Abstract
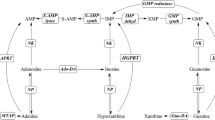
In our search for thermophilic and acid-tolerant nucleoside 2′-deoxyribosyltransferases (NDTs), we found a good candidate in an enzyme encoded by Chroococcidiopsis thermalis PCC 7203 (CtNDT). Biophysical and biochemical characterization revealed CtNDT as a homotetramer endowed with good activity and stability at both high temperatures (50–100 °C) and a wide range of pH values (from 3 to 7). CtNDT recognizes purine bases and their corresponding 2′-deoxynucleosides but is also proficient using cytosine and 2′-deoxycytidine as substrates. These unusual features preclude the strict classification of CtNDT as either a type I or a type II NDT and further suggest that this simple subdivision may need to be updated in the future. Our findings also hint at a possible link between oligomeric state and NDT’s substrate specificity. Interestingly from a practical perspective, CtNDT displays high activity (80–100%) in the presence of several water-miscible co-solvents in a proportion of up to 20% and was successfully employed in the enzymatic production of several therapeutic nucleosides such as didanosine, vidarabine, and cytarabine.





Similar content being viewed by others
References
Alford RF, Leaver-Fay A, Jeliazkov JR, O'Meara MJ, DiMaio FP, Park H, Shapovalov MV, Renfrew PD, Mulligan VK, Kappel K, Labonte JW, Pacella MS, Bonneau R, Bradley P, Dunbrack RL Jr, Das R, Baker D, Kuhlman B, Kortemme T, Gray JJ (2017) The Rosetta all-atom energy function for macromolecular modeling and design. J Chem Theory Comput 13(6):3031–3048. https://doi.org/10.1021/acs.jctc.7b00125
Anand R, Kaminski PA, Ealick SE (2004) Structures of purine 2′-deoxyribosyltransferase, substrate complexes, and the ribosylated enzyme intermediate at 2.0 Å resolution. Biochemistry 43(9):2384–2393. https://doi.org/10.1021/bi035723k
Armstrong SR, Cook WJ, Short SA, Ealick SE (1996) Crystal structures of nucleoside 2-deoxyribosyltransferase in native and ligand-bound forms reveal architecture of the active site. Structure 4(1):97–107
Arroyo M, Torres-Guzmán R, de la Mata I, Castillón MP, Acebal C (2000) Prediction of penicillin V acylase stability in water-organic co-solvent monophasic systems as a function of solvent composition. Enzym Microb Technol 27(1–2):122–126
Bagarolo ML, Porcelli M, Martino E, Feller G, Cacciapuoti G (2015) Multiple disulfide bridges modulate conformational stability and flexibility in hyperthermophilic archaeal purine nucleoside phosphorylase. Biochim Biophys Acta 1854(10 Pt A):1458–1465. https://doi.org/10.1016/j.bbapap.2015.06.010
Biasini M, Bienert S, Waterhouse A, Arnold K, Studer G, Schmidt T, Kiefer F, Gallo Cassarino T, Bertoni M, Bordoli L, Schwede T (2014) SWISS-MODEL: modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Res 42(Web Server issue):W252–W258. https://doi.org/10.1093/nar/gku340
Billi D, Friedmann EI, Helm RF, Potts M (2001) Gene transfer to the desiccation-tolerant cyanobacterium Chroococcidiopsis. J Bacteriol 183(7):2298–2305. https://doi.org/10.1128/JB.183.7.2298-2305.2001
Britos CN, Lapponi MJ, Cappa VA, Rivero CW, Trelles JA (2016) Biotransformation of halogenated nucleosides by immobilized Lactobacillus animalis 2′-N-deoxyribosyltransferase. J Fluor Chem 186:91–96. https://doi.org/10.1016/j.jfluchem.2016.04.012
Brown PH, Schuck P (2006) Macromolecular size-and-shape distributions by sedimentation velocity analytical ultracentrifugation. Biophys J 90(12):4651–4661. https://doi.org/10.1529/biophysj.106.081372
Case DA, Berryman JT, Betz RM, Cerutti DS, Cheatham TE III, Darden TA, Duke RE, Giese TJ, Gohlke H, Götz AW, Homeyer N, Izadi S, Janowski P, Kaus J, Kovalenko A, Lee TS, LeGrand S, Li P, Luchko T, Luo R, Madej B, Merz KM, Monard G, Needham P, Nguyen H, Nguyen HT, Omelyan I, Onufriev A, Roe DR, Roitberg A, Salomon-Ferrer R, Simmerling CL, Smith W, Swails J, Walker RC, Wang J, Wolf RM, Wu X, York DM, Kollman PA (2015) Assisted model building with energy refinement AMBER 14. University of California, San Francisco
Castro GR, Knubovets T (2003) Homogeneous biocatalysis in organic solvents and water-organic mixtures. Crit Rev Biotechnol 23(3):195–231
