Abstract
Epigenetic regulation plays an essential role in immunity and inflammation in endometriosis. In this study, we aimed to explore differences in m6A regulators between endometriosis patients and normal women and analyze the effect of m6A modification on immune and inflammatory microenvironment. The samples for analysis were downloaded from the Gene Expression Omnibus database, including ectopic endometrium (EC), eutopic endometrium (EU), and normal eutopic endometrium (NM) samples from non-endometriosis women. The validation process involved utilizing our previous RNA-sequencing data. Subsequently, a correlation analysis was performed to ascertain the relationship between m6A and the inflammatory microenvironment profile, encompassing infiltrating immunocytes, immune-inflammation reaction gene sets, and human leukocyte antigen genes. LASSO analyses were used to develop risk signature. The findings of this study indicate that the m6A regulators FTO were observed to be significantly up-regulated, while YTHDF2, CBLL1, and METTL3 were down-regulated in endometriosis tissues. The CIBERSORT analysis revealed that the local inflammatory microenvironment of ectopic lesions plays a crucial role in the development of endometriosis. Notably, M2 macrophages exhibited a significant difference between the EC and NM groups. Moreover, M2 macrophages demonstrated a positive correlation with FTO (0.39) and a negative correlation with CBLL1 (− 0.35). Furthermore, consistent clustering of EC and EU samples resulted in the identification of three distinct cell subtypes. Among different cell subtypes, significant differences were in immunoinfiltrating cells, plasma cells, naive CD4 T cells, memory activated CD4 T cells, gamma delta T cells, resting NK cells and activated NK cells but not in macrophages. Furthermore, the identification of various compounds capable of targeting these m6A genes was achieved. In conclusions, our integrated bioinformatics analysis results demonstrated that m6A-related genes METTL3, CBLL1 and YTHDF2 may be useful biomarkers for endometriosis in ectopic endometrium. The potential therapeutic approach of targeting m6A regulators holds promise for the treatment of endometriosis.








Similar content being viewed by others
Data Availability
Publicly available datasets from GEO datasets were analyzed in this study. All data are contained within the manuscript and additional files. And the entire RNA-seq dataset is available at the sequencing Read Archive (SRA) database (https://www.ncbi.nlm.nih.gov/sra/PRJNA769152).
Abbreviations
- CBLL1:
-
Casitas B-lineage proto-oncogene like 1
- CCK-8:
-
Cell counting kit-8
- ELAVL1:
-
ELAV like RNA binding protein 1
- EMT:
-
Epithelial-to-mesenchymal transition
- FTO:
-
Fat mass and obesity-associated protein
- HLAs:
-
Human leukocyte antigen gene
- LRPPRC:
-
Leucine-rich pentatricopeptide repeat containing
- LASSO:
-
Least absolute shrinkage and selection operator
- METTL3/14:
-
Methyltransferase-like 3/14
- m6A:
-
N6-methyladenosine
- OS:
-
Overall survival
- ROC:
-
Receiver operating characteristic curve
- RPMI-1640:
-
Roswell park memorial institute 1640
- YTHDC1, 2:
-
YT521-B homology domain-containing protein 1 1/2
- YTHDF1/2/3:
-
YTH N6-methyladenosine RNA binding protein F1/2/3
