Abstract
Objective
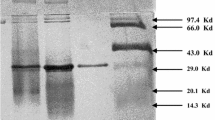
The biochemical properties of a putative thermostable cycloisomaltooligosaccharide (CI) glucanotransferase gene from Thermoanaerobacter thermocopriae were determined using a recombinant protein (TtCITase) expressed in Escherichia coli and purified to a single protein.
Results
The 171-kDa protein displayed maximum activity at pH 6.0, and enzyme activity was stable at pH 5.0–11.0. The optimal temperature was 60 °C in 1 h incubation, and thermal stability of the protein was 63% at 60 °C for 24 h. TtCITase produced CI-7 to CI-17, as well as CI-18, CI-19, and CI-20, which are relatively large CIs. Additionally, an unusual kinetic feature of TtCITase was its negative cooperative behavior in the dextran T2000 cleavage reaction.
Conclusions
Based on our results, TtCITase can be applied to produce relatively large CIs at high temperature.






Similar content being viewed by others
References
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Britton HTK, Robinson RA (1931) Universal buffer solutions and the dissociation constant of veronal. J Chem Soc. https://doi.org/10.1039/JR9310001456
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrrisat B (2009) The Carbohydrate-Active ENZymes Database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37:D233–D238
Funane K, Terasawa K, Mizuno Y, Ono H, Miyagi T, Gibu S, Tokashiki T, Kawabata Y, Kim YM, Kimura A, Kobayashi M (2007) A novel cyclic isomaltooligosaccharide (cycloisomaltodecaose, CI-10) produced by Bacillus circulans T-3040 displays remarkable inclusion ability compared with cyclodextrins. J Biotechnol 130:188–192
Funane K, Terasawa K, Mizuno Y, Ono H, Gibu S, Tokashiki T, Kawabata Y, Kim YM, Kimura A, Kobayashi M (2008) Isolation of Bacillus and Paenibacillus bacterial strains that produce large molecules of cyclic isomaltooligosaccharides. Biosci Biotechnol Biochem 72:3277–3280
Funane K, Kawabata Y, Suzuki R, Kim YM, Kang HK, Fujimoto Z, Kimura A, Kobayashi M (2011) Deletion analysis of the C-terminal region of cycloisomaltooligosaccahride glucanotransferase from Bacillus circulans T-3040. Biochim Biophys Acta 1814:428–434
Funane K, Ichinose H, Araki M, Suzuki R, Kimura K, Fujimoto Z, Kobayashi M, Kimura A (2014) Evidence for cycloisomaltooligosaccharide production from starch by Bacillus circulans T-3040. Appl Microbiol Biotechnol 98:3947–3954
Huber RE, Thompson DJ (1973) Study on a honey bee sucrose exhibiting unusual kinetics and tranglucolytic activity. Biochemistry 12:4011–4020
Ichinose H, Suzuki R, Miyazaki T, Kimura K, Momma M, Suzuki N, Fujimoto Z, Kimura A, Funane K (2017) Paenibacillus sp. 598 K 6-α-glucosyltransferase is essential for cycloisomaltooligosaccharides synthesis from α-(1-4)-glucan. Appl Microbiol Biotechnol 101:4115–4128
Khalikova E, Susi P, Korpela T (2005) Microbial dextran-hydrolyzing enzymes: fundamentals and applications. Microbiol Mol Biol Rev 69:306–325
Kim YM, Kim D (2010) Characterization of novel thermostable dextranase from Thermotoga lettingae TMO. Appl Microbiol Biotechnol 85:581–587
Kim YM, Kimura A, Kim D (2011) Novel quantitative method for the degree of branching in dextran. Food Sci Biotechnol 20:537–541
Kitamura M, Okuyama M, Tanzawa F, Mori H, Kitago Y, Watanabe N, Kimura A, Tanaka I, Yao M (2008) Structural and functional analysis of a glycoside hydrolase family 97 enzyme from Bacteroides thetaiotaomicron. J Biol Chem 283:36328–36337
Kobayashi M, Funane K, Oguma T (1995) Inhibition of dextran and mutan synthesis by cycloisomaltooligosaccharides. Biosci Biotechnol Biochem 59:1861–1865
Mitchell FL, Miles SM, Neres J, Bichenkova EV, Bryce RA (2010) Tryptophan as a molecular shovel in the glycosyl transfer activity of Trypanasoma cruzi trans-sialidase. Biophys J 98:38–40
Neet KE, Ainslie GR Jr (1980) Hysteretic enzymes. In: Purich DL (ed) Methods in enzymology, vol 64. Academic Press, New York, pp 192–226
Oguma T, Horiuchi T, Kobayashi M (1993) Novel cyclic dextrins, cycloisomaltooligosaccharides, from Bacillus sp. T-3040 culture. Biosci Biotechnol Biochem 57:1225–1227
Oguma T, Tobe K, Kobayashi M (1994) Purification and properties of a novel enzyme from Bacillus spp. T-3040, which catalyzes the conversion of dextran to cyclic isomaltooligosaccharides. FEBS Lett 345:135–138
Oguma T, Kitao S, Kobayashi M (2014) Purification and characterization of cycloisomalto-oligosaccharide glucanotransferase and cloning of cit from Bacillus circulans U-155. J Appl Glycosci 61:93–98
Ozaki H, Yamada K (1991) Isolation of Streptomyces sp. producing glucose-tolerant β-glucosidases and properties of the enzyme. Agric Biol Chem 55:979–987
Saburi W, Mori H, Saito S, Okuyama M, Kimura A (2006) Structural elements in dextran glucosidase responsible for high specificity to long chain substrate. Biosci Biotechnol Biochem 1764:688–698
Suzuki R, Terasawa K, Kimura K, Fujimoto Z, Momma M, Kobayashi M, Kimura K, Funane K (2012) Biochemical characterization of a novel cycloisomaltooligosaccharide glucanotransferase from Paenibacillus sp. 598K. Biochem Biophys Acta 1824:919–924
Suzuki N, Kishine N, Fujimoto Z, Sakurai M, Momma M, Ko JA, Nam SH, Kimura A, Kim YM (2016) Crystal structure of thermophilic dextranase from Thermoanaerobacter pseudethanolicus. J Biochem 159:331–339
Wongchawalit J, Yamamoto T, Nakai H, Kim YM, Sato N, Nishimoto N, Okuyama M, Mori H, Saji O, Chanchao C, Wangsiri S, Surarit S, Svasti J, Chiba S, Kimura A (2006) Purification and characterization of α-glucosidase I from Japanese honeybee (Apis cerana japonica) and molecular cloning of its cDNA. Biosci Biotechnol Biochem 70:2889–2898
Yang SJ, Ko JA, Kim HS, Jo MH, Lee HN, Park BR, Kim YM (2018) Biochemical characterization of alkaliphilic cyclodextran glucanotransferase from an alkaliphilic bacterium, Paenibacillus daejeonensis. J Microbiol Biotechnol 28:2029–2035
Acknowledgments
This research was carried out with support from the “Cooperative Research Program for Agriculture Science and Technology Development (Project No. PJ0131262019)” Rural Development Administration, Republic of Korea and Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2015R1D1A1A01058227).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, SJ., Choi, SJ., Park, BR. et al. Thermostable CITase from Thermoanaerobacter thermocopriae shows negative cooperativity. Biotechnol Lett 41, 625–632 (2019). https://doi.org/10.1007/s10529-019-02666-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-019-02666-6