Abstract
Objective
Pectinase is an industrially important enzyme which is employed in an array of commercial processes; cost of production, however, impedes its application. The main objective of this study was to design a two-layered strategy for the reduction of production cost, firstly by using a yeast co-culture in an immobilized form on an agricultural waste matrix, corncob (CB), secondly by utilizing orange peels (OP) as substrate.
Results
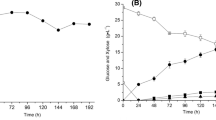
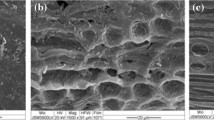
Two yeast strains, Saccaromyces cerevisiae MK-157 and Geotrichum candidum AA15 were cultivated as mono-, as well as, co-culture after immobilization on CB and pectinase production was monitored. Initial experiments revealed that co-culture is beneficial to get sustainable product in subsequent 2nd and 3rd production cycles. The factors affecting pectinase production in consecutive three production cycles were studied by employing Plackett–Burman design and the significant factors were optimized through Box–Behnken design. Under optimized conditions, 17.89 IU mL−1 of pectinase was obtained. Scanning electron micrographs presented damaged immobilized yeast cells on CB after the 3rd production cycle.
Conclusion
The pectinase production was improved substantially by using immobilized co-culture and hence the strategy was found effective at lab scale. Since, pectinase is applied in orange juice clarification, therefore, the study can be extended to move forward towards circular economy.


Similar content being viewed by others
References
Abbas M, Khan MM, Mughal SM, Ji P (2015) Comparison of infection of Citrus tristeza closterovirus in Kinnow mandarin (Citrus reticulata) and Mosambi sweet orange (Citrus sinensis) in Pakistan. Crop Prot. https://doi.org/10.1016/j.cropro.2015.09.006
Abebe Alamineh E (2018) Extraction of pectin from orange peels and characterizing its physical and chemical properties. Am J Appl Chem. https://doi.org/10.11648/j.ajac.20180602.13
Ahmed A, Sohail M (2019) Characterization of pectinase from Geotrichum candidum AA15 and its potential application in orange juice clarification. J King Saud Univ Sci. https://doi.org/10.1016/j.jksus.2019.07.002
Ahmed A, Khan MN, Ahmad A et al (2019) Optimization of pectinase production from Geotrichum candidum AA15 using response surface methodology. Pakistan J Bot 51:743–750. https://doi.org/10.30848/PJB2019-2(41)
Almeida C, Brányik T, Moradas-Ferreira P, Teixeira J (2003) Continuous production of pectinase by immobilized yeast cells on spent grains. J Biosci Bioeng 96:513–518. https://doi.org/10.1263/jbb.96.513
Carvalho EA, Nunes LV, dos Goes LM et al (2018) Peach-palm (Bactris gasipaes Kunth) waste as substrate for xylanase production by Trichoderma stromaticum AM7. Chem Eng Commun. https://doi.org/10.1080/00986445.2018.1425208
de la Torre I, Ravelo M, Segarra S et al (2017) Study on the effects of several operational variables on the enzymatic batch saccharification of orange solid waste. Bioresour Technol. https://doi.org/10.1016/j.biortech.2017.08.094
Dhillon SS, Gill RK, Gill SS, Singh M (2004) Studies on the utilization of citrus peel for pectinase production using fungus Aspergillus niger. Int J Environ Stud. https://doi.org/10.1080/0020723032000143346
dos Santos TC, dos Santos Reis N, Silva TP et al (2018) Production, optimisation and partial characterisation of enzymes from filamentous fungi using dried forage cactus pear as substrate. Waste Biomass Valoriz. https://doi.org/10.1007/s12649-016-9810-z
Ejaz U, Ahmed A, Sohail M (2018) Statistical optimization of immobilization of yeast cells on corncob for pectinase production. Biocatal Agric Biotechnol 14:450–456. https://doi.org/10.1016/j.bcab.2018.04.011
Fiedurek J, Ilczuk Z, Łobarzewski J, Pleszczynska M (1992) Optimization of pectinolytic enzymes biosynthesis by immobilized mycelium of Aspergillus niger 71. Zentralbl Mikrobiol. https://doi.org/10.1016/s0232-4393(11)80357-4
