Abstract
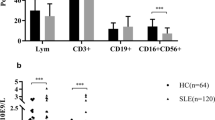
Genetic studies demonstrate that the Aiolos polymorphisms contribute to the susceptibility to autoimmune diseases. The purpose of the study was to investigate the Aiolos expression in lymphocytes and monocytes in the peripheral blood from patients with SLE and RA, and to explore the correlation between Aiolos expression in cell subsets and laboratory measurements. Peripheral blood mononuclear cells (PBMC) from 32 patients with SLE, 35 patients with RA, and 37 healthy controls were purified. Aiolos expression in PBMC subsets was examined by flow cytometry. In SLE patients, a much higher percentage of Aiolos + CD8+ T cells and Aiolos + CD14+ monocytes was found, when compared with healthy controls (p = 8.29 × 10−5 and p = 1.01 × 10−5, respectively). Furthermore, the percentage of CD4+ and CD8+ T cells, CD19+ B cells, and CD14+ monocytes expressing Aiolos in RA patients was also determined and each found higher than that in healthy controls (p = 0.009, p = 4.11 × 10−5, p = 0.001, and p = 1.11 × 10−5, respectively). The percentage of Aiolos + CD8+ T cells was weakly correlated with ESR in SLE patients and RF in RA patients (r s = 0.37, p = 0.038; r s = 0.34, p = 0.044, respectively). On the other hand, the percentage of Aiolos + CD14+ monocytes was significantly correlated with multiple laboratory measurements, including ESR, creatinine, CRP, LDH, proteinuria, albumin, and ACCPA in patients (r s = 0.62, p < 0.001; r s = 0.65, p < 0.001; r s = 0.44, p = 0.010; r s = 0.42, p = 0.022; r s = 0.52, p = 0.013; r s = 0.34, p = 0.048, respectively). To our knowledge, it is the first study to demonstrate overexpression of Aiolos in PBMC subsets in SLE and RA patients. The results indicate that overexpression of Aiolos may contribute to pathogenesis of SLE and RA.

Similar content being viewed by others
References
Antica M, Cicin-Sain L, Kapitanovic S, Matulic M, Dzebro S, Dominis M (2008) Aberrant Ikaros, Aiolos, and Helios expression in Hodgkin and non-Hodgkin lymphoma. Blood 111(6):3296–3297
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS et al (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31(3):315–324
Billot K, Parizot C, Arrouss I, Mazier D, Debre P, Rogner UC et al (2010) Differential aiolos expression in human hematopoietic subpopulations. Leuk Res 34(3):289–293
Billot K, Soeur J, Chereau F, Arrouss I, Merle-Beral H, Huang ME et al (2011) Deregulation of Aiolos expression in chronic lymphocytic leukemia is associated with epigenetic modifications. Blood 117(6):1917–1927
Blanco P, Pitard V, Viallard JF, Taupin JL, Pellegrin JL, Moreau JF (2005) Increase in activated CD8+ T lymphocytes expressing perforin and granzyme B correlates with disease activity in patients with systemic lupus erythematosus. Arthritis Rheum 52(1):201–211
Bombardier C, Gladman DD, Urowitz MB, Caron D, Chang CH (1992) Derivation of the SLEDAI. A disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum 35(6):630–640
Cai X, Qiao Y, Diao C, Xu X, Chen Y, Du S et al (2014) Association between polymorphisms of the IKZF3 gene and systemic lupus erythematosus in a Chinese Han population. PLoS One 9(10):e108661
Carvalheiro H, da Silva JA, Souto-Carneiro MM (2013) Potential roles for CD8(+) T cells in rheumatoid arthritis. Autoimmun Rev 12(3):401–409
Davignon JL, Hayder M, Baron M, Boyer JF, Constantin A, Apparailly F et al (2013) Targeting monocytes/macrophages in the treatment of rheumatoid arthritis. Rheumatology (Oxford) 52(4):590–598
Dhaouadi T, Sfar I, Haouami Y, Abdelmoula L, Turki S, Hassine LB et al (2013) Polymorphisms of Toll-like receptor-4 and CD14 in systemic lupus erythematosus and rheumatoid arthritis. Biomark Res 1(1):20
Duhamel M, Arrouss I, Merle-Beral H, Rebollo A (2008) The Aiolos transcription factor is up-regulated in chronic lymphocytic leukemia. Blood 111(6):3225–3228
Eguchi K (2001) Apoptosis in autoimmune diseases. Intern Med 40(4):275–284
Eisenberg R, Albert D (2006) B-cell targeted therapies in rheumatoid arthritis and systemic lupus erythematosus. Nat Clin Pract Rheumatol 2(1):20–27
Gizinski AM, Fox DA (2014) T cell subsets and their role in the pathogenesis of rheumatic disease. Curr Opin Rheumatol 26(2):204–210
Kinne RW, Emmrich F, Freesmeyer M (2010) Clinical impact of radiolabeled anti-CD4 antibodies in the diagnosis of rheumatoid arthritis. Q J Nucl Med Mol Imaging 54(6):629–638
Koipally J, Renold A, Kim J, Georgopoulos K (1999) Repression by Ikaros and Aiolos is mediated through histone deacetylase complexes. EMBO J 18(11):3090–3100
