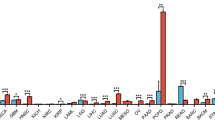
For tumors with chimeric NTRK genes, entrectinib and larotrectinib can be prescribed regardless of tumor localization. We compared changes in the transcriptional activity of genes in brain tumors (BT) and thyroid cancer (TC) with rearrangement (NTRK+) and without rearrangement (NTRK-) of the NTRK genes using The Cancer Genome Atlas (TCGA) and Gene Expression Omnibus (GEO) databases. We revealed an increase in the transcription of the JUN gene in NTRK+ samples in comparison with NTRK- samples: by 1.6 times for BT (p=0.239) and by 2.5 times for TC (p=0.003). The transcription of eight HOX genes in NTRK+ BT samples was also increased (by 85-725 times, p<0.05) in comparison with NTRK-. In NTRK+ TC samples, the level of miR-31 and miR-542 was statistically significantly higher (by 3 and 2.5 times, respectively) than in NTRK-samples. For the NTRK+ BT samples, the levels of miR-10b, miR-182, and miR-21 more than 5-fold surpassed the corresponding values in NTRK-samples (p<0.05). These findings reflect differences in activation of gene transcription resulting from NTRK gene rearrangement in BT and TC.
Similar content being viewed by others
References
Arévalo JC, Wu SH. Neurotrophin signaling: many exciting surprises! Cell. Mol. Life Sci. 2006;63(13):1523-1537. doi: https://doi.org/10.1007/s00018-006-6010-1
Vaishnavi A, Le AT, Doebele RC. TRKing down an old oncogene in a new era of targeted therapy. Cancer Discov. 2015;5(1):25-34. doi: https://doi.org/10.1158/2159-8290.CD-14-0765
Kechin A, Borobova V, Kel A, Ivanov A, Filipenko M. ErbB4 Is a Potential Key Regulator of the Pathways Activated by NTRK-Fusions in Thyroid Cancer. Appl. Sci. 2022;12(5):2506. doi: https://doi.org/10.3390/app12052506
Suntsova M, Gaifullin N, Allina D, Reshetun A, Li X, Mendeleeva L, Surin V, Sergeeva A, Spirin P, Prassolov V, Morgan A, Garazha A, Sorokin M, Buzdin A. Atlas of RNA sequencing profiles for normal human tissues. Sci. Data. 2019;6(1):36. doi: https://doi.org/10.1038/s41597-019-0043-4
Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15(12):550. doi: https://doi.org/10.1186/s13059-014-0550-8
Gazon H, Barbeau B, Mesnard JM, Peloponese JM Jr. Hijacking of the AP-1 Signaling Pathway during Development of ATL. Front. Microbiol. 2018;8:2686. doi: https://doi.org/10.3389/fmicb.2017.02686
Pramanik R, Qi X, Borowicz S, Choubey D, Schultz RM, Han J, Chen G. p38 isoforms have opposite effects on AP-1-dependent transcription through regulation of c-Jun. The determinant roles of the isoforms in the p38 MAPK signal specificity. J. Biol. Chem. 2003;278(7):4831-4839. doi: https://doi.org/10.1074/jbc.M207732200
Chen SL, Lin ST, Tsai TC, Hsiao WC, Tsao YP. ErbB4 (JM-b/CYT-1)-induced expression and phosphorylation of c-Jun is abrogated by human papillomavirus type 16 E5 protein. Oncogene. 2007;26(1):42-53. doi: https://doi.org/10.1038/sj.onc.1209768
Gonçalves CS, Le Boiteux E, Arnaud P, Costa BM. HOX gene cluster (de)regulation in brain: from neurodevelopment to malignant glial tumours. Cell. Mol. Life Sci. 2020;77(19):3797-3821. doi: https://doi.org/10.1007/s00018-020-03508-9
Deforzh E, Uhlmann EJ, Das E, Galitsyna A, Arora R, Saravanan H, Rabinovsky R, Wirawan AD, Teplyuk NM, El Fatimy R, Perumalla S, Jairam A, Wei Z, Mirny L, Krichevsky AM. Promoter and enhancer RNAs regulate chromatin reorganization and activation of miR-10b/HOXD locus, and neoplastic transformation in glioma. Mol. Cell. 2022;82(10):1894-1908.e5. doi: https://doi.org/10.1016/j.molcel.2022.03.018
Wang C, Wang Y, Xu W, Lin X, Xi J, Wang S, Lin L, Yuan F, Wang A, Wang C, Luo X, Xu Q, Yin R, Zhang Y, Huang X, Chen Y. Generation of an induced pluripotent stem cell line from an Ohtahara syndrome patient with the hemizygous mutation p.Q503Afs*28 (c.1507_1508del) in the ARX gene. Stem Cell Res. 2021;59:102621. doi: https://doi.org/10.1016/j.scr.2021.102621
Wei S, Li J, Tang M, Zhang K, Gao X, Fang L, Liu W. STAT3 and p63 in the Regulation of Cancer Stemness. Front. Genet. 2022;13:909251. doi: https://doi.org/10.3389/fgene.2022.909251
Long S, Wang J, Weng F, Xiang D, Sun G. Extracellular Matrix Protein 1 Regulates Colorectal Cancer Cell Proliferative, Migratory, Invasive and Epithelial-Mesenchymal Transition Activities Through the PI3K/AKT/GSK3β/Snail Signaling Axis. Front. Oncol. 2022;12:889159. doi: https://doi.org/10.3389/fonc.2022.889159
Yu T, Ma P, Wu D, Shu Y, Gao W. Functions and mechanisms of microRNA-31 in human cancers. Biomed. Pharmacother. 2018;108:1162-1169. doi: https://doi.org/10.1016/j.biopha.2018.09.132
Yang C, Wang MH, Zhou JD, Chi Q. Upregulation of miR-542-3p inhibits the growth and invasion of human colon cancer cells through PI3K/AKT/survivin signaling. Oncol. Rep. 2017;38(6):3545-3553. doi: https://doi.org/10.3892/or.2017.6054
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 175, No. 1, pp. 91-99, January, 2023
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kechin, A.A., Koryukov, M.A., Smertina, M.A. et al. Differences in Transcriptomic Profiles of Brