Abstract
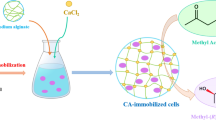
Immobilization of cis-epoxysuccinate hydrolase-containing E. coli for d(−)-tartaric acid production was screened by various methods. The highest recovery of activity was obtained by entrapment in κ-carrageenan gel. 23.6 g biomass/l and 43.4 g κ-carrageenan/l were the best immobilization conditions optimized by response surface methodology with 83% yield (114 U/g). Cell autolysis was observed after immobilization. Immobilized cells showed high pH (5–10) stability, thermal (up to 65°C) stability, conversion rate (>99.5%), enantioselectivity (ee > 99.6%), and were less affected by metal ions and surfactants compared with free cells. Conversion rate for immobilized cells preserved 93% after 10 repeated batches (5% for free cells).




Similar content being viewed by others
References
Bucko M, Vikartovska A, Lacik I, Kollarikova G, Gemeiner P, Patoprsty V, Brygin M (2005) Immobilization of a whole-cell epoxide-hydrolyzing biocatalyst in sodium alginate-cellulose sulfate-poly(methylene-co-guanidine) capsules using a controlled encapsulation process. Enzyme Microb Technol 36:118–126
Chibata I, Tosa T, Sato T (1974) Immobilized aspartase-containing microbial cells: preparation and enzymatic properties. Appl Environ Microbiol 27:878–885
End N, Schöning KU (2004) Immobilized biocatalysts in industrial research and production. Top Curr Chem 242:273–317
Gåserød O, Smidsrød O, Skjåk-Bræk G (1998) Microcapsules of alginate-chitosan-I: a quantitative study of the interaction between alginate and chitosan. Biomaterials 19:1815–1825
Ghosh AK, Koltun ES, Bilcer G (2001) Tartaric acid and tartrates in the synthesis of bioactive molecules. Synthesis 9:1281–1301
Kroutil W, Orru RVA, Faber K (1998) Stabilization of Nocardia EH1 epoxide hydrolase by immobilization. Biotechnol Lett 20:373–377
Kurillova L, Gemeiner P, Vikartovska A, Mikova H, Rosenberg M, Ilavsky M (2000) Calcium pectate gel beads for cell entrapment.6. Morphology of stabilized and hardened calcium pectate gel beads with cells for immobilized biotechnology. J Microencapsul 17:279–296
Pan HF, Xie ZP, Bao WN, Zhang JG (2008a) Isolation and identification of a novel cis-epoxysuccinate hydrolase-producing Bordetella sp. BK-52 and optimization of enzyme production. Wei Sheng Wu Xue Bao 48:1075–1081
Pan HF, Xie ZP, Bao WN, Zhang JG (2008b) Optimization of culture conditions to enhance cis-epoxysuccinate hydrolase production in Escherichia coli by response surface methodology. Biochem Eng J 42:133–138
Rosenberg M, Miková H, Krištofíková L (1999) Production of l-tartaric acid by immobilized bacterial cells Nocardia tartaricans. Biotechnol Lett 21:491–495
Sun ZH, Zheng P, Dai XT, Li H, Jin M (1995) Production of l(+)-tartaric acid by immobilized Nocardia tartaricans SW13–57. Sheng Wu Gong Cheng Xue Bao 11:372–376
Takata I, Yamamoto K, Tosa T, Chibata I (1980) Immobilization of Brevibacterium flavum with carrageenan and its application for continuous production of l-malic acid. Enzyme Microb Technol 2:30–36
Vikartovska A, Bucko M, Gemeiner P, Nahalka J, Patoprsty V, Hrabarova E (2004) Flow calorimetry-a useful tool for determination of immobilized cis-epoxysuccinate hydrolase activity from Nocardia tartaricans. Artif Cells Blood Substit Biotechnol 32:77–89
Willaert R, De Vuyst L (2006) Continuous production of l(+)-tartaric acid from cis-epoxysuccinate using a membrane recycle reactor. Appl Microbiol Biotechnol 71:155–163
Zhang JG, Qian YJ (2000) Production of l-(+)-tartaric acid by immobilized Corynebacterium sp. JZ-1. Sheng Wu Gong Cheng Xue Bao 16:188–192
Acknowledgment
This work was funded by Hangzhou Bioking Biochemical Engineering Co., Ltd.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pan, H., Bao, W., Xie, Z. et al. Immobilization of Escherichia coli cells with cis-epoxysuccinate hydrolase activity for d(−)-tartaric acid production. Biotechnol Lett 32, 235–241 (2010). https://doi.org/10.1007/s10529-009-0134-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-009-0134-y