissimilarity between native and nonnative plant species in the aquatic environment. New Phytol. https://doi.org/10.1111/j.1469-8137.2011.03955.x
Santos MJ, Khanna S, Hestir EL et al (2009) Use of hyperspectral remote sensing to evaluate efficacy of aquatic plant management in the Sacramento–San Joaquin River Delta, California. Invasive Plant Sci Manag 2:216–229. https://doi.org/10.1614/IPSM-08-115.1
Scheffer M, Jeppesen E (2007) Regime shifts in Shallow Lakes. Ecosystems 10:1–3. https://doi.org/10.1007/s10021-006-9002-y
Scheffer M, van Nes EH, Martens K (2007) Shallow lakes theory revisited: various alternative regimes driven by climate, nutrients, depth and lake size. In: Gulati RD, Lammens E, Pauw N, Donk E (eds) Shallow Lakes in a changing world. Springer, Netherlands, pp 455–466
Schultz R, Dibble E (2012) Effects of invasive macrophytes on freshwater fish and macroinvertebrate communities: the role of invasive plant traits. Hydrobiologia 684:1–14. https://doi.org/10.1007/s10750-011-0978-8
Sisneros D, Lichtwardt M, Greene T (1998) Low-dose metering of endothall for aquatic plant control in flowing water. J Aquat Plant Manag 36:69–72
Sommer T, Mejia F (2013) A place to call home: a synthesis of delta smelt habitat in the upper San Francisco estuary. San Franc Estuary Watershed Sci. https://doi.org/10.15447/sfews.2013v11iss2art4
Ta J, Anderson LWJ, Christman MA et al (2017) Invasive aquatic vegetation management in the Sacramento–San Joaquin River Delta: status and recommendations. San Franc Estuary Watershed Sci 15:5. https://doi.org/10.15447/sfews.2017v15iss4art5
USFWS (2019) Biological Opinion for the aquatic invasive plant control program located in the Sacramento–San Joaquin Delta, its surrounding tributaries and Suisun Marsh, California. Sacramento, CA, USA
Ustin SL, Khanna S, Lay MC (2021) Remote sensing of the Sacramento–San Joaquin Delta to enhance mapping for invasive and native aquatic plant species. Davis, CA, USA
Valley RD, Crowell W, Welling CH, Proulx N (2006) Effects of a low-dose fluridone treatment eutrophic Minnesota Lake dominated. J Aquat Plant Manag 44:19–25
Van TK, Steward KK (1986) The use of controlled-release fluridone fibers for control of hydrilla (hydrilla verticillata). Weed Sci 34:70–76. https://doi.org/10.1017/s0043174500026485
Wagner KI, Hauxwell J, Rasmussen PW et al (2007) Whole-lake herbicide treatments for eurasian watermilfoil in four wisconsin lakes: effects on vegetation and water clarity. Lake Reserv Manag 23:83–94. https://doi.org/10.1080/07438140709353912
Walters C (1986) Adaptive management of revewable resources. Macmillan Publishers Ltd., Basingstoke, UK
Wersal RM, Madsen JD, McMillan BR, Gerard PD (2006) Environmental Factors Affecting Biomass and Distribution of Stuckenia pectinata in the Heron Lake System, Minnesota, USA. Wetlands 26:313–321. https://doi.org/10.1672/0277-5212(2006)26[313:EFABAD]2.0.CO;2
Williams SL, Grosholz ED (2008) The invasive species challenge in estuarine and coastal environments: marrying management and science. Estuaries Coasts 31:3–20. https://doi.org/10.1007/s12237-007-9031-6
Wilson CE, Darbyshire SJ, Jones R (2007) The biology of invasive alien plants in Canada. 7. Cabomba caroliniana A. Gray. Can J Plant Sci 87:615–638. https://doi.org/10.4141/P06-068
Yarrow M, Marin VH, Finlayson M, Tironi A, Delgado LE, Fischer F (2009) The ecology of Egeria densa Planchon (Liliopsida: alismatales): a wetland ecosystem engineer? Rev Chil Hist Nat 82:299–313
Zuur A, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
This research received no specific grant from any funding agency, commercial or not-for-profit sectors. No conflicts of interest have been declared. This research was made possible by generous data sharing from the Division of Boating and Waterways, California Department of Parks and Recreation and the Center for Spatial Technologies and Remote Sensing at University of California, Davis. We are grateful to Dr. Nicholas Rasmussen and researchers at the Interagency Ecological Program (IEP) Science Management Team for reviewing the manuscript and offering valuable guidance. They also contributed to research by donating the time of their scientists to this project. We are especially grateful to Dr. Susan L. Ustin for the use of her lab resources and staff help, and to Jeffrey Caudill at the Division of Boating and Waterways for guidance on understanding the treatment program executed by his agency.
Funding
The authors did not receive any external funds from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
SK contributed to the study concept and design, data collection, image analysis, data visualization, manuscript writing, and manuscript editing. JWG contributed to statistical analysis, data visualization, writing, and manuscript editing. JLC contributed to the study concept, data collection, manuscript writing and editing. EG contributed to data creation, manuscript writing and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khanna, S., Gaeta, J.W., Conrad, J.L. et al. Multi-year landscape-scale efficacy analysis of fluridone treatment of invasive submerged aquatic vegetation in the Sacramento–San Joaquin Delta. Biol Invasions 25, 1827–1843 (2023). https://doi.org/10.1007/s10530-023-03013-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-023-03013-7