Abstract
Objective
Marine actinomycetes from the genus Salinispora have an unexploited biotechnological potential. To accurately estimate their application potential however, data on their cultivation, including biomass growth kinetics, are needed but only incomplete information is currently available.
Results
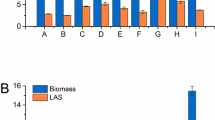
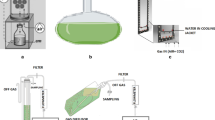
This work provides some insight into the effect of temperature, salinity, nitrogen source, glucose concentration and oxygen supply on growth rate, biomass productivity and yield of Salinispora tropica CBN-440T. The experiments were carried out in unbaffled shake flasks and agitated laboratory-scale bioreactors. The results show that the optimum growth temperature lies within the range 28–30 °C, salinity is close to sea water and the initial glucose concentration is around 10 g/L. Among tested nitrogen sources, yeast extract and soy peptone proved to be the most suitable. The change from unbaffled to baffled flasks increased the volumetric oxygen transfer coefficient (kLa) as did the use of agitated bioreactors. The highest specific growth rate (0.0986 h−1) and biomass productivity (1.11 g/L/day) were obtained at kLa = 28.3 h−1. A further increase in kLa was achieved by increasing stirrer speed, but this led to a deterioration in kinetic parameters.
Conclusions
Improvement of S. tropica biomass growth kinetics of was achieved mainly by identifying the most suitable nitrogen sources and optimizing kLa in baffled flasks and agitated bioreactors.



Similar content being viewed by others
References
Amanullah A, Justen P, Davies A, Paul GC, Nienow AW, Thomas CR (2000) Agitation induced mycelial fragmentation of Aspergillus oryzae and Penicillium chrysogenum. Biochem Eng J 5:109–114
Andryukov BG, Mikhaylov VV, Besednova NN, Zaporozhets TS, Bynina MP, Matosova EV (2018) The bacteriocinogenic potential of marine microorganisms. Russ J Mar Biol 44:433–441
Contador CA, Rodriguez V, Andrews BA, Asenjo JA (2015) Genome-scale reconstruction of Salinispora tropica CNB-440 metabolism to study strain-specific adaptation. Antonie Van Leeuwenhoek 108:1075–1090
Heenan NC, Adams CM, Hosken WR, Fleet HG (2002) Growth medium for culturing probiotic bacteria for applications in vegetarian food products. LWT Food Sci Technol 35:171–176
Jensen RP, Dwight R, Fenical W (1991) Distribution of Actinomycetes in near-shore tropical marine sediments. Appl Environ Microbiol 57:1102–1108
Jensen RP, Moore SB, Ferical W (2015) The marine actinomycete genus Salinispora: a model organism for secondary metabolite discovery. Nat Prod Rep 32:738–751
Large KP, Ison AP, Williams DJ (1998) The effect of agitation rate on lipid utilisation and clavulanic acid production in Streptomyces clavuligerus. J Biotechnol 63:111–119
Maldonado AL, Fenical W, Jensen RP, Kauffman AC, Mincer JT, Ward CA, Bull TA, Goodfellow M (2005) Salinispora arenicola gen. nov., sp. nov. and Salinispora tropica sp. nov., obligate marine actinomycetes belonging to the family Micromonosporaceae. Int J Syst Evol Microbiol 55:1759–1766
Manam RR, Macherla RV, Tsueng G, Dring WX, Weiss J, Neuteboom CTS, Lam SK, Poots CB (2009) Antiprotealide is a natural product. J Nat Prod 72:295–297
Mincer TJ, Jensen PR, Kauffman CA, Fenical W (2002) Widespread and persistent populations of a major new marine actinomycete taxon in ocean sediments. Appl Environ Microbiol 68:5005–5011