- WTAP:
-
Wilms’ tumor-associated protein
References
Agostinis C, Balduit A, Mangogna A, Zito G, Romano F, Ricci G, Kishore U, Bulla R (2020) Immunological basis of the endometriosis: the complement system as a potential therapeutic target. Front Immunol 11:599117. https://doi.org/10.3389/fimmu.2020.599117
Aparicio LA, Valladares M, Blanco M, Alonso G, Figueroa A (2012) Biological influence of Hakai in cancer: a 10-year review. Cancer Metastasis Rev 31(1–2):375–386. https://doi.org/10.1007/s10555-012-9348-x
Bacci M, Capobianco A, Monno A, Cottone L, Di Puppo F, Camisa B, Mariani M, Brignole C, Ponzoni M, Ferrari S, Panina-Bordignon P, Manfredi AA, Rovere-Querini P (2009) Macrophages are alternatively activated in patients with endometriosis and required for growth and vascularization of lesions in a mouse model of disease. Am J Pathol 175(2):547–556. https://doi.org/10.2353/ajpath.2009.081011
Baka S, Frangou-Plemenou M, Panagiotopoulou E, Makrakis E, Kaltsakas G, Hassiakos D, Kondi-Pafiti A (2011) The expression of human leukocyte antigens class I and II in women with endometriosis or adenomyosis. Gynecol Endocrinol 27(6):419–424. https://doi.org/10.3109/09513590.2010.495429
Becker CM, Bokor A, Heikinheimo O, Horne A, Jansen F, Kiesel L, King K, Kvaskoff M, Nap A, Petersen K, Saridogan E, Tomassetti C, van Hanegem N, Vulliemoz N, Vermeulen N (2022) ESHRE Guideline: Endometriosis Hum Reprod Open 2022(2):c9. https://doi.org/10.1093/hropen/hoac009
Bertero A, Brown S, Madrigal P, Osnato A, Ortmann D, Yiangou L, Kadiwala J, Hubner NC, de Los MI, Sadée C, Lenaerts AS, Nakanoh S, Grandy R, Farnell E, Ule J, Stunnenberg HG, Mendjan S, Vallier L (2018) The SMAD2/3 interactome reveals that TGF-β controls m(6)A mRNA methylation in pluripotency. Nature 555(7695):256–259. https://doi.org/10.1038/nature25784
Bian PP, Liu SY, Luo QP, Xiong ZT (2022) YTHDF2 is a novel diagnostic marker of endometrial adenocarcinoma and endometrial atypical hyperplasia/ intraepithelial neoplasia. Pathol Res Pract 234:153919. https://doi.org/10.1016/j.prp.2022.153919
Budi EH, Duan D, Derynck R (2017) Transforming growth factor-β receptors and smads: regulatory complexity and functional versatility. Trends Cell Biol 27(9):658–672. https://doi.org/10.1016/j.tcb.2017.04.005
Capobianco A, Rovere-Querini P (2013) Endometriosis, a disease of the macrophage. Front Immunol 4:9. https://doi.org/10.3389/fimmu.2013.00009
Chen B, Khodadoust MS, Liu CL, Newman AM, Alizadeh AA (2018) Profiling tumor infiltrating immune cells with CIBERSORT. Methods Mol Biol 1711:243–259. https://doi.org/10.1007/978-1-4939-7493-1_12
Chen XY, Zhang J, Zhu JS (2019) The role of m(6)A RNA methylation in human cancer. Mol Cancer 18(1):103. https://doi.org/10.1186/s12943-019-1033-z
Chen J, Fang Y, Xu Y, Sun H (2022) Role of m6A modification in female infertility and reproductive system diseases. Int J Biol Sci 18(9):3592–3604. https://doi.org/10.7150/ijbs.69771
Gabriel M, Fey V, Heinosalo T, Adhikari P, Rytkönen K, Komulainen T, Huhtinen K, Laajala TD, Siitari H, Virkki A, Suvitie P, Kujari H, Aittokallio T, Perheentupa A, Poutanen M (2020) A relational database to identify differentially expressed genes in the endometrium and endometriosis lesions. Sci Data 7(1):284. https://doi.org/10.1038/s41597-020-00623-x
