Abstract
Heritability estimates of general intelligence in adulthood generally range from 75 to 85%, with all heritability due to additive genetic influences, while genetic dominance and shared environmental factors are absent, or too small to be detected. These estimates are derived from studies based on the classical twin design and are based on the assumption of random mating. Yet, considerable positive assortative mating has been reported for general intelligence. Unmodeled assortative mating may lead to biased estimates of the relative magnitude of genetic and environmental factors. To investigate the effects of assortative mating on the estimates of the variance components of intelligence, we employed an extended twin-family design. Psychometric IQ data were available for adult monozygotic and dizygotic twins, their siblings, the partners of the twins and siblings, and either the parents or the adult offspring of the twins and siblings (N = 1314). Two underlying processes of assortment were considered: phenotypic assortment and social homogamy. The phenotypic assortment model was slightly preferred over the social homogamy model, suggesting that assortment for intelligence is mostly due to a selection of mates on similarity in intelligence. Under the preferred phenotypic assortment model, the variance of intelligence in adulthood was not only due to non-shared environmental (18%) and additive genetic factors (44%) but also to non-additive genetic factors (27%) and phenotypic assortment (11%).This non-additive nature of genetic influences on intelligence needs to be accommodated in future GWAS studies for intelligence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Considerable evidence from classical twin studies shows that individual differences in general intelligence in adults are largely explained by additive genetic factors. Heritability estimates range from 75 to 85% (Plomin 1999; Ando et al. 2001; Luciano et al. 2001; Bouchard and McGue 1981; Posthuma et al. 2001a). These estimates are based on the classical twin design (CTD) in which the phenotypic resemblance of monozygotic (MZ) and dizygotic (DZ) twins is compared (Plomin et al. 2001a). MZ twin correlations of ~.80 and DZ twin correlations of ~.40 are typically reported and suggest absence of both shared environmental influences and genetic dominance. The CTD, however, relies on the assumption that mating of spouse pairs is random, and has to assume that either shared environmental influences or non-additive genetic factors (i.e., dominance and epistasis) are absent as these two sources of variation cannot be estimated simultaneously in the CTD.
Strong assortative mating for intelligence has however been reported, with spousal correlations for intelligence ranging from .20 to .50 (Reynolds et al. 2000; van Leeuwen et al. 2008; Mascie-Taylor 1989; Jencks et al. 1972; Loehlin 1978). Assortative mating for a heritable trait leads to a non-random distribution of the genetic variants important for that trait as spouses will be more similar genetically than expected by chance. Unmodeled assortative mating will consequently increase the correlation of DZ twin pairs, while the MZ twin correlation remains unaffected. As shared environmental factors are expected if the DZ twin correlation is more than half the MZ twin correlation, whereas genetic dominance is expected if the DZ twin correlation is less than half the MZ twin correlation, increased DZ twin correlations may deflate estimates of genetic dominance due to unmodeled assortative mating in a CTD (Keller et al. 2009b).
A second mechanism that tends to inflate DZ twin correlations is cultural transmission (Fulker 1982). Cultural transmission (CT) refers to the transmission of environmental factors that are related to the trait (e.g., general intelligence) from the parental generation to the offspring generation, and is shared between siblings. CT—when unmodeled is thus part of the shared environmental ‘C’ component in the offspring generation. Consequently, unmodeled CT results in increased DZ twin correlations relative to the MZ twin correlations, thereby inflating estimates of shared environmental effects, which may go undetected in the CTD, in the presence of genetic dominance.
To disentangle all these different sources of variation, we set out to investigate the influences of assortative mating, CT, additive genetic factors, genetic dominance, and shared- and non-shared environmental influences on general intelligence in an adult population using an extended twin-family design. Two mechanisms underlying assortative mating were considered: social homogamy and phenotypic assortment. In the present study, social homogamy is defined as assortment that takes place within groups that are differentiated environmentally, and positive phenotypic assortment is defined as assortment based on similar phenotypes, i.e., similar level of intelligence (Falconer and Mackay 1989). A third form of assortative mating, i.e., social interaction, in which mates resemble each other more as a function of the time they have been together, was not considered in the present study because the data were not sufficient to model this type of assortment.
These different mechanisms of mate selection result in different expectations for familial resemblance (Fisher 1918; Rice et al. 1978; Heath and Eaves 1985; Falconer and Mackay 1989). Basically, under social homogamy, resemblance between relatives is a function of shared environment (social resemblance), whereas, under phenotypic assortment, trait resemblance is a function of genetic resemblance between relatives, such that phenotypic correlations between individuals decrease with increasing genetic distance, as well as environmental resemblance.
To determine whether the effects of assortative mating, CT, genetic dominance, additive genetic factors, and shared and non-shared environmental factors are important for general intelligence, and for the verbal and performance intelligence sub dimensions, in adulthood, we collected psychometric IQ data in 1,314 individuals from 317 families. Families consisted of twins and their non-twin siblings, the spouses of these twins and siblings, and either the parents or the children of the twins and siblings.
Method
Sample
This study was part of a large ongoing project on the genetics of cognition. In a first wave of data collection (Posthuma et al. 2001a, b), data on psychometric IQ from twins and their non-twin siblings were collected between 1997 and 2001. To allow the modeling of complex processes such as assortative mating and CT, the data set was extended with psychometric IQ data from relatives from multiple generations (parents, children, and spouses of the twins and their non-twin siblings) between 2007 and 2009 in a second wave of data collection. To recruit participants for the second wave of data collection, non-registered family members of the twins and their non-twin siblings were approached after permission from their registered family members. All participants were registered in the Netherlands Twin Register (NTR) (Boomsma et al. 2006). In the present study, data were available for 1,314 participants (317 families, 45.7% men). On average 4.1 subjects per family participated.
