Abstract
Objective
To develop a method combining enzymatic catalysis and resting-cell biotransformation to produce allitol from low cost substrate d-glucose.
Results
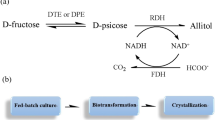
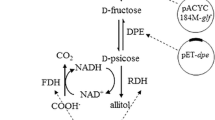
The recombinant E. coli expressing d-psicose-3-epimerase (DPE), ribitol dehydrogenase (RDH) and formate dehydrogenase (FDH) for allitol production from d-fructose was constructed. The optimizations of the cell catalytic conditions and the cell cultivation conditions were made. Then, 63.4 g allitol L−1 was obtained from 100 g d-fructose L−1 in 4 h catalyzed by the recombinant E. coli cells. In order to decrease the substrate cost, d-glucose was used as the substrate instead of d-fructose and immobilized glucose isomerase was used to convert d-glucose into d-fructose. In order to simplify allitol production process from d-glucose, one-pot reaction using the mixed catalysts was used and the reaction conditions were optimized. Finally, 12.7 g allitol L−1 was obtained from 50 g d-glucose L−1 catalyzed by the mixed catalysts of immobilized glucose isomerase and the recombinant E. coli cells.
Conclusions
Allitol can be efficiently produced from low cost substrate d-glucose by using the method combining enzymatic catalysis and resting-cell biotransformation, which is the first report.









Similar content being viewed by others
References
Cutayar JM, Poillon D (1989) High cell density culture of E. coli in a fed-batch system with dissolved oxygen as substrate feed indicator. Biotechnol Lett 11:155–160. https://doi.org/10.1007/BF01026048
Hassanin HAM, Wang X, Mu W, Zhang T, Jiang B (2015) Cloning and characterization of a new ribitol dehydrogenase from Providencia alcalifaciens RIMD 1656011. J Sci Food Agric 96:2917–2924. https://doi.org/10.1002/jsfa.7589
Hassanin HAM, Letsididi R, Koko MYF, Mu W, Elferga A, Jiang B (2016a) Synthesis of allitol from D-psicose using ribitol dehydrogenase and formate dehydrogenase. Trop J Pharm Res 15:2701–2708. https://doi.org/10.4314/tjpr.v15i12.23
Hassanin HAM, Mu W, Koko MYF, Zhang T, Masamba K, Jiang B (2016b) Allitol: production, properties and applications. Int J Food Sci Technol 52:91–97. https://doi.org/10.1111/ijfs.13290
Izumori K (2006) Izumoring: a strategy for bioproduction of all hexoses. J Biotechnol 124:717–722. https://doi.org/10.1016/j.jbiotec.2006.04.016
Jin LQ, Xu Q, Liu ZQ, Jia DX, Liao CJ, Chen DS, Zheng YG (2017) Immobilization of recombinant glucose isomerase for efficient production of high fructose corn syrup. Appl Biochem Biotechnol 183:293–306. https://doi.org/10.1007/s12010-017-2445-0
Jumde VR, Eisink NN, Witte MD, Minnaard AJ (2016) C3 epimerization of glucose, via regioselective oxidation and reduction. J Org Chem 81:11439–11443. https://doi.org/10.1021/acs.joc.6b02074
Kornberg HL, Lambourne LTM, Sproul AA (2000) Facilitated diffusion of fructose via the phosphoenolpyruvate/glucose phosphotransferase system of Escherichia coli. Proc Natl Acad Sci USA 97:1808–1812. https://doi.org/10.1073/pnas.97.4.1808
Korz DJ, Rinas U, Hellmuth K, Sanders EA, Deckwer WD (1995) Simple fed-batch technique for high cell density cultivation of Escherichia coli. J Biotechnol 39:59–65. https://doi.org/10.1016/0168-1656(94)00143-Z
Li C, Zhang CJ, Lin JQ, Gao L, Lin HB, Lin JQ (2017) Enzymatic fructose removal from D-psicose bioproduction model solution and the system modeling and simulation. J Chem Technol Biotechnol 93:1249–1260. https://doi.org/10.1002/jctb.5483
Li C, Lin JQ, Gao L, Lin HB, Lin JQ (2018a) Modeling and simulation of enzymatic gluconic acid production using immobilized enzyme and CSTR–PFTR circulation reaction system. Biotechnol Lett 40:649–657. https://doi.org/10.1007/s10529-018-2509-4
Li C, Lin JQ, Guo QQ, Zhang CJ, Du K, Lin HB, Lin JQ (2018b) D-psicose 3-epimerase secretory overexpression, immobilization, and d-psicose biotransformation, separation and crystallization. J Chem Technol Biotechnol 93:350–357. https://doi.org/10.1002/jctb.5360
Muniruzzaman S, Kobayashi H, Izumori K (1994) Production of d-talitol from d-tagatose by Aureobasidium pullulans strain 113B. J Ferm Bioeng 78:346–350. https://doi.org/10.1016/0922-338X(94)90278-X
Restaino OF, Bhaskar U, Paul P, Li L, De Rosa M, Dordick JS, Linhardt RJ (2013) High cell density cultivation of a recombinant E. coli strain expressing a key enzyme in bioengineered heparin production. Appl Microbiol Biotechnol 97:3893–3900. https://doi.org/10.1007/s00253-012-4682-z
Zhu Y, Li H, Liu P, Yang J, Zhang X, Sun Y (2015) Construction of allitol synthesis pathway by multi-enzyme coexpression in Escherichia coli and its application in allitol production. J Ind Microbiol Biotechnol 42:661–669. https://doi.org/10.1007/s10295-014-1578-1
Acknowledgements
This research was funded by the Key R & D Plan of Shandong Province in 2019 (2019GSF107015), China. The authors would like to thank Chengjia Zhang, Caiyun Sun from the Core Facilities for Life and Environmental Sciences, State Key Lab of Microbial Technology for help and guidance in the experiments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wen, X., Lin, H., Ren, Y. et al. Optimization for allitol production from d-glucose by using immobilized glucose isomerase and recombinant E. coli expressing d-psicose-3-epimerase, ribitol dehydrogenase and formate dehydrogenase. Biotechnol Lett 42, 2135–2145 (2020). https://doi.org/10.1007/s10529-020-02917-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02917-x