Abstract
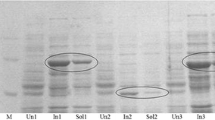
A 1,125-bp long ORF encoding a novel gentisate 1,2-dioxygenase with two-domain bicupins was cloned from Silicibacter pomeroyi DSS-3 and expressed in Escherichia coli. The resulting product was purified to homogeneity and partially characterized. Non-reductive SDS-PAGE and gel filtration showed that the active recombinant gentisate 1,2-dioxygenase had an estimated molecular mass of 132 kDa, and reductive SDS-PAGE indicated an approximate size of 45 kDa. The enzyme thus appears to be a homotrimeric protein. This is in contrast to the homotetrameric or dimeric protein of the gentisate 1,2-dioxygenases that have been characterized thus far. The K m and K cat/K m for gentisate were 12 μM and 653 × 104 M−1 s−1; the pI was 4.6–4.8. It was optimally active at 40°C and pH 8.0.






Similar content being viewed by others
References
Adams MA, Singh VK, Keller BO, Jia Z (2006) Structural and biochemical characterization of gentisate 1,2-dioxygenase from Escherichia coli O157:H7. Mol Microbiol 61:1469–1484
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Chua CH, Feng YM, Yeo CC, Khoo HE, Poh CL (2001) Identification of amino acid residues essential for catalytic activity of gentisate 1,2-dioxygenase from Pseudomonas alcaligenes NCIB 9867. FEMS Microbiol Lett 204:141–146
Clewley JP (1995) Macintosh sequence analysis software. DNAstar’s LaserGene. Mol Biotechnol 3:221–224
Crawford RL, Hutton SW, Chapman PJ (1975) Purification and properties of gentisate 1,2-dioxygenase from Moraxella osloensis. J Bacteriol 121:794–799
Feng YM, Khoo HE, Poh CL (1999) Purification and characterization of gentisate 1,2-dioxygenases from Pseudomonas alcaligenes NCIB 9867 and Pseudomonas putida NCIB 9869. Appl Environ Microbiol 65:946–950
Fu W, Oriel P (1998) Gentisate 1,2-dioxygenase from Haloferax sp. D1227. Extremophiles 2:439–446
Gao X, Tan CL, Yeo CC, Poh CL (2005) Molecular and biochemical characterization of the xlnD-encoded 3-hydroxybenzoate 6-hydroxylase involved in the degradation of 2,5-xylenol via the gentisate pathway in Pseudomonas alcaligenes NCIMB 9867. J Bacteriol 187:7696–7702
Harpel MR, Lipscomb JD (1990) Gentisate 1,2-dioxygenase from Pseudomonas. Purification, characterization, and comparison of the enzymes from Pseudomonas testosteroni and Pseudomonas acidovorans. J Biol Chem 265:6301–6311
Jeanmougin F, Thompson JD, Gouy M, Higgins DG, Gibson TJ (1998) Multiple sequence alignment with Clustal X. Trends Biochem Sci 23:403–405
Jones DC, Cooper RA (1990) Catabolism of 3-hydroxybenzoate by the gentisate pathway in Klebsiella pneumoniae M5a1. Arch Microbiol 54:489–495
Kloetzel MC, Abadir BY (1955) Synthetic analogs of cortical hormones. Part II. 3-substituted α-2,5-trihydroxyacetophenone derivatives. J Am Chem Soc 77:3823–3826
Kumar SK, Tamura K, Nei M (2004) MEGA3: an integrated software for molecular evolutionary genetics analysis and sequence alignment. Brief Bioinform 5:150–163
Lack L (1959) The enzymic oxidation of gentisic acid. Biochim Biophys Acta 34:117–123
Luo S, Liu DQ, Liu H, Zhou NY (2006) Site-directed mutagenesis of gentisate 1,2-dioxygenases from Klebsiella pneumoniae M5a1 and Ralstonia sp. strain U2. Microbiol Res 161:138–144
Schwede T, Kopp J, Guex N, Peitsch MC (2003) SWISS-MODEL: an automated protein homology-modeling server. Nucleic Acids Res 31:3381–3385
Shen XH, Jiang CY, Huang Y, Liu ZP, Liu SJ (2005) Functional identification of novel genes involved in the glutathione-independent gentisate pathway in Corynebacterium glutamicum. Appl Environ Microbiol 71:3442–3452
Studier FW, Moffatt BA (1986) Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J Mol Biol 189:113–130
Suarez M, Ferrer E, Martin M (1996) Purification and biochemical characterization of gentisate 1,2-dioxygenase from Klebsiella pneumoniae M5a1. FEMS Microbiol Lett 143:89–95
Werwath J, Arfmann HA, Pieper DH, Timmis KN, Wittich RM (1998) Biochemical and genetic characterization of a gentisate 1,2-dioxygenase from Sphingomonas sp. strain RW5. J Bacteriol 180:4171–4176
Wheelis ML, Palleroni NJ, Stanier RY (1967) The metabolism of aromatic acids by Pseudomonas testosteroni and Pseudomonas acidovorans. Arch Mikrobiol 59:302–314
Zhou NY, Fuenmayor SL, Williams PA (2001) nag genes of Ralstonia (formerly Pseudomonas) sp. strain U2 encoding enzymes for gentisate catabolism. J Bacteriol 183:700–708
Acknowledgments
This work was supported by Postdoctoral Science Foundation of China (Grant No. 2005038032). We also thank F. Xiang, Z. Li and L.Y. Zhou for their assistance during this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, D., Zhu, T., Fan, L. et al. Identification of a novel gentisate 1,2-dioxygenase from Silicibacter pomeroyi . Biotechnol Lett 29, 1529–1535 (2007). https://doi.org/10.1007/s10529-007-9421-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-007-9421-7