Zygosity of same-sex twins was based on DNA polymorphisms (114 pairs, 83%) or, if information on DNA markers was not available, on questions about physical similarity and confusion of the twins by family members and strangers. Agreement between zygosity diagnoses from DNA and survey was 97% (Willemsen et al. 2005). All five zygosity groups were well represented: MZ males (MZM: 20.6%), MZ females (MZF: 25.4%), DZ males (DZM: 12.4%), DZ females (DZF: 22.4%) and DZ opposite sex (DOS: 19.2%). The biological relatedness of all family members was determined based on questions about family structure. Average age of the participants was 41.11 years (SD = 15.06; range: 15.71–79.87).
Mean ages (standard deviations) of the parent, twin and offspring generations were 63.81 (5.17), 39.81 (13.55), and 28.86 (6.89), respectively. Mean age differed significantly between parent- and twin generations (t = 21.48, p < .001), between parent- and offspring generations (t = 50.89, p < .001), and between twin and offspring generations (t = 10.27, p < .001). The twin generation consisted of two birth cohorts (for an extensive description see (Posthuma et al. 2001a). Table 1 shows frequencies of all relatives that are included in the sample, grouped by zygosity of the twins. Spouses and children of twins were more willing to participate than spouses and children of siblings.
Tasks and instruments
General intelligence, operationalized as scores on a psychometric intelligence test (Full Scale IQ, FSIQ), was assessed with the Dutch version of the WAIS-IIIR (Wechsler 1997). Participants assessed in the first wave of data collection (770 participants: twins and siblings) completed eleven subtests of the WAIS-IIIR: Block design, Letter-number sequencing, Information, Matrix reasoning, Similarities, Picture completion, Arithmetic, Vocabulary, Digit symbol-coding, Digit symbol pairing and Digit symbol-free recall. Participants assessed in the second wave of data collection (544 participants: twins, siblings, parents, offspring of twins and siblings and spouses of twins and siblings) completed seven subtests of the WAIS-IIIR: Block design, Letter-number sequencing, Information, Matrix reasoning, Arithmetic, Vocabulary and Digit symbol-coding. Correlation between FSIQ assessed with eleven subtests, and FSIQ assessed with seven subtests, was very high (Pearson’s r = .97, N = 770, p < .001). 59 participants participated in both the first and the second wave of data collection, test–retest reliability over 7–10 years was substantial (Pearson’s r = .85, N = 59, p < .001, based on seven subtests). For those subjects for whom data from two waves of data collection were available, data from the first wave were used in the analyses. The present sample is representative of the Dutch population with respect to educational level (Posthuma et al. 2001a). Because effects of age and sex on FSIQ scores were still present after WAIS-IIIR standardization procedures, and because the present sample size exceeded the WAIS-IIIR standardization sample, residual effects of sex and age were partialled out to avoid spuriously increased similarities in MZ and same-sex DZ twin pairs (McGue and Bouchard 1984). In total, sex and age corrected FSIQ scores were available for 1,314 participants (see Table 1).
To eliminate possible discrepancies between FSIQ data collected in the first and in the second wave of data collection, Z-transformed scores were used in the analyses. FSIQ scores were Z-transformed in both groups separately (wave 1 and wave 2) such that the scores had equal means and variances in both waves. For convenience, these z-scores were transformed such that the overall mean was 100, and the SD was 15, as is standard in IQ research.
Power
Power simulations have shown that large sample sizes are required to distinguish contributions of phenotypic assortment and social homogamy to mate selection (Heath and Eaves 1985). According to Heath and Eaves (1985), data on DZ and sibling pairs and their spouses are more informative to resolve the nature of the process of mate selection than data on MZ pairs and their spouses because the high phenotypic correlation between MZ twin pairs complicates resolving phenotypic assortment and social homogamy. Both MZ and DZ twin pairs are, however, required to disentangle genetic and environmental influences on individual differences in general intelligence. A mixed homogamy model, in which both social homogamy and positive phenotypic assortment act simultaneously, requires a larger sample size than the one currently available (Heath and Eaves 1985). The size and composition of the present sample should, however, be sufficient to allow distinction between pure social homogamy and pure positive phenotypic assortment (Heath and Eaves 1985). As the sample size was also too small to explicitly examine possible sex and age effects on the genetic and environmental parameters, age- and sex corrected FSIQ scores were used in all analyses.