Cook WJ, Short SA, Ealick SE (1990) Crystallization and preliminary X-ray investigation of recombinant Lactobacillus leichmannii nucleoside deoxyribosyltransferase. J Biol Chem 265(5):2682–2683
Crespo N, Sánchez-Murcia PA, Gago F, Cejudo-Sánches J, Galmes MA, Fernández-Lucas J, Mancheño JM (2017) 2′-Deoxyribosyltransferase from Leishmania mexicana, an efficient biocatalyst for one-pot, one-step synthesis of nucleosides from poorly soluble purine bases. Appl Microbiol Biotechnol 101(19):7187–7200. https://doi.org/10.1007/s00253-017-8450-y
De Benedetti EC, Rivero CW, Britos CN, Lozano ME, Trelles JA (2012) Biotransformation of 2,6-diaminopurine nucleosides by immobilized Geobacillus stearothermophilus. Biotechnol Prog 28(5):1251–1256. https://doi.org/10.1002/btpr.1602
De Clercq E (2005) Recent highlights in the development of new antiviral drugs. Curr Opin Microbiol 8(5):552–560. https://doi.org/10.1016/j.mib.2005.08.010
Del Arco J, Cejudo-Sánches J, Esteban I, Clemente-Suárez VJ, Hormigo D, Perona A, Fernández-Lucas J (2017) Enzymatic production of dietary nucleotides from low-soluble purine bases by an efficient, thermostable and alkali-tolerant biocatalyst. Food Chem 237:605–611. https://doi.org/10.1016/j.foodchem.2017.05.136
DeLano WL (2013) The PyMOL molecular graphics system. 1.3 edn. Schrödinger, LLC, Portland
Fernández-Lucas J, Acebal C, Sinisterra JV, Arroyo M, de la Mata I (2010) Lactobacillus reuteri 2′-deoxyribosyltransferase, a novel biocatalyst for tailoring of nucleosides. Appl Environ Microbiol 76(5):1462–1470. https://doi.org/10.1128/AEM.01685-09
Fernández-Lucas J, Fresco-Taboada A, de la Mata I, Arroyo M (2012) One-step enzymatic synthesis of nucleosides from low water-soluble purine bases in non-conventional media. Bioresour Technol 115:63–69. https://doi.org/10.1016/j.biortech.2011.11.127
Fresco-Taboada A, de la Mata I, Arroyo M, Fernández-Lucas J (2013) New insights on nucleoside 2′-deoxyribosyltransferases: a versatile biocatalyst for one-pot one-step synthesis of nucleoside analogs. Appl Microbiol Biotechnol 97(9):3773–3785. https://doi.org/10.1007/s00253-013-4816-y
Gill SC, von Hippel PH (1989) Calculation of protein extinction coefficients from amino acid sequence data. Anal Biochem 182(2):319–326
Gupta MN, Batra R, Tyagi R, Sharma A (1997) Polarity index: the guiding solvent parameter for enzyme stability in aqueous-organic cosolvent mixtures. Biotechnol Prog 13(3):284–288. https://doi.org/10.1021/bp9700263
Holm L, Laakso LM (2016) Dali server update. Nucleic Acids Res 44(W1):W351–W355. https://doi.org/10.1093/nar/gkw357
Iglesias LE, Lewkowicz ES, Medici R, Bianchi P, Iribarren AM (2015) Biocatalytic approaches applied to the synthesis of nucleoside prodrugs. Biotechnol Adv 33(5):412–434. https://doi.org/10.1016/j.biotechadv.2015.03.009
Kaminski PA (2002) Functional cloning, heterologous expression, and purification of two different N-deoxyribosyltransferases from Lactobacillus helveticus. J Biol Chem 277(17):14400–14407. https://doi.org/10.1074/jbc.M111995200
Kaminski PA, Dacher P, Dugue L, Pochet S (2008) In vivo reshaping the catalytic site of nucleoside 2′-deoxyribosyltransferase for dideoxy- and didehydronucleosides via a single amino acid substitution. J Biol Chem 283(29):20053–20059. https://doi.org/10.1074/jbc.M802706200
Kaul P, Banerjee UC (2008) Predicting enzyme behavior in nonconventional media: correlating nitrilase function with solvent properties. J Ind Microbiol Biotechnol 35(7):713–720. https://doi.org/10.1007/s10295-008-0332-y
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJ (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10(6):845–858. https://doi.org/10.1038/nprot.2015.053
Khmelnitsky YL, Mozhaev VV, Belova AB, Sergeeva MV, Martinek K (1991) Denaturation capacity: a new quantitative criterion for selection of organic solvents as reaction media in biocatalysis. Eur J Biochem 198(1):31–41
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685
Lapponi MJ, Rivero CW, Zinni MA, Britos CN, Trelles JA (2016) New developments in nucleoside analogues biosynthesis: a review. J Mol Catal B Enzym 133:218–233. https://doi.org/10.1016/j.molcatb.2016.08.015