Kurreeman FA, Stahl EA, Okada Y, Liao K, Diogo D, Raychaudhuri S et al (2012) Use of a multiethnic approach to identify rheumatoid- arthritis-susceptibility loci, 1p36 and 17q12. Am J Hum Genet 90(3):524–532
Lessard CJ, Adrianto I, Ice JA, Wiley GB, Kelly JA, Glenn SB et al (2012) Identification of IRF8, TMEM39A, and IKZF3-ZPBP2 as susceptibility loci for systemic lupus erythematosus in a large-scale multiracial replication study. Am J Hum Genet 90(4):648–660
Li Y, Lee PY, Reeves WH (2010) Monocyte and macrophage abnormalities in systemic lupus erythematosus. Arch Immunol Ther Exp (Warsz) 58(5):355–364
Liote F, Boval-Boizard B, Weill D, Kuntz D, Wautier JL (1996) Blood monocyte activation in rheumatoid arthritis: increased monocyte adhesiveness, integrin expression, and cytokine release. Clin Exp Immunol 106(1):13–19
Morgan B, Sun L, Avitahl N, Andrikopoulos K, Ikeda T, Gonzales E et al (1997) Aiolos, a lymphoid restricted transcription factor that interacts with Ikaros to regulate lymphocyte differentiation. EMBO J 16(8):2004–2013
Nakase K, Ishimaru F, Avitahl N, Dansako H, Matsuo K, Fujii K et al (2000) Dominant negative isoform of the Ikaros gene in patients with adult B-cell acute lymphoblastic leukemia. Cancer Res 60(15):4062–4065
Narvi E, Nera KP, Terho P, Mustonen L, Granberg J, Lassila O (2007) Aiolos controls gene conversion and cell death in DT40 B cells. Scand J Immunol 65(6):503–513
Nuckel H, Frey UH, Sellmann L, Collins CH, Duhrsen U, Siffert W (2009) The IKZF3 (Aiolos) transcription factor is highly upregulated and inversely correlated with clinical progression in chronic lymphocytic leukaemia. Br J Haematol 144(2):268–270
Romero F, Martinez AC, Camonis J, Rebollo A (1999) Aiolos transcription factor controls cell death in T cells by regulating Bcl-2 expression and its cellular localization. EMBO J 18(12):3419–3430
Rus V, Nguyen V, Puliaev R, Puliaeva I, Zernetkina V, Luzina I et al (2007) T cell TRAIL promotes murine lupus by sustaining effector CD4 Th cell numbers and by inhibiting CD8 CTL activity. J Immunol 178(6):3962–3972
Schmitt C, Tonnelle C, Dalloul A, Chabannon C, Debre P, Rebollo A (2002) Aiolos and Ikaros: regulators of lymphocyte development, homeostasis and lymphoproliferation. Apoptosis 7(3):277–284
Smiljanovic B, Grun JR, Biesen R, Schulte-Wrede U, Baumgrass R, Stuhlmuller B et al (2012) The multifaceted balance of TNF-alpha and type I/II interferon responses in SLE and RA: how monocytes manage the impact of cytokines. J Mol Med (Berl) 90(11):1295–1309
Sun J, Matthias G, Mihatsch MJ, Georgopoulos K, Matthias P (2003) Lack of the transcriptional coactivator OBF-1 prevents the development of systemic lupus erythematosus-like phenotypes in Aiolos mutant mice. J Immunol 170(4):1699–1706
Tan EM, Cohen AS, Fries JF, Masi AT, McShane DJ, Rothfield NF et al (1982) The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 25(11):1271–1277
Tarr T, Derfalvi B, Gyori N, Szanto A, Siminszky Z, Malik A et al (2015) Similarities and differences between pediatric and adult patients with systemic lupus erythematosus. Lupus 24(8):796–803
Townsend MJ (2014) Molecular and cellular heterogeneity in the Rheumatoid Arthritis synovium: clinical correlates of synovitis. Best Pract Res Clin Rheumatol 28(4):539–549
Wang JH, Avitahl N, Cariappa A, Friedrich C, Ikeda T, Renold A et al (1998) Aiolos regulates B cell activation and maturation to effector state. Immunity 9(4):543–553
Yamagiwa T, Fukunishi S, Tachibana T, Okamura H, Yoshiya S, Kashiwamura S (2012) Abrogation of Treg function deteriorates rheumatoid arthritis. Mod Rheumatol 22(1):80–88
Zhuang Y, Li D, Fu J, Shi Q, Lu Y, Ju X (2014) Overexpression of AIOLOS inhibits cell proliferation and suppresses apoptosis in Nalm-6 cells. Oncol Rep 31(3):1183–1190
Zhuang Y, Lu Y, Li D, Sun N, Ju X (2015) Upregulation of AIOLOS induces apoptosis and enhances etoposide chemosensitivity in Jurkat leukemia cells. Oncol Rep 33(3):1319–1325
Acknowledgments
We especially appreciate all the SLE and RA patients making this study accomplished. This study was funded by the National Natural Science Foundation of China (No. 81401330).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Cai, X., Liu, X., Du, S. et al. Overexpression of Aiolos in Peripheral Blood Mononuclear Cell Subsets from Patients with Systemic Lupus Erythematosus and Rheumatoid Arthritis. Biochem Genet 54, 73–82 (2016). https://doi.org/10.1007/s10528-015-9702-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10528-015-9702-0