Fiedurek J, Szczodrak J, Rogalski J (1995) Seeds as natural matrices for immobilization of Aspergillus niger mycelium producing pectinases. J Appl Bacteriol. https://doi.org/10.1111/j.1365-2672.1995.tb03426.x
Genisheva Z, Mussatto SI, Oliveira JM, Teixeira JA (2011) Evaluating the potential of wine-making residues and corn cobs as support materials for cell immobilization for ethanol production. Ind Crops Prod 34:979–985. https://doi.org/10.1016/j.indcrop.2011.03.006
Habrylo O, Evangelista DE, Castilho PV et al (2018) The pectinases from Sphenophorus levis: potential for biotechnological applications. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2018.01.172
Ibrahim D, Weloosamy H, Sheh-Hong L (2014) Potential use of nylon scouring pad cubes attachment method for pectinase production by Aspergillus niger HFD5A-1. Process Biochem. https://doi.org/10.1016/j.procbio.2014.01.012
Kratchanova M, Pavlova E, Panchev I (2004) The effect of microwave heating of fresh orange peels on the fruit tissue and quality of extracted pectin. Carbohydr Polym. https://doi.org/10.1016/j.carbpol.2004.01.009
Laopaiboon L, Laopaiboon P (2012) Ethanol production from sweet sorghum juice in repeated-batch fermentation by Saccharomyces cerevisiae immobilized on corncob. World J Microbiol Biotechnol 28:559–566. https://doi.org/10.1007/s11274-011-0848-6
Lee SE, Lee CG, Kang DH et al (2012) Preparation of corncob grits as a carrier for immobilizing yeast cells for ethanol production. J Microbiol Biotechnol. https://doi.org/10.4014/jmb.1202.02049
Nighojkar S, Phanse Y, Sinha D et al (2006) Production of polygalacturonase by immobilized cells of Aspergillus niger using orange peel as inducer. Process Biochem. https://doi.org/10.1016/j.procbio.2005.12.009
Noreen A, Nazli ZIH, Akram J et al (2017) Pectins functionalized biomaterials; a new viable approach for biomedical applications: a review. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2017.03.029
Oberoi HS, Vadlani PV, Madl RL et al (2010) Ethanol production from orange peels: two stage hydrolysis and fermentation studies using optimized parameters through experimental design. J Agric Food Chem. https://doi.org/10.1021/jf903163t
Plessas S, Bekatorou A, Kanellaki M et al (2007) Use of immobilized cell biocatalysts in baking. Process Biochem. https://doi.org/10.1016/j.procbio.2007.05.023
Poondla V, Yannam SK, Gummadi SN et al (2016) Enhanced production of pectinase by Saccharomyces cerevisiae isolate using fruit and agro-industrial wastes: its application in fruit and fiber processing. Biocatal Agric Biotechnol. https://doi.org/10.1016/j.bcab.2016.02.007
Qadir F, Shariq M, Ahmed A, Sohail M (2018) Evaluation of a yeast co-culture for cellulase and xylanase production under solid state fermentation of sugarcane bagasse using multivariate approach. Ind Crops Prod. https://doi.org/10.1016/j.indcrop.2018.07.021
Rosyida VT, Indrianingsih AW, Maryana R, Wahono SK (2015) Effect of temperature and fermentation time of crude cellulase production by Trichoderma reesei on straw substrate. Energy Proc. https://doi.org/10.1016/j.egypro.2015.01.065
Shariq M, Muhammad F, Ahmad A et al (2018) Production and characterization of endoglucanase from an indigenous yeast strain. Pak J Bot 50:2413–2421
Wang W, Chen W, Zou M et al (2018) Applications of power ultrasound in oriented modification and degradation of pectin: a review. J Food Eng. https://doi.org/10.1016/j.jfoodeng.2018.04.016
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qadir, F., Ejaz, U. & Sohail, M. Co-culturing corncob-immobilized yeasts on orange peels for the production of pectinase. Biotechnol Lett 42, 1743–1753 (2020). https://doi.org/10.1007/s10529-020-02897-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-020-02897-y