Gentles AJ, Newman AM, Liu CL, Bratman SV, Feng W, Kim D, Nair VS, Xu Y, Khuong A, Hoang CD, Diehn M, West RB, Plevritis SK, Alizadeh AA (2015) The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat Med 21(8):938–945. https://doi.org/10.1038/nm.3909
Gou Y, Wang H, Wang T, Wang H, Wang B, Jiao N, Yu Y, Cao Y, Wang H, Zhang Z (2023) Ectopic endometriotic stromal cells-derived lactate induces M2 macrophage polarization via Mettl3/Trib1/ERK/STAT3 signalling pathway in endometriosis. Immunology 168(3):389–402. https://doi.org/10.1111/imm.13574
Gu X, Zhang Y, Li D, Cai H, Cai L, Xu Q (2020) N6-methyladenosine demethylase FTO promotes M1 and M2 macrophage activation. Cell Signal 69:109553. https://doi.org/10.1016/j.cellsig.2020.109553
Guo L, Yang H, Zhou C, Shi Y, Huang L, Zhang J (2021) N6-methyladenosine RNA modification in the tumor immune microenvironment: novel implications for immunotherapy. Front Immunol 12:773570. https://doi.org/10.3389/fimmu.2021.773570
Hever A, Roth RB, Hevezi P, Marin ME, Acosta JA, Acosta H, Rojas J, Herrera R, Grigoriadis D, White E, Conlon PJ, Maki RA, Zlotnik A (2007) Human endometriosis is associated with plasma cells and overexpression of B lymphocyte stimulator. Proc Natl Acad Sci U S A 104(30):12451–12456. https://doi.org/10.1073/pnas.0703451104
Hogg C, Horne AW, Greaves E (2020) Endometriosis-associated macrophages: origin, phenotype, and function. Front Endocrinol (lausanne) 11:7. https://doi.org/10.3389/fendo.2020.00007
Hu X, Li J, Fu M, Zhao X, Wang W (2021) The JAK/STAT signaling pathway: from bench to clinic. Signal Transduct Target Ther 6(1):402. https://doi.org/10.1038/s41392-021-00791-1
Jiang L, Zhang M, Wu J, Wang S, Yang X, Yi M, Zhang X, Fang X (2020) Exploring diagnostic m6A regulators in endometriosis. Aging 12(24):25916–25938. https://doi.org/10.18632/aging.202163
Johnson NP, Hummelshoj L, Adamson GD, Keckstein J, Taylor HS, Abrao MS, Bush D, Kiesel L, Tamimi R, Sharpe-Timms KL, Rombauts L, Giudice LC (2017) World endometriosis society consensus on the classification of endometriosis. Hum Reprod 32(2):315–324. https://doi.org/10.1093/humrep/dew293
Koninckx PR, Ussia A, Adamyan L, Wattiez A, Gomel V, Martin DC (2019) Pathogenesis of endometriosis: the genetic/epigenetic theory. Fertil Steril 111(2):327–340. https://doi.org/10.1016/j.fertnstert.2018.10.013
Kotlyar AM, Mamillapalli R, Flores VA, Taylor HS (2021) Tofacitinib alters STAT3 signaling and leads to endometriosis lesion regression. Mol Hum Reprod. https://doi.org/10.1093/molehr/gaab016
Koukoura O, Sifakis S, Spandidos DA (2016) DNA methylation in endometriosis (Review). Mol Med Rep 13(4):2939–2948. https://doi.org/10.3892/mmr.2016.4925
Laganà AS, Salmeri FM, Ban FH, Ghezzi F, Vrtačnik-Bokal E, Granese R (2020) Evaluation of M1 and M2 macrophages in ovarian endometriomas from women affected by endometriosis at different stages of the disease. Gynecol Endocrinol 36(5):441–444. https://doi.org/10.1080/09513590.2019.1683821
Li R, Song Y, Chen X, Chu M, Wang ZW, Zhu X (2021a) METTL3 increases cisplatin chemosensitivity of cervical cancer cells via downregulation of the activity of RAGE. Mol Ther Oncolytics 22:245–255. https://doi.org/10.1016/j.omto.2021.05.013
Li X, Xiong W, Long X, Dai X, Peng Y, Xu Y, Zhang Z, Zhang L, Liu Y (2021b) Inhibition of METTL3/m6A/miR126 promotes the migration and invasion of endometrial stromal cells in endometriosis. Biol Reprod. https://doi.org/10.1093/biolre/ioab152
Li Y, Peng H, Jiang P, Zhang J, Zhao Y, Feng X, Pang C, Ren J, Zhang H, Bai W, Liu W (2022a) Downregulation of methyltransferase-Like 14 promotes ovarian cancer cell proliferation through stabilizing TROAP mRNA. Front Oncol 12:824258. https://doi.org/10.3389/fonc.2022.824258
Li Y, Su R, Deng X, Chen Y, Chen J (2022b) FTO in cancer: functions, molecular mechanisms, and therapeutic implications. Trends Cancer 8(7):598–614. https://doi.org/10.1016/j.trecan.2022.02.010
Lin X, Chai G, Wu Y, Li J, Chen F, Liu J, Luo G, Tauler J, Du J, Lin S, He C, Wang H (2019) RNA m(6)A methylation regulates the epithelial mesenchymal transition of cancer cells and translation of Snail. Nat Commun 10(1):2065. https://doi.org/10.1038/s41467-019-09865-9
Lin Z, Xu Q, Miao D, Yu F (2021) An inflammatory response-related gene signature can impact the immune status and predict the prognosis of hepatocellular carcinoma. Front Oncol 11:644416. https://doi.org/10.3389/fonc.2021.644416
Liu J, Eckert MA, Harada BT, Liu SM, Lu Z, Yu K, Tienda SM, Chryplewicz A, Zhu AC, Yang Y, Huang JT, Chen SM, Xu ZG, Leng XH, Yu XC, Cao J, Zhang Z, Liu J, Lengyel E, He C (2018) m(6)A mRNA methylation regulates AKT activity to promote the proliferation and tumorigenicity of endometrial cancer. Nat Cell Biol 20(9):1074–1083. https://doi.org/10.1038/s41556-018-0174-4
Liu M, Zhao Z, Cai Y, Bi P, Liang Q, Yan Y, Xu Z (2021) YTH domain family: potential prognostic targets and immune-associated biomarkers in hepatocellular carcinoma. Aging 13(21):24205–24218. https://doi.org/10.18632/aging.203674
Mak MP, Tong P, Diao L, Cardnell RJ, Gibbons DL, William WN, Skoulidis F, Parra ER, Rodriguez-Canales J, Wistuba II, Heymach JV, Weinstein JN, Coombes KR, Wang J, Byers LA (2016) A patient-derived, pan-cancer EMT signature identifies global molecular alterations and immune target enrichment following epithelial-to-mesenchymal transition. Clin Cancer Res 22(3):609–620. https://doi.org/10.1158/1078-0432.CCR-15-0876
Oerum S, Meynier V, Catala M, Tisné C (2021) A comprehensive review of m6A/m6Am RNA methyltransferase structures. Nucleic Acids Res 49(13):7239–7255. https://doi.org/10.1093/nar/gkab378
Parisi L, Gini E, Baci D, Tremolati M, Fanuli M, Bassani B, Farronato G, Bruno A, Mortara L (2018) Macrophage polarization in chronic inflammatory diseases: killers or builders? J Immunol Res 2018:8917804. https://doi.org/10.1155/2018/8917804
Ramírez-Pavez TN, Martínez-Esparza M, Ruiz-Alcaraz AJ, Marín-Sánchez P, Machado-Linde F, García-Peñarrubia P (2021) The role of peritoneal macrophages in endometriosis. Int J Mol Sci 22(19):10792. https://doi.org/10.3390/ijms221910792
Shen L, Wang Q, Huang W, Wang Q, Yuan Q, Huang Y, Lei H (2011) High prevalence of endometrial polyps in endometriosis-associated infertility. Fertil Steril 95(8):2722–2724. https://doi.org/10.1016/j.fertnstert.2011.04.067
Szukiewicz D (2022) Epigenetic regulation and T-cell responses in endometriosis—something other than autoimmunity. Front Immunol 13:943839. https://doi.org/10.3389/fimmu.2022.943839
Tong J, Wang X, Liu Y, Ren X, Wang A, Chen Z, Yao J, Mao K, Liu T, Meng FL, Pan W, Zou Q, Liu J, Zhou Y, Xia Q, Flavell RA, Zhu S, Li HB (2021) Pooled CRISPR screening identifies m(6)A as a positive regulator of macrophage activation. Sci Adv. https://doi.org/10.1126/sciadv.abd4742
Vallvé-Juanico J, Houshdaran S, Giudice LC (2019) The endometrial immune environment of women with endometriosis. Hum Reprod Update 25(5):564–591. https://doi.org/10.1093/humupd/dmz018
Vigano P, Ottolina J, Bartiromo L, Bonavina G, Schimberni M, Villanacci R, Candiani M (2020) Cellular components contributing to fibrosis in endometriosis: a literature review. J Minim Invasive Gynecol 27(2):287–295. https://doi.org/10.1016/j.jmig.2019.11.011
Vitale I, Manic G, Coussens LM, Kroemer G, Galluzzi L (2019) Macrophages and metabolism in the tumor microenvironment. Cell Metab 30(1):36–50. https://doi.org/10.1016/j.cmet.2019.06.001
Wan S, Sun Y, Zong J, Meng W, Yan J, Chen K, Wang S, Guo D, Xiao Z, Zhou Q, Yin Z, Yang M (2023) METTL3-dependent m(6)A methylation facilitates uterine receptivity and female fertility via balancing estrogen and progesterone signaling. Cell Death Dis 14(6):349. https://doi.org/10.1038/s41419-023-05866-1
Wang X, Feng J, Xue Y, Guan Z, Zhang D, Liu Z, Gong Z, Wang Q, Huang J, Tang C, Zou T, Yin P (2016) Structural basis of N(6)-adenosine methylation by the METTL3-METTL14 complex. Nature 534(7608):575–578. https://doi.org/10.1038/nature18298
Wang Y, Nicholes K, Shih IM (2020) The origin and pathogenesis of endometriosis. Annu Rev Pathol 15:71–95. https://doi.org/10.1146/annurev-pathmechdis-012419-032654
Wang H, Liang Z, Gou Y, Li Z, Cao Y, Jiao N, Tan J, Yu Y, Zhang Z (2022) FTO-dependent N(6)-Methyladenosine regulates the progression of endometriosis via the ATG5/PKM2 Axis. Cell Signal 98:110406. https://doi.org/10.1016/j.cellsig.2022.110406
Wanna-Udom S, Terashima M, Lyu H, Ishimura A, Takino T, Sakari M, Tsukahara T, Suzuki T (2020) The m6A methyltransferase METTL3 contributes to transforming growth factor-beta-induced epithelial-mesenchymal transition of lung cancer cells through the regulation of JUNB. Biochem Biophys Res Commun 524(1):150–155. https://doi.org/10.1016/j.bbrc.2020.01.042
Xiao F, Liu X, Guo SW (2020) Platelets and regulatory T cells may induce a type 2 immunity that is conducive to the progression and fibrogenesis of endometriosis. Front Immunol 11:610963. https://doi.org/10.3389/fimmu.2020.610963
Yang L, Chen X, Qian X, Zhang J, Wu M, Yu A (2022) Comprehensive analysis of the transcriptome-wide m6A methylome in endometrioid ovarian cancer. Front Oncol 12:844613. https://doi.org/10.3389/fonc.2022.844613
Young VJ, Ahmad SF, Duncan WC, Horne AW (2017) The role of TGF-β in the pathophysiology of peritoneal endometriosis. Hum Reprod Update 23(5):548–559. https://doi.org/10.1093/humupd/dmx016
Yu R, Li Q, Feng Z, Cai L, Xu Q (2019) m6A reader YTHDF2 regulates LPS-induced inflammatory response. Int J Mol Sci. https://doi.org/10.3390/ijms20061323
Zhai J, Li S, Sen S, Opoku-Anane J, Du Y, Chen ZJ, Giudice LC (2020) m(6)A RNA methylation regulators contribute to eutopic endometrium and myometrium dysfunction in adenomyosis. Front Genet 11:716. https://doi.org/10.3389/fgene.2020.00716
Zhang YN, Zhang YS, Yu Q, Guo ZZ, Ma JL, Yan L (2018) Higher prevalence of endometrial polyps in infertile patients with endometriosis. Gynecol Obstet Invest 83(6):558–563. https://doi.org/10.1159/000487946
Zhang Z, Luo K, Zou Z, Qiu M, Tian J, Sieh L, Shi H, Zou Y, Wang G, Morrison J, Zhu AC, Qiao M, Li Z, Stephens M, He X, He C (2020) Genetic analyses support the contribution of mRNA N(6)-methyladenosine (m(6)A) modification to human disease heritability. Nat Genet 52(9):939–949. https://doi.org/10.1038/s41588-020-0644-z
Zheng F, Du F, Qian H, Zhao J, Wang X, Yue J, Hu N, Si Y, Xu B, Yuan P (2021) Expression and clinical prognostic value of m6A RNA methylation modification in breast cancer. Biomark Res 9(1):28. https://doi.org/10.1186/s40364-021-00285-w
Zheng W, Xiang D, Wen D, Luo M, Liang X, Cao L (2023) Identification of key modules and candidate genes associated with endometriosis based on transcriptome data via bioinformatics analysis. Pathol Res Pract 244:154404. https://doi.org/10.1016/j.prp.2023.154404
Zhou WJ, Yang HL, Shao J, Mei J, Chang KK, Zhu R, Li MQ (2019) Anti-inflammatory cytokines in endometriosis. Cell Mol Life Sci 76(11):2111–2132. https://doi.org/10.1007/s00018-019-03056-x
Zhu T, Roundtree IA, Wang P, Wang X, Wang L, Sun C, Tian Y, Li J, He C, Xu Y (2014) Crystal structure of the YTH domain of YTHDF2 reveals mechanism for recognition of N6-methyladenosine. Cell Res 24(12):1493–1496. https://doi.org/10.1038/cr.2014.152
Acknowledgements
The authors thank the whole team for assistance.
Funding
This study is supported by the medical research fund of Guangdong Province (A2022266), Guangzhou municipal science and technology bureau (202201020263), Guangdong basic and applied basic research foundation (2023A1515011109) and Guangdong basic and applied basic research foundation (2022A1515110697). The funders (Weilin Zheng and Lixing Cao) provided important supports during design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
WLZ wrote the main manuscript text. WLZ and ZYF contributed to the data analysis and experimental verification. XT contributed to the pathological analysis. XFL and LXC revised the article critically. LXC and WLZ designed the study. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
The medical ethics committee at the Guangdong Second Provincial General Hospital (Approval No. 2022-KY-KZ-252-02) approved this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10528_2024_10725_MOESM1_ESM.tif
Supplementary file1 (TIF 2061 kb)—Figure S1: Workflow for the m6A regulators methylation modification patterns involved in immume microenvironment. Exploration of potential therapeutic agents in endometriosis by using data downloaded from the GEO database. Identification of the macrophage M2-related biomarkers through the CIBERSORT and WGCNA algorithms.
10528_2024_10725_MOESM2_ESM.tif
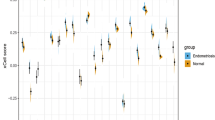
Supplementary file2 (TIF 5036 kb)—Figure S2: Expressions of m6A phenotype-related genes in ectopic and eutopic endometrium and normal endometrium. (A, B) The m6A genes based on ANOVA analysis in box plot and heat map of m6A genes for EC, EU and NM groups difference analysis, respectively. *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001. METTL3, p = 0.0095; RBM15, p = 0.0073; CBLL1, p = 0.00019; YTHDF2, p = 1.44E-06; YTHDC1, p = 0.0051; ELAVL1, p = 9.39E-07; LRPPRC, p = 0.00177; FTO, p = 0.01077.