Analyses
This study consisted of three parts. First, a saturated model was fitted to the data to estimate model free correlations between pairs of different genetic and social relatedness, and to test whether correlations between DZ twins differed from correlations between regular siblings. The saturated model is exclusively used to estimate correlations between family members, and does not yet include any assumptions with respect to mating behavior. A total number of 102 different correlations between relatives can be estimated when all correlations are subdivided by sex of the twin pairs. As our sample size was relatively small, the precision of such specific correlations was low, especially for more distant relatives (Keller et al. 2009a). It was therefore decided to not further investigate possible sex and age effects on the variances and covariances. Note that previous analyses in a partly overlapping sample showed no significant sex effects on variances and covariances of sub dimensions of general intelligence, while effects of age were significant with direction depending on the dimension; see (Posthuma et al. 2001b), these age effects, however, disappeared when the dimensions of intelligence were combined into one general measure of intelligence (i.e., Full Scale IQ). Significance of the difference between the DZ twin correlation and the correlation between regular siblings was tested by constraining these correlations to be equal (e.g., DZ twin correlation = sibling correlation, nieces/nephews through DZ twins = nieces/nephews through siblings, etcetera). A significant worsening of the model fit is indicative for a special twin environment. A model without special twin environment and without sex and age effects on the variances and covariances, would leave us with 15 different relations: twin–twin MZ, twin–twin DZ/sibling, parent-offspring, cousins avuncular through MZ, cousins avuncular through DZ/sibling, niece/nephews through MZ, niece/nephews through DZ/sibling, spouse pairs, sister/brother in law through MZ, sister/brother in law through DZ/sibling, spouse–spouse through MZ, spouse–spouse through DZ/sibling, parent–offspring in law, aunt/uncle cousin in law through MZ, and aunt/uncle cousin in law through DZ/sibling. Note that the grandparent-grandchild correlation was not estimated since none of the families comprised three generations.
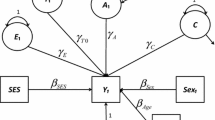
Second, within a genetic model, the two competing assortment models (i.e., social homogamy versus phenotypic assortment) were fitted to the data, to investigate whether social homogamy or positive phenotypic assortment was the most likely underlying process of assortative mating for general intelligence. The fit of both models was compared to the fit of the saturated model. Under the social homogamy model, assortative mating is due to a common environment that renders individuals with common social backgrounds more alike. Mate selection is purely based on environmental similarities. Consequently, correlations are expected to be similar for any combination of sibling-spouse pairsFootnote 1 (r spouses = r co-twin–spouse = r spouse1–spouse2). In contrast, under the positive phenotypic assortment model mate selection is purely based on the phenotype of the spouses (i.e., similar general intelligence). Consequently, for a heritable trait like intelligence, correlations between sibling and spouse pairs are expected to decline with the distance of the genetic relationship (r spouse > r co-twin–spouse > r spouse1–spouse2), and cross-sibling-spouse correlations are expected to be higher for MZ twins compared to DZ twins, depending on the extent to which the phenotype under study reflects the genotype. Preference of the assortment model was based on maximum likelihood estimation. The preferred assortment process was modeled in subsequent analyses of the relative contribution of genetic and environmental factors. Both assortment models are depicted in Fig. 1.
Full assortment models for a DZ twin pair with parents, spouses, and offspring: social homogamy (upper panel) and positive phenotypic assortment (lower panel). A additive genetic effects, D genetic dominance, E non-shared environmental effects, C shared environmental effects, f cultural transmission path, w gene-environment correlation, q variance additive genetic effects, x variance shared environmental effects, ρ z assortative mating co-path, P parent, T DZ twin, Sp spouse, O offspring. Please note that additional siblings (and their spouses and offspring) are not included in the figure for reasons of convenience
Third, individual differences in general intelligence were modeled as a function of genetic and environmental effects, taking into account the preferred underlying process of assortative mating (ASM; i.e., phenotypic assortment or social homogamy) as well as CT, additive genetic factors (A), genetic dominance (D), and shared non-parental (C) and non-shared (E) environmental factors (Eaves et al. 1999). ‘A’ represents additive effects of alleles summed over all loci. ‘D’ represents the extent to which the effects of alleles (at a locus or across loci) are not additives but interact with each other (i.e., genetic dominance, epistasis). Genetic dominance and effects of epistasis cannot be distinguished in this model. ‘ASM’ represents genetic influences due to assortative mating. ‘C’ represents non-parental common environmental influences that render offspring of the same family more alike. CT represents shared environmental factors due to CT. Parents transmit not only their genetic material, but also their environment to their children. CT refers to the transmission of environmental factors that are related to the trait (e.g., general intelligence) from the parental phenotype to their offspring’s environment (Maes et al. 2009; Keller et al. 2009b). Presence of both CT and genetic transmission will result in a correlation between A and CT (i.e., rGE). ‘E’ represents all environmental influences that result in differences between members of a family. E also includes measurement error.
Because of the limited size of the sample, we assumed that assortative mating, genetic inheritance, shared environmental influences, and CT remain constant from generation to generation. This implies that phenotypic variances and correlations between relatives are equal over generations as a state of equilibrium has been reached (Falconer and Mackay 1989).
Analyses were carried out using the raw data option in Mx (Neale 1994; Maes et al. 2009). All analyses that were performed for FSIQ were subsequently repeated for the verbal intelligence and performance intelligence sub dimensions of the WAIS-IIIR.
Results
Within the saturated model, correlations via DZ twin pairs and regular sibling pairs could be constrained to equal without a significant worsening of the model fit (Table 2, model S-1 versus S-2: χ2(6) = 6.08, ns), implying that there was no special twin environment that renders members of a twin pair more similar for general intelligence than regular siblings. This is in line with Posthuma et al. (2001a). Consequently, special twin environmental effects were excluded from the subsequent genetic models.
