Abstract
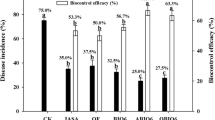
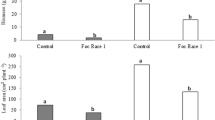
More effective ways of applying biocontrol products should be developed based both on the characteristics of the biocontrol agents and the normal practices of the agricultural producer. A new system was developed to improve the biocontrol efficacy of Fusarium wilt for watermelon production, and this system was tested in pot and field experiments. Biocontrol was achieved by applying a novel bioorganic fertilizer product (BIO) to Fusarium-infested soil. The best biocontrol was obtained by application of a bioorganic fertilizer, BIO, into soil during the nursery phase of watermelon seedling followed by a second application to Fusarium-infested soil when watermelon seedlings were transplanted. In comparison with the controls, the incidence of the disease was reduced by 60–100% in the pot experiment and by 59–73% in the field experiment when the BIO was applied during the nursery stage. After application of BIO during the nursery stage, the number of colony-forming units of Fusarium oxysporum in rhizospheric soil was significantly (P < 0.05) inhibited compared to the controls. An in vitro experiment showed that the antagonist Paenibacillus polymyxa in the BIO could effectively colonize the rhizosphere of watermelon and proliferate along the extending plant roots. This inhibited growth of Fusarium oxysporum in the rhizosphere of watermelon and protected the watermelon roots from attack by the pathogens. The method used for biocontrol Fusarium wilt disease in watermelon should be a useful strategy to improve field efficacy of other biocontrol agents.






Similar content being viewed by others
References
Alabouvette C, Lemanceau P, Steinberg C (1993) Recent advances in the biological control of fusarium wilts. Pestic Sci 37:365–373
Alabouvette C, Olivain C, Steinberg C (2006) Biological control of plant diseases: the European situation. Eur J Plant Pathol 114:329–341
Booth C (1971) The genus Fusarium. The Eastern Press, London, pp 147–149
Borrero C, Trillas MI, Ordovás J, Tello J, Avilés M (2004) Predictive factors for the suppression of Fusarium wilt of tomato in plant growth media. Phytopathology 94:1094–1101
Brunner K, Zeilinger S, Ciliento R, Woo SL, Lorito M, Kubicek CP, Mach RL (2005) Improvement of the fungal biocontrol agent Trichoderma atroviride to enhance both antagonism and induction of plant systemic disease resistance. Appl Environ Microb 71:3959–3965
Cook RJ (1993) Making greater use of introduced microorganisms for biological control of plant pathogens. Annu Rev Phytopathol 31:53–80
Cotxarrera L, Trillas-Gay MI, Steinberg C, Alabouvette C (2002) Use of sewage sludge compost and Trichoderma asperellum isolates to suppress Fusarium wilt of tomato. Soil Biol Biochem 34:467–476
Dijksterhuis J, Sanders M, Gorris LGM, Smid EJ (1999) Antibiosis plays a role in the context of direct interaction during antagonism of Paenibacillus polymixa towards Fusarium oxysporum. J Appl Microbiol 86:13–21
Duffy BK, Weller DM (1995) Use of Gaeumannomyces graminis var graminis alone and in combination with fluorescent Pseudomonas spp. to suppress take-all of wheat. Plant Dis 79:907–911
Fuchs JG, Moënne Loccoz Y, Défago G (1999) Ability of nonpathogenic Fusarium oxysporum Fo47 to protect tomato against Fusarium wilt. Biol Control 14:105–110
Georgakopoulos DG, Fiddaman P, Leifert C, Malathrakis NE (2002) Biological control of cucumber and sugar beet damping-off caused by Pythium ultimum with bacterial and fungal antagonists. J Appl Microbiol 92:1078–1086
Haggag WM, Timmusk S (2008) Colonization of peanut roots by biofilm-forming Paenibacillus polymyxa initiates biocontrol against crown rot disease. J Appl Microbiol 104:961–969
Hervás A, Landa B, Jiménez-Díaz RM (1997) Influence of chickpea genotype and Bacillus sp. on protection from Fusarium wilt by seed treatment with nonpathogenic Fusarium oxysporum. Eur J Plant Pathol 103:631–642