10528_2024_10725_MOESM3_ESM.tif
Supplementary file3 (TIF 701 kb)—Figure S3: Verification of m6A expressions in EC, EU and NM. A–C shows the m6A genes based on Wilcox’s test in box plot of EC versus EU, EC versus NM, and EU versus NM, respectively. Ns denotes that statistical differences are not significant. *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001.
10528_2024_10725_MOESM4_ESM.tif
Supplementary file4 (TIF 6372 kb)—Figure S4: Spearman correlation analysis of the m6A modifification regulators in EC, EU and NM, respectively. The darker the color in the figure, the stronger the correlation, and the blank indicates that the statistical p-value is greater than or equal to 0.05, which does not reach the significant level.
10528_2024_10725_MOESM5_ESM.tif
Supplementary file5 (TIF 653 kb)—Figure S5: The forest map drawn by the multivariate logistic model established for EU and NM samples and three related genes in GSE141549. Figure S5B was showed the riskScore of EU in the training, and the Figure S4C showed the AUC of the classifier was 0.658, and the effect was poor.
10528_2024_10725_MOESM6_ESM.tif
Supplementary file6 (TIF 3275 kb)—Figure S6: The m6A genes associated with the immune microenvironment. (A) The relationship between gene METTL3 expression and B cell memory is depicted in A. The x-axis represents the expression level of gene METTL3, while the y-axis represents the proportion of immune cells in B cell memory. The figure illustrates a negative correlation between B cell memory content and METTL3 gene expression, as evidenced by a correlation coefficient of − 0.24 and a statistically significant p-value of 0.047 (p<0.05). These findings suggest a potential association between low METTL3 expression and high B cell memory expression. Similar results were observed in (B)–(L), further supporting the potential relationship between these factors. (B)METTL3 and B cell naive; (C) CBLL1 and T cells memory resting; (D) CBLL1 and T cells regulatory(Tregs); (E) CBLL1 and Mast cells resting; (F) YTHDF2 and T cells CD4 memory resting; (G) YTHDF2 and T cells regulatory (Tregs); (H) YTHDF2 and T cells tollicular helper; (I) YTHDF2 and NK cells resting; (J) FTO and T cells CD4 memory resting; (K) FTO and T cells regulatory(Tregs); (L) FTO and Dendritic cells activated.
10528_2024_10725_MOESM7_ESM.tif
Supplementary file7 (TIF 2412 kb)—Figure S7: (A) The boxplot of immune-related genes. (B) The correlation diagram of immune-related genes and m6A genes. *p < 0.05; **p < 0.01; ***p < 0.001, ns, no significant.
10528_2024_10725_MOESM8_ESM.tif
Supplementary file8 (TIF 2643 kb)—Figure S8: (A) The boxplot of HLA related genes. (B) The correlation diagram of HLA genes and m6A genes. *p < 0.05; **p < 0.01; ***p < 0.001, ns, no significant.
10528_2024_10725_MOESM9_ESM.tif
Supplementary file9 (TIF 1653 kb)—Figure S9: A(a-d) Consistency clustering of EC and EU samples. B–D are boxplots of immune-related genes, HLA-related genes and inflammatory genes in different subtypes, respectively.
10528_2024_10725_MOESM11_ESM.xlsx
Supplementary file11 (XLSX 15 kb)—Table S2: Clinical and histopathological characteristics of patients included in the study. DIE, Deep infiltrating endometriosis.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, W., Fu, Z., Tan, X. et al. Bioinformatic Analysis of m6A Regulator-Mediated RNA Methylation Modification Patterns and Immune Microenvironment Characterization in Endometriosis. Biochem Genet (2024). https://doi.org/10.1007/s10528-024-10725-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10528-024-10725-5