Figure 2 shows observed phenotypic correlations (and 95% confidence intervals) and expected genetic correlations between relatives grouped by degree of additive genetic similarity (A) and genetic dominance (D) similarity. Note that the degree of additive genetic similarity increases over time within a population undergoing phenotypic assortment for all pairs of relatives except MZ twin pairs. Similarly, dominance genetic similarity increases over time as a result of phenotypic assortment for cousins avuncular through MZ/DZ/sibling, niece/nephews through MZ/DZ/sibling, sister/brother in law through MZ/DZ/sibling, spouse–spouse through MZ/DZ/sibling and aunt/uncle cousin in law through MZ/DZ/sibling within a population undergoing assortative mating, while under random assortment there would be no similarity in D for these pairs of relatives (Fisher 1918; Nagylaki 1978; Lynch and Walsh 1998). Under social homogamy, spousal correlations are increased but do not differ as a function of genetic relatedness (i.e., correlations between direct spouse pairs are expected to be the same as correlations between spouses in law). Social homogamy does not affect the genetic relatedness of relatives. Please see the Appendix for coefficients of relatedness between relatives for A and D for a population undergoing phenotypic assortment. Within a population undergoing social homogamy, additive genetic similarity and dominance genetic similarity remain unaltered over time.
Mean correlation (95% CI) of general intelligence between relatives grouped by degree of theoretical additive genetic similarity and dominance genetic similarity. Observed observed correlation, PA expected genetic similarity (A + D) under phenotypic assortment, No PA, expected genetic similarity (A + D) assuming no phenotypic assortment; 95% CI = 95% confidence interval; correlations are constrained to be equal across twins and regular siblings and across sex; MZ twin–twin MZ, DZ twin–twin DZ/sibling, PO parent-offspring, AVMZ cousins avuncular through MZ, AVDZ cousins avuncular through DZ/sibling; COMZ niece/nephews through MZ, CODZ niece/nephews through DZ/sibling, SP spouse pairs; SMZ sister/brother in law through MZ, SDZ sister/brother in law through DZ/sibling, SMZS spouse–spouse through MZ, SDZS spouse–spouse through DZ/sibling, SAVMZ aunt/uncle cousin in law through MZ, SAVDZ aunt/uncle cousin in law through DZ/sibling, POS parent-offspring in law. Please note that the degree of additive genetic similarity increases within a population undergoing phenotypic assortment for all pairs of relatives except MZ twin pairs. Similar, dominance genetic similarity is induced by phenotypic assortment for AVMZ, AVDZ, COMZ, CODZ, SMZ, SDZ, SMZS, SDZS, SAVMZ, and SAVDZ within a population undergoing phenotypic assortment, where under random assortment there would be none (Fisher 1918; Nagylaki 1978; Lynch and Walsh 1998). For the expected correlations we assumed \( \hat{h}^{2} \) = .44, \( \hat{d}^{2} \) = .27, as estimated under the reduced model and ρ z = .37 (i.e., the observed spousal correlation); Please see the Appendix for coefficients for σ 2 A and σ 2 D . Under social homogamy, spousal correlations are increased but do not differ as a function of genetic relatedness (i.e., correlations between direct spouse pairs are expected to be the same as correlations between spouses in law). Social homogamy does not affect the genetic relatedness of relatives
Figure 2 shows a higher correlation for family members who share 100% of their genetic material (i.e., MZ twin pairs, r = .82), compared to family members who share on average 50% of A and 25% of D (i.e., DZ twin pairs and regular siblings; r = .37). The figure illustrates that this steep decrease in phenotypic resemblance between relatives did not persist for relatives with decreasing genetic relatedness and as one would expect under random mating. Correlations were also substantial for spouse pairs who do not share genetic material under the assumption of random mating; these spouse correlations generally decreased with increasing distance. This pattern of correlations suggests strong influence of genetic factors as well as a considerable contribution of assortative mating.
Table 2 shows the model fitting results of the social homogamy (SH) model (model SH-1: χ2(6) = 23.12, p < .01), and the phenotypic assortment (PA) model (model PA-1: χ2(6) = 21.58, p < .01) compared to the constrained saturated model (i.e., model S-2, in which correlations for all relations through DZ and sibling pairs were constrained to be equal). Both assortment models caused a significant decrease in model fit, but the SH model fitted the data relatively worse than the PA model (according to χ2 difference test, Akaike’s information criterion (AIC), Bayesian information criterion (BIC), and Deviance information criterion (DIC)).
The worsening of the model fit for both the SH and the PA model was largely attributable to observed spousal correlations being higher than spousal correlations expected under both assortment models. Discrepancies between the saturated model and the genetic model were largest under the SH model. Moreover, under the SH model, correlations were expected to be similar for any combination of sibling-spouse pairs, while in the present data, spousal correlations generally decreased with increasing distance between relatives. In the SH model, such a decrease in observed spousal correlations can only be accommodated by increasing the D component (which was indeed considerable in the SH model solution), which resulted in overall misfit. Based on test statistics (χ2 difference test, AIC, BIC, and DIC) and biological interpretation of the parameter estimates in the SH model (i.e., substantial contribution of D and very small A, a scenario that is biologically unlikely (Falconer and Mackay 1989)), the PA model seems to describe the observed data pattern better. However, as the difference in misfit (compared to the saturated model) between SH and PA was small (23.12 vs. 21.58), we chose to further examine the relative contributions of genetic and environmental factors under both assortment models.