Niewerth D, Jansen G, Riethoff VFL, Meerloo J, Kale JA, Moore SB, Assaraf GY, Anderl LJ, Zweegman S, Kaspers LJG, Cloos J (2014) Antileukemic activity and mechanism of drug resistance to the marine Salinispora tropica proteasome inhibitor Salinosporamide A (Marizomib). Mol Pharmacol 86:12–19
Nouioui I, Carro L, García-López M, Meier-Kolthoff PJ, Woyke T, Kyrpides CN, Pukall R, Klenk PH, Goodfellow M, Göker M (2018) Genome-based taxonomic classification of the phylum actinobacteria. Front Microbiol 9:1–119
Olmos E, Mehmood N, Husein LH, Goergen JL, Fick M, Delaunay S (2013) Effects of bioreactor hydrodynamics on the physiology of Streptomyces. Bioprocess Biosyst Eng 36:259–272
Potts CB, Lam SK (2010) Generating a generation of proteasome inhibitors: from microbial fermentation to total synthesis of salinosporamide A (Marizomib) and other salinosporamides. Mar Drugs 8:835–880
Richter TKS (2014) Discovery, biosynthesis and evolutionary history of sioxanthin, a novel glycosylated carotenoid from marine bacteria Salinispora. UC San Diego. https://escholarship.org/uc/item/7mn416k3. Accessed 27 Oct 2020
Richter SKT, Hughes CC, Moor SB (2015) Sioxanthin, a novel glycosylated carotenoid, reveals an unusual subclustered biosynthetic pathway. Environ Microbiol 17:2158–2171
Roubos JA, Krabben P, Luiten RGM, Verbruggen HB, Heijnen JJ (2001) A quantitative approach to characterizing cell lysis caused by mechanical agitation of Streptomyces clavuligerus. Biotechnol Prog 17:336–347
Stiborova H, Branska B, Vesela T, Lovecka P, Stranska M, Hajslova J, Jiru M, Patakova P, Demnerova K (2016) Transformation of raw feather waste into digestible peptides and amino acids. J Chem Technol Biotechnol 91:1629–1637
Subramani R, Aalbersberg W (2012) Marine actinomycetes: an ongoing source of novel bioactive metabolites. Microbiol Res 167:571–580
Tribe LA, Briens CL, Margaritis A (1995) Determination of the volumetric mass transfer coefficient (kLa) using the dynamic “gas out-gas in” method: analysis of errors caused by dissolved oxygen probes. Biotechnol Bioeng 46:388–392
Tsueng G, Lam KS (2008a) A low-sodium-salt formulation for the fermentation of salinosporamides by Salinispora tropica strain NPS21184. Appl Microbiol Biotechnol 78:821–826
Tsueng G, Lam KS (2008b) Growth of Salinispora tropica strains CNB440, CNB476, and NPS21184 in nonsaline, low-sodium media. Appl Microbiol Biotechnol 80:873–880
Tsueng G, Lam KS (2010) A preliminary investigation on the growth requirement for monovalent cations, divalent cations and medium ionic strength of marine actinomycete Salinispora. Appl Microbiol Biotechnol 86:1525–1534
Tsueng G, Teisan S, Lam KS (2008) Defined salt formulations for the growth of Salinispora tropica strain NPS21184 and the production of salinosporamide A (NPI-0052) and related analogs. Appl Microbiol Biotechnol 78:827–832
Zangh JJ, Moore BS, Tang X (2018) Engineering Salinispora tropica for heterologous expression of natural product biosynthetic gene clusters. Appl Microbiol Biotechnol 102:8437–8446
Funding
This research was supported by the Ministry of Education, Youth and Sports of the Czech Republic, Ministry of Agriculture of the Czech Republic (institutional support MZE-RO1918), and by Insubria University “Fondo di Ateneo per la Ricerca” 2017–2018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jezkova, Z., Binda, E., Potocar, T. et al. Laboratory scale cultivation of Salinispora tropica in shake flasks and mechanically stirred bioreactors. Biotechnol Lett 43, 1715–1722 (2021). https://doi.org/10.1007/s10529-021-03121-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-021-03121-1