Abstract
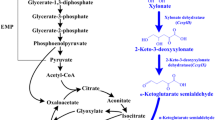
Escherichia coli W3110 was previously engineered to produce xylitol from a mixture of glucose plus xylose by expressing xylose reductase (CbXR) and deleting xylulokinase (ΔxylB), combined with either plasmid-based expression of a xylose transporter (XylE or XylFGH) (Khankal et al., J Biotechnol, 2008) or replacing the native crp gene with a mutant (crp*) that alleviates glucose repression of xylose transport (Cirino et al., Biotechnol Bioeng 95:1167–1176, 2006). In this study, E. coli K-12 strains W3110 and MG1655 and wild-type E. coli B were compared as platforms for xylitol production from glucose-xylose mixtures using these same strategies. The engineered strains were compared in fed-batch fermentations and as non-growing resting cells. Expression of CRP* in the E. coli B strains tested was unable to enhance xylose uptake in the presence of glucose. Xylitol production was similar for the (crp*, ΔxylB)-derivatives of W3110 and MG1655 expressing CbXR (average specific productivities of 0.43 g xylitol g cdw−1 h−1 in fed-batch fermentation). In contrast, results varied substantially between different ΔxylB-derivative strains co-expressing either XylE or XylFGH. The differences in genetic background between these host strains can therefore profoundly influence metabolic engineering strategies.


Similar content being viewed by others
References
Aiba H, Nakamura T, Mitani H et al (1985) Mutations that alter the allosteric nature of cAMP receptor protein of Escherichia coli. EMBO J 4:3329–3332
Atlung T, Nielsen HV, Hansen FG (2002) Characterisation of the allelic variation in the rpoS gene in thirteen K-12 and six other non-pathogenic Escherichia coli strains. Mol Genet Genomics 266:873–881
Belduz AO, Lee EJ, Harman JG (1993) Mutagenesis of the cyclic AMP receptor protein of Escherichia coli: targeting positions 72 and 82 of the cyclic nucleotide binding pocket. Nucleic Acids Res 21:1827–1835
Blattner FR, Plunkett G, Bloch CA et al (1997) The complete genome sequence of Escherichia coli K-12. Science 277:1453–1469
Causey TB, Zhou S, Shanmugam KT et al (2003) Engineering the metabolism of Escherichia coli W3110 for the conversion of sugar to redox-neutral and oxidized products: Homoacetate production. Proc Natl Acad Sci USA 100:825–832
Cirino PC, Chin JW, Ingram LO (2006) Engineering Escherichia coli for xylitol production from glucose-xylose mixtures. Biotechnol Bioeng 95:1167–1176
Dien BS, Nichols NN, Bothast RJ (2001) Recombinant Escherichia coli engineered for production of L-lactic acid from hexose and pentose sugars. J Ind Microbiol Biotechnol 27:259–264
Eppler T, Boos W (1999) Glycerol-3-phosphate-mediated repression of malT in Escherichia coli does not require metabolism, depends on enzyme IIA(Glc) and is mediated by cAMP levels. Mol Microbiol 33:1221–1231
Hayashi K, Morooka N, Yamamoto Y et al (2006) Highly accurate genome sequences of Escherichia coli K-12 strains MG1655 and W3110. Mol Syst Biol 2:2006.0007. doi:10.1038/msb4100049
Hengge-Aronis R (2002) Signal transduction and regulatory mechanisms involved in control of the sigma(S) (RpoS) subunit of RNA polymerase. Microbiol Mol Biol Rev 66:373–395
Herring CD, Palsson BO (2007) An evaluation of comparative genome sequencing (CGS) by comparing two previously-sequenced bacterial genomes. BMC Genomics 8:274. doi:10.1186/1471-2164-8-274
Hogema BM, Arents JC, Bader R et al (1998) Inducer exclusion in Escherichia coli by non-PTS substrates: The role of the PEP to pyruvate ratio in determining the phosphorylation state of enzyme IIA(Glc). Mol Microbiol 30:487–498
Jishage M, Ishihama A (1997) Variation in RNA polymerase sigma subunit composition within different stocks of Escherichia coli W3110. J Bacteriol 179:959–963
Karimova G, Ladant D, Ullmann A (2004) Relief of catabolite repression in a cAMP-independent catabolite gene activator mutant of Escherichia coli. Res Microbiol 155:76–79
Khankal R, Chin JW, Cirino PC (2008) Role of xylose transporters in xylitol production from engineered Escherichia coli. J Biotechnol 134:246–252
Kwon SG, Park SW, Oh DK (2006) Increase of xylitol productivity by cell-recycle fermentation of Candida tropicalis using submerged membrane bioreactor. J Biosci Bioeng 101:13–18
Luli GW, Strohl WR (1990) Comparison of growth, acetate production, and acetate inhibition of Escherichia coli strains in batch and fed-batch fermentations. Appl Environ Microbiol 56:1004–1011
Noronha SB, Yeh HJC, Spande TF et al (2000) Investigation of the TCA cycle and the glyoxylate shunt in Escherichia coli BL21 and JM109 using C-13-NMR/MS. Biotechnol. Bioeng 68:316–327
Phue JN, Shiloach J (2004) Transcription levels of key metabolic genes are the cause for different glucose utilization pathways in E. coli B (BL21) and E. coli K (JM109). J Biotechnol 109:21–30
Phue JN, Kedem B, Jaluria P et al (2007) Evaluating microarrays using a semiparametric approach: Application to the central carbon metabolism of Escherichia coli BL21 and JM109. Genomics 89:300–305
Sabourin D, Beckwith J (1975) Deletion of the Escherichia coli crp gene. J Bacteriol 122:338–340
Saier MH, Crasnier M (1996) Inducer exclusion and the regulation of sugar transport. Res Microbiol 147:482–489
Shiloach J, Kaufman J, Guillard AS et al (1996) Effect of glucose supply strategy on acetate accumulation, growth, and recombinant protein production by Escherichia coli BL21 (lambda DE3) and Escherichia coli JM109. Biotechnol Bioeng 49:421–428
Song SG, Park C (1997) Organization and regulation of the D-xylose operons in Escherichia coli K-12: XylR acts as a transcriptional activator. J Bacteriol 179:7025–7032
Subbarayan PR, Sarkar M (2004) A comparative study of variation in codon 33 of the rpoS gene in Escherichia coli K-12 stocks: Implications for the synthesis of sigma(S). Mol Genet Genomics 270:533–538
Vijayendran C, Polen T, Wendisch VF et al (2007) The plasticity of global proteome and genome expression analyzed in closely related W3110 and MG1655 strains of a well-studied model organism, Escherichia coli K-12. J Biotechnol 128:747–761
Acknowledgments
We acknowledge W. Boos (University of Konstanz) for providing strain ET25. This research was made possible by a grant from the NSF (BES0519516) and support from the Penn State Associate Vice President for Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Reza Khankal and Francesca Luziatelli contributed equally to the paper.
Rights and permissions
About this article
Cite this article
Khankal, R., Luziatelli, F., Chin, J.W. et al. Comparison between Escherichia coli K-12 strains W3110 and MG1655 and wild-type E. coli B as platforms for xylitol production. Biotechnol Lett 30, 1645–1653 (2008). https://doi.org/10.1007/s10529-008-9720-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-008-9720-7