Lawrence KA, Jewett MW, Rosa PA, Gherardini FC (2009) Borrelia burgdorferi bb0426 encodes a 2′-deoxyribosyltransferase that plays a central role in purine salvage. Mol Microbiol 72(6):1517–1529. https://doi.org/10.1111/j.1365-2958.2009.06740.x
Lewkowicz E, Iribarren A (2006) Nucleoside phosphorylases. Curr Org Chem 10(11):1197–1215. https://doi.org/10.2174/138527206777697995
Linder MB, Szilvay GR, Nakari-Setala T, Penttila ME (2005) Hydrophobins: the protein-amphiphiles of filamentous fungi. FEMS Microbiol Rev 29(5):877–896. https://doi.org/10.1016/j.femsre.2005.01.004
López-Blanco JR, Canosa-Valls AJ, Li Y, Chacón P (2016) RCD+: fast loop modeling server. Nucleic Acids Res 44(W1):W395–W400. https://doi.org/10.1093/nar/gkw395
Mikhailopulo I (2007) Biotechnology of nucleic acid constituents—state of the art and perspectives. Curr Org Chem 11(4):317–335. https://doi.org/10.2174/138527207780059330
Mitsukawa Y, Hibi M, Matsutani N, Horinouchi N, Takahashi S, Ogawa J (2017) Enzymatic synthesis of 2′-O-methylribonucleosides with a nucleoside hydrolase family enzyme from Lactobacillus buchneri LBK78. J Biosci Bioeng 123(6):659–664. https://doi.org/10.1016/j.jbiosc.2017.01.005
Miyamoto Y, Masaki T, Chohnan S (2007) Characterization of N-deoxyribosyltransferase from Lactococcus lactis subsp. lactis. Biochim Biophys Acta 1774(10):1323–1330. https://doi.org/10.1016/j.bbapap.2007.08.008
Moreno J, Fágáin CO (1997) Activity and stability of native and modified alanine aminotransferase in cosolvent systems and denaturants. J Mol Catal B Enzym 2(6):271–279. https://doi.org/10.1016/s1381-1177(96)00018-5
Okuyama K, Shibuya S, Hamamoto T, Noguchi T (2003) Enzymatic synthesis of 2′-deoxyguanosine with nucleoside deoxyribosyltransferase-II. Biosci Biotechnol Biochem 67(5):989–995. https://doi.org/10.1271/bbb.67.989
Oliva C, Sánchez-Murcia PA, Rico E, Bravo A, Menéndez M, Gago F, Jiménez-Ruiz A (2017) Structure-based domain assignment in Leishmania infantum EndoG: characterization of a pH-dependent regulatory switch and a C-terminal extension that largely dictates DNA substrate preferences. Nucleic Acids Res 45(15):9030–9045. https://doi.org/10.1093/nar/gkx629
Parker WB (2009) Enzymology of purine and pyrimidine antimetabolites used in the treatment of cancer. Chem Rev 109(7):2880–2893. https://doi.org/10.1021/cr900028p
Patel RN (2017) Biocatalysis for synthesis of pharmaceuticals. Bioorg Med Chem 26:1252–1274. https://doi.org/10.1016/j.bmc.2017.05.023
Salomon-Ferrer R, Götz AW, Poole D, Le Grand S, Walker RC (2013) Routine microsecond molecular dynamics simulations with AMBER on GPUs. 2. Explicit solvent particle mesh Ewald. J Chem Theory Comput 9(9):3878–3888. https://doi.org/10.1021/ct400314y
Sikowitz MD, Cooper LE, Begley TP, Kaminski PA, Ealick SE (2013) Reversal of the substrate specificity of CMP N-glycosidase to dCMP. Biochemistry 52(23):4037–4047. https://doi.org/10.1021/bi400316p
Somervuo P, Holm L (2015) SANSparallel: interactive homology search against Uniprot. Nucleic Acids Res 43(W1):W24–W29. https://doi.org/10.1093/nar/gkv317
Van Holde KE (1985) Physical biochemistry, 2nd edn. Prentice-Hall, Englewood Cliffs
Vichier-Guerre S, Dugué L, Bonhomme F, Pochet S (2016) Expedient and generic synthesis of imidazole nucleosides by enzymatic transglycosylation. Org Biomol Chem 14(14):3638–3653. https://doi.org/10.1039/c6ob00405a
Yokozeki K, Tsuji T (2000) A novel enzymatic method for the production of purine-2′-deoxyribonucleosides. J Mol Catal B Enzym 10(1–3):207–213. https://doi.org/10.1016/s1381-1177(00)00121-1
Funding
This work was supported by grants SAN151610 from the Santander Foundation and 2017/UEM23 from Universidad Europea de Madrid (to J.F.-L.) and SAF2015-64629-C2-2-R from the Spanish MEC/MICINN (to F.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals by any of the authors.
Electronic supplementary material
ESM 1
(PDF 995 kb)
Rights and permissions
About this article
Cite this article
Del Arco, J., Sánchez-Murcia, P.A., Mancheño, J.M. et al. Characterization of an atypical, thermostable, organic solvent- and acid-tolerant 2′-deoxyribosyltransferase from Chroococcidiopsis thermalis. Appl Microbiol Biotechnol 102, 6947–6957 (2018). https://doi.org/10.1007/s00253-018-9134-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9134-y