Social homogamy model
Within the full SH model (i.e., model SH-1), the effect of D (57%) was substantial, effects of CT (23%), and E (18%) were modest, the effect of C (2%) was small, and the effects of A (0%) and ASM (0%) were estimated at zero. The model induced correlation (rGE) between A and CT was estimated at zero (see Fig. 3). Model fitting results are presented in Table 2. Eliminating non-parental shared environmental factors (C) from the model (model SH-2: χ2(1) < 1, ns) did not result in a significant worsening of the model fit. Elimination of all other factors (i.e., A + D, CT, rGE, and/or ASM), however, did result in a significant worsening of the model fit. Under the reduced SH model (model SH-2), individual differences in general intelligence were explained by AFootnote 2 (estimated at 0%), D (58%), CT (24%), and E (18%). The presence of D in the absence of A, as noted above is considered biologically implausible (Falconer and Mackay 1989).
Standardized variance components for general intelligence based on full (left) and reduced (right) SH and PA models. ASM assortative mating (phenotypic assortment under the PA model; social homogamy under the SH model), A additive genetic factors, D genetic dominance, E non-shared environmental factors, C shared environmental factors, CT cultural transmission, rGE correlation between A and CT, Under the PA model, rGE is negative since the unstandardized parameter estimate of CT is negative; model numbering corresponds to model numbering in Table 2
Phenotypic assortment model
Within the full PA model, effects of A (58%) and ASM (23%) were substantial, effects of CT (8%) and E (11%) were modest, and effects of C (0%) and D (0%) were estimated at zero. The model induced correlation (rGE) between A and CT was estimated at −0.36 (see Fig. 3). Note that standardized variance components are by definition positive but that the unstandardized parameter estimate of CT was negative. The negative effect of CT consequently induced a negative correlation between the parental and offspring phenotypes.
Eliminating shared environmental factors C (model PA-2: χ2(1) < 1, ns) or CT (model PA-3: χ2(1) = 1.61, ns) from the model did not result in a significant worsening of the model fit. Subsequently eliminating genetic factors A + D (model PA-4: χ2(2) = 300.76, p < .001) or D (model PA-5: χ2(1) = 28.03, p < .001), or the effect of ASM (model PA-6: χ2(1) = 41.28, p < .001) from the model resulted in a significant worsening of the model fit, implying that individual differences in general intelligence are to a large extent explained by genetic factors but also by phenotypic assortment. When the genetic factors A and D were fixed to zero, the relative influence of ASM decreased from 11 to 0%, which implies that phenotypic assortment is based on a phenotype that completely reflects the genotype. Within model PA-3, the estimate of D, increased dramatically to 27% when CT was eliminated from the model. Consequently, eliminating D from the model while CT was already fixed to zero led to a significant worsening of the model fit.
From the above it is clear that although D was estimated at zero in the full model, it accommodated the variance that was previously ascribed to CT. When non-significant CT was dropped from the model, D then becomes highly significant. To test the reverse, i.e., whether the estimate of CT changed when D was eliminated from the model first (i.e., CT is included in the model), we conducted a second series of nested models: eliminating D from a model including CT did not change the estimate of CT (model PA-7: CT = 8%) and did not result in a worse model fit (model PA-7: χ2(1) < 1, ns). That is, although D and CT were both not significant when the other effect was estimated freely, both effects could not be eliminated simultaneously (model PA-5: χ2(1) = 28.03, p < .001 and model PA-8: χ2(2) = 29.64, p < .001). In this second series of nested models (i.e., models PA-7 to PA-10), additive genetic factors (model PA-9: χ2(1) = 116.00, p < .001) and effects of phenotypic assortment (model PA-10: χ2(1) = 43.82, p < .001) were also significant.
Estimates of D and negative CT are identified by a similar pattern of correlations. D is identified by a higher DZ correlation relative to the parent-offspring correlation since dominance effects are correlated in DZ twin pairs (.25) but not in parent-offspring pairs. Negative CT is also identified by higher DZ correlation relative to the parent-offspring correlation as negative CT results from an inhibiting effect from parents on their offspring’s general intelligence. However, like the effect of positive CT, the effect of negative CT is similar for both members of a DZ twin pair, resulting in an increased DZ twin correlation. Thus, both D and CT are expected to increase the DZ twin correlation relative to the parent-offspring correlation. Apparently, the current study design lacks information and power to reliably estimate both parameters simultaneously.
Summarizing the results for both reduced models: model PA-3 included additive genetic factors (44%), genetic dominance (27%), phenotypic assortment (11%) and non-shared environmental factors (18%). Model PA-7 included additive genetic factors (58%), phenotypic assortment (23%), negative CT (8%), and non-shared environmental factors (11%); correlation between A and CT was −0.36. Comparing both reduced PA models (model PA-3 and model PA-7) showed no significant difference in likelihood, but showed lower AIC, BIC, and DIC for model PA-3. Based on these criteria, model PA-3 was to be preferred.
Parameter estimates of the full and reduced SH and PA assortment models are presented in Fig. 3. Results were replicated for both verbal and performance intelligence (see online supplement for details).
Discussion
The aim of the present study was to investigate individual differences in intelligence in adults while taking non-random mating of spouses into account. To this end, two different assortment models were fitted to the data, a social homogamy (SH) model and a phenotypic assortment (PA) model. For general intelligence, as well as for verbal and performance intelligence, the SH model fitted the data comparatively worse than the PA model. The most parsimonious model under SH was a model including genetic dominance, CT, and non-shared environmental factors. The effect of additive genetic factors was estimated close to zero. Under PA, we ended up with two alternative models as the estimates of genetic dominance and negative CT were confounded: (i) a model including negative CT but not genetic dominance, or (ii) a model including genetic dominance but not negative CT. Both PA models also included additive genetic variance, variance explained by assortative mating, and non-shared environmental variance. Similar results were obtained for verbal intelligence and performance intelligence subscales (see online supplement for details).