Hervás A, Landa B, Datnoff LE (1998) Effects of commercial and indigenous microorganisms on Fusarium wilt development in chickpea. Biol Control 13:166–176
Hoitink HAJ, Boehm MJ (1999) Biocontrol within the context of soil microbial communities: a substrate-dependent phenomenon. Annu Rev Phytopathol 37:427–446
Hoitink HAJ, Fahy PC (1986) Basis for the control of soil-borne plant pathogens with composts. Annu Rev Phytopathol 24:93–114
Hoitink HAJ, Schmitthener AF, Herr LJ (1975) Composted bark for control of root rot in ornamentals. Ohio Rep 60:25–26
Kamilova F, Validov S, Azarova T, Mulders I, Lugtenberg B (2005) Enrichment for enhanced competitive plant root tip colonizers selects for a new class of biocontrol bacteria. Environ Microbiol 7:1809–1817
Komada H (1975) Development of a selective medium for quantitative isolation of Fusarium oxysporum from natural soil. Rev Plant Protect Res 8:114–125
Leeman M, Den Ouden EM, Van Pelt JA, Cornelissen C, Matamala-Garros A, Bakker PAHM, Schippers B (1996) Suppression of Fusarium wilt of radish by co-inoculation of fluorescent Pseudomonas spp. and root-colonizing fungi. Eur J Plant Pathol 102:21–31
Litterick A, Harrier L, Wallace P, Watson C, Wood M (2004) The role of uncomposted materials, composts, manures and compost extracts in reducing pest and disease incidence and severity in sustainable temperate agricultural and horticultural crop production—a review. Crit Rev Plant Sci 23:453–479
Lumsden RD, Lewis JA, Millner PD (1983) Effect of composted sewage sludge on several soilborne pathogens and diseases. Phytopathology 73:1543–1548
Martyn RD (1996) Fusarium wilt of watermelon. In: Zitter TA, Hopkins DL, Thomas CA (eds) Compendium of cucurbit diseases. The American Phytopathology Society, St. Paul, pp 13–14
Minuto A, Spadaro D, Garibaldi A, Gullino LM (2006) Control of soilborne pathogens of tomato using a commercial formulation of Streptomyces griseoviridis and solarization. Crop Prot 25:468–475
Nemec S, Datnofft LE, Strandberg J (1996) Efficacy of biocontrol agents in planting mixes to colonize plant roots and control root diseases of vegetables and citrus. Crop Prot 15:735–742
Raza W, Yang W, Shen Q-R (2008) Paenibacillus polymyxa: antibiotics hydrolytic enzymes and hazard assessment. J Plant Pathol 90:419–430
Ryu C-M, Kim J, Choi O, Kim SH, Park CS (2006) Improvement of biological control capacity of Paenibacillus polymyxa E681 by seed pelleting on sesame. Biol Control 39:282–289
Sabuquillo P, De Cal A, Melgarejo P (2006) Biocontrol of tomato wilt by Penicillium oxalicum formulations in different crop. Biol Control 37:256–265
Sivan A, Chet I (1992) Microbial control of plant disease. In: Mitchell R (ed) Environmental microbiology. Wiley, New York, pp 335–354
Timmusk S, Grantcharova N, Wagner HGE (2005) Paenibacillus polymyxa invades plant roots and forms biofilm. Appl Environ Microb 71:7292–7300
Trillas MI, Casanova E, Corxarrera L, Ordovas J, Borrero C, Aviles M (2006) Composts from agricultural waste and the Trichoderma asperellum strain T-34 suppress Rhizoctonia solani in cucumber seedlings. Biol Control 39:32–38
Wu HS, Yang XM, Fan JQ, Miao WG, Ling N, Shen QR (2008) Suppression of Fusarium wilt of watermelon by a bio-organic fertilizer containing combinations of antagonistic microorganisms. BioControl 54:287–295
Zhang SS, Raza W, Yang XM, Hu J, Huang QW, Xu YC, Liu XH, Ran W, Shen QR (2008) Control of Fusarium wilt disease of cucumber plants with the application of a bio-organic fertilizer. Biol Fertil Soils 44:1073–1080
Acknowledgment
This research was financially supported by the Nature Science Foundation of China (40871126) and the Science and Technology Ministry of China (2007CB109304).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Reijo Karjalainen.
Rights and permissions
About this article
Cite this article
Ling, N., Xue, C., Huang, Q. et al. Development of a mode of application of bioorganic fertilizer for improving the biocontrol efficacy to Fusarium wilt. BioControl 55, 673–683 (2010). https://doi.org/10.1007/s10526-010-9290-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10526-010-9290-1