The overall misfit observed for the SH model is likely to be due to the high estimate of genetic dominance, which is, in the light of practically absent additive genetic effects, biologically unlikely (Falconer and Mackay 1989). We assume that the estimate of genetic dominance is increased in order to accommodate the observed decrease in spousal correlations with increasing genetic distance, a pattern of correlations that is not expected under an SH model (see Fig. 2).
With respect to the PA models, the present study lacked the information to disentangle effects of genetic dominance and negative CT. Based on test statistics, the PA model including genetic dominance (but not CT) fitted the data relatively better than the PA model including negative CT (but not genetic dominance). Moreover, significant negative CT seems somewhat unlikely in the context of general intelligence, as it would, for example, imply that smarter parents suppress their children’s cognitive abilities. An alternative explanation of negative CT, however, is possible incomplete genetic isomorphism across adult ages with e.g., increased genetic contribution in young to middle adulthood and decreased genetic contribution at later ages (Pedersen et al. 1992; Brandt et al. 1993; Finkel et al. 1998) or different (sets of) genes that might be related to general intelligence in different stages in life (e.g., Deary et al. 2002). Further analyses are required to disentangle different explanations for negative CT in the context of general intelligence.
In the present design, negative CT and genetic dominance are largely confounded, consequently genetic dominance could only be detected when effects of CT were eliminated from the model. Results for general intelligence showed that estimates of genetic dominance increased from 0 to 27% if negative CT was eliminated from the model. Based on test statistics and interpretation of the parameter estimates, we suggest that the PA model including additive genetic factors, genetic dominance deviation, positive assortative mating, and non-shared environmental factors (Model PA-3 in Table 2) provides the most plausible description of the observed data. Such a model would support the hypothesis that in adults, genetic dominance might go undetected due to the presence of assortative mating when assortment is not adequately modeled. Note that negative CT, if present, could mask the presence of genetic dominance as well.
Although, the design applied in the present study allows one to model the effects of assortative mating on the estimates of the variance components of intelligence, some limitations should be noted. First, mating behavior was assumed to be due to pure SH or pure PA, in which SH was defined as purely environmental similarities and PA as assortment purely based on phenotypic similarities. It is however not unlikely that mating behavior is influenced by both processes, i.e., mixed assortment. Moreover, social stratification may itself be driven by genetic stratification between populations such that assortment due to SH may in fact have a genetic background. Similarly, PA may be purely environmental in the case that the trait under study is not influenced by genetic factors.
Second, it is possible that cohort differences in assortment exist, i.e., that the process underlying assortative mating differs for different birth cohorts. For example, mating in the first half of the 20th century may generally have been based on similarity in social milieus for spouses, while urbanization and increasing equality of educational opportunities between men and women may have increased the influence of PA in latter generations. Studies including large generational cohorts are required to model both processes simultaneously, or to model assortment changes over time.
Third, satisfying the distinction between negative CT and genetic dominance was difficult as these two effects were largely confounded in the present study design. Different relatives, such as half-sibs, adoptees, or twins that have grown up in separate households, would need to be included to disentangle those two processes.
Fourth, within the present study we did not model the correlation between genetic and non-shared environmental factors as this correlation is not identified as long as no specific non-shared environmental factors are measured and included in the model. In the context of general intelligence, correlation between genes and non-shared environmental factors such as e.g., education and profession, has, however, been suggested by Haworth et al. (2010). Ignoring the correlation between genes and non-shared environmental factors may lead to an overestimation of the genetic effects (Purcell 2002).
Thus far, only a few studies have suggested the presence of genetic dominance for general intelligence in adults (Chipuer et al. 1990; Fulker and Eysenck 1979). The results of these studies were, however, based on combined samples (i.e., different samples from different studies were combined within one study) with different measures of intelligence. A disadvantage of such a combined design is that general intelligence is assessed using different intelligence tests at different points in time, which may affect estimates of the correlation between relatives. Correlations between individuals measured with different tests and/or in different points in time may be relatively decreased compared to correlations between relatives assessed with the same test and/or at a similar moment in time. This, in turn, may lead to biased estimates of the variance components. The advantage of the present study is that a single intelligence test was used for all participants. The present study is also unique in its design as it includes adult MZ and DZ twins, their non-twin siblings and the parents, spouses and adult offspring of the twins and non-twin siblings.
Reynolds et al. (2000) emphasized the importance of considering assortative mating in a twin-family study on educational attainment and fluid ability in adults. In that study, effects of SH and PA were modeled simultaneously (i.e., mixed assortment) in a sample of 116 twin-spouse sets; effects of CT and genetic dominance were however not considered in this study. Both SH and PA contributed to the spousal similarities for educational attainment and fluid ability in a multivariate design. Considering both SH and positive PA in the context of general intelligence requires larger sample sizes than we had currently available (Heath and Eaves 1985). A mixed model might however nicely fit to the pattern of phenotypic correlations between relatives that we observe in the present study, i.e., a decrease in correlations with increasing genetic distance (attributable to PA) and generally high correlations between relatives with no genetic relationship (attributable to SH).
Results from the present study have several implications. First, our results suggest that the well recognized high influence of additive genetic factors on individual differences in intelligence in adults may partly reflect more complex processes such as genetic dominance and positive assortative mating. The extended twin-family design evidently allows the disentanglement of various sources of individual differences in intelligence, and this design could also prove important in the context of a wide variety of other traits for which assortative mating has been reported, such as human height, body mass index, smoking behavior, personality traits, and psychiatric disorders (Silventoinen et al. 2003; Agrawal et al. 2006; Glicksohn and Golan 1999; Maes et al. 1998). Heritability estimates for these traits are generally based on twin correlations, while effects of assortative mating are not considered. Consequently, current knowledge about causes of individual differences in numerous traits may need to be reconsidered with effects of assortative mating are taken into account.
Second, our finding that genetic dominance explains part of the variance in adult intelligence is interesting in the context of the well-known increase in heritability of intelligence over age. It is generally recognized that shared environmental influences disappear after adolescence as children leave their parental home. An alternative is that dominance variance is present in children as well, but goes undetected due to larger shared environmental variance or effects of CT in childhood. In addition, the reported effects of shared environmental variance in childhood may be overestimated due to assortative mating that is not accounted for. To test this hypothesis, the CTD should be extended with parents of young twins.
Third, the conclusion that in adulthood, the genetic variation of general intelligence may not be merely additives in nature may be important in the context of gene finding studies for general intelligence. Genome wide association (GWA) studies generally test for main effects of alleles, and do not consider interaction (Plomin et al. 2001b; Seshadri et al. 2007; Butcher et al. 2008), this has two implications. The extent of ‘missing heritability’ is lower, since it is only the unexplained part of the additive variance that is missing, not the non-additive genetic variance. Moreover, considering non-additive genetic effects within GWA studies for general intelligence might enhance their gene finding success. A major problem in this context, however, is that even larger sample sizes are required to detect non-additive alleles in GWA studies. Other approaches, such as the candidate gene approach, or functional pathway analyses might prove more suited to better our understanding of the contribution of the additive genetic factors and genetic dominance deviation as these studies do not suffer from power problems such as GWA studies.
To conclude, we demonstrated that the high heritability of intelligence is not only due to additive genetic factors but also to non-additive genetic factors or to negative CT, and the consequences of assortment. Analyses of verbal intelligence and performance intelligence support these results. Future studies of intelligence need to accommodate both assortment and non-additive genetic influences. Such studies could for example use genomic marker data to distinguish underlying mechanisms of spouse correlations (e.g., assortative mating due to PA would show increased genetic relatedness between spouses relative to random individuals of a population, whereas assortative mating due to SH would not). Moreover, gene finding studies may benefit from genetic resemblance between spouses since genetic variants that are shared between spouses more often than expected by chance, are possibly the same variants that account for part of the variance in general intelligence.
Notes
All relations are expressed in relation to the twin. Twin–spouse relations do also incorporate spousal relations between the parents of the twins and between the sibling with its spouse.
Please note that A is included in the model because a model that includes D and not A is biologically implausible, but can still be estimated at zero (Falconer and Mackay 1989).
References
Agrawal A, Heath AC, Grant JD, Pergadia ML, Statham DJ, Bucholz KK et al (2006) Assortative mating for cigarette smoking and for alcohol consumption in female Australian twins and their spouses. Behav Genet 36:553–566
Ando J, Ono Y, Wright MJ (2001) Genetic structure of spatial and verbal working memory. Behav Genet 31:615–624
Boomsma DI, de Geus EJ, Vink JM, Stubbe JH, Distel MA, Hottenga JJ et al (2006) Netherlands twin register: from twins to twin families. Twin Res Hum Genet 9:849–857
Bouchard TJ Jr, McGue M (1981) Familial studies of intelligence: a review. Science 212:1055–1059
Brandt J, Welsh KA, Breitner JC, Folstein MF, Helms M, Christian JC (1993) Hereditary influences on cognitive functioning in older men. A study of 4000 twin pairs. Arch Neurol 50(6):599–603
Butcher LM, Davis OS, Craig IW, Plomin R (2008) Genome-wide quantitative trait locus association scan of general cognitive ability using pooled DNA and 500 K single nucleotide polymorphism microarrays. Genes Brain Behav 7:435–446
Chipuer HM, Rovine MJ, Plomin R (1990) Lisrel modeling—genetic and environmental-influences on Iq revisited. Intelligence 14:11–29
Deary IJ, Whiteman MC, Pattie A, Starr JM, Hayward C, Wright AF, Carothers A, Whalley LJ (2002) Ageing: cognitive change and the APOE ε 4 allele. Nature 418:932
Eaves L, Heath A, Martin N, Maes H, Neale M, Kendler K et al (1999) Comparing the biological and cultural inheritance of personality and social attitudes in the Virginia 30, 000 study of twins and their relatives. Twin Res 2:62–80
Falconer DS, Mackay TFC (1989) Introduction to quantitative genetics, 4th edn. Pearson Education Limited, Harlow
Finkel D, Wille DE, Matheny AP (1998) Preliminary results from a Twin study of infant-caregiver attachment. Behav Genetics (28)1:1–8
Fisher R (1918) The correlation between relatives on the supposition of Mendelian inheritance. Trans R Soc Edinborough 52:399–433
Fulker DW (1982) Extensions of the classical twin method. In: Genetics Human (ed) Part A: the unfolding genome. Alan R. Liss Inc., New York, pp 395–406
Fulker DW, Eysenck HJ (1979) Nature and nurture: heredity. In: Eysenck HJ (ed) The structure and measurement of intelligence. Springer, New York, pp 102–131
Glicksohn J, Golan H (1999) Personality, cognitive style and assortative mating. Personal Individ Differ 30:1199–1209
Haworth CM, Wright MJ, Luciano M, Martin NG, de Geus EJ, van Beijsterveldt CE et al (2010) The heritability of general cognitive ability increases linearly from childhood to young adulthood. Mol Psychiatry 15:1112–1120
Heath AC, Eaves LJ (1985) Resolving the effects of phenotype and social background on mate selection. Behav Genet 15:15–30
Jencks D, Smith M, Acland H, Bane MJ, Cohen D, Gintis H et al (1972) Inequality: a reassessment of the effect of family and schooling in America. Basic Books, New York
Keller MC, Medland SE, Duncan LE (2009a) Are extended twin family designs worth the trouble? A comparison of the bias, precision, and accuracy of parameters estimated in four twin family models. Behav Genet 12(1):19–25
Keller MC, Medland SE, Duncan LE, Hatemi PK, Neale MC, Maes HH et al (2009b) Modeling extended twin family data I: description of the Cascade model. Twin Res Hum Genet 12:8–18
Loehlin JC (1978) Heredity-environment analyses of Jencks’s IQ correlations. Behav Genet 8:415–436
Luciano M, Wright M, Smith GA, Geffen GM, Geffen LB, Martin NG (2001) Genetic covariance among measures of information processing speed, working memory, and IQ. Behav Genet 31:581–592
Lynch M, Walsh B (1998) Resemblance between relatives. In: Lynch M, Walsh B (eds) Genetics and aanalyses of quantitative traits. Sinauer Associates Inc, Sunderland
Maes HH, Neale MC, Kendler KS, Hewitt JK, Silberg JL, Foley DL et al (1998) Assortative mating for major psychiatric diagnoses in two population-based samples. Psychol Med 28:1389–1401
Maes HH, Neale MC, Medland SE, Keller MC, Martin NG, Heath AC et al (2009) Flexible Mx specification of various extended twin kinship designs. Twin Res Hum Genet 12:26–34
Mascie-Taylor CG (1989) Spouse similarity for IQ and personality and convergence. Behav Genet 19:223–227
McGue M, Bouchard TJ Jr (1984) Adjustment of twin data for the effects of age and sex. Behav Genet 14:325–343
Nagylaki T (1978) The correlation between relatives with assortative mating. Ann Hum Genet 42:131–137
Neale MC (1994) Mx: statistical modeling [Computer software]
Pedersen NL, McClearn GE, Plomin R, Nesselroade JR (1992) Effects of early rearing environment on twin similarity in the last half of the life span. Br J Dev Psychol 10:255–267
Plomin R (1999) Genetics and general cognitive ability. Nature 402:C25–C29
Plomin R, DeFries JC, McClearn GE, McGuffin P (2001a) Behavioral genetics, 4th edn. Worth Publishers, New York
Plomin R, Hill L, Craig IW, McGuffin P, Purcell S, Sham P et al (2001b) A genome-wide scan of 1842 DNA markers for allelic associations with general cognitive ability: a five-stage design using DNA pooling and extreme selected groups. Behav Genet 31:497–509
Posthuma D, de Geus EJ, Boomsma DI (2001a) Perceptual speed and IQ are associated through common genetic factors. Behav Genet 31:593–602
Posthuma D, Neale MC, Boomsma DI, de Geus EJ (2001b) Are smarter brains running faster? Heritability of alpha peak frequency, IQ, and their interrelation. Behav Genet 31:567–579
Purcell S (2002) Variance components models for gene-environment interaction in twin analysis. Twin Res 5:554–571
Reynolds CA, Baker LA, Pedersen NL (2000) Multivariate models of mixed assortment: phenotypic assortment and social homogamy for education and fluid ability. Behav Genet 30:455–476
Rice J, Cloninger CR, Reich T (1978) Multifactorial inheritance with cultural transmission and assortative mating. I. Description and basic properties of the unitary models. Am J Hum Genet 30:618–643
Seshadri S, DeStefano AL, Au R, Massaro JM, Beiser AS, Kelly-Hayes M et al (2007) Genetic correlates of brain aging on MRI and cognitive test measures: a genome-wide association and linkage analysis in the Framingham study. BMC Med Genet 8(Suppl 1):S15
Silventoinen K, Kaprio J, Lahelma E, Viken RJ, Rose RJ (2003) Assortative mating by body height and BMI: finnish twins and their spouses. Am J Hum Biol 15:620–627
van Leeuwen M, van den Berg SM, Boomsma DI (2008) A twin-family study of general IQ. Learn Individ Differ 18:76–88
Wechsler D (1997) Wechsler adult intelligence scale-third edition. Swets and Zeitlinger, Lisse
Willemsen G, Posthuma D, Boomsma DI (2005) Environmental factors determine where the Dutch live: results from the Netherlands twin register. Twin Res Hum Genet 8:312–317
Acknowledgments
This study was supported by NWO/MaGW VIDI-016-065-318, HFSP RG0154, and NWO/MaGW VENI-415-08-025. The authors thank Dorret Boomsma for helpful comments on previous versions of the manuscript and facilities of the NTR. The authors also thank Marjolein Hoogeboom, Madelon Grit and Özgül Uysal for assisting in the data collection.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Eric Turkheimer.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Vinkhuyzen, A.A., van der Sluis, S., Maes, H.H.M. et al. Reconsidering the Heritability of Intelligence in Adulthood: Taking Assortative Mating and Cultural Transmission into Account. Behav Genet 42, 187–198 (2012). https://doi.org/10.1007/s10519-011-9507-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10519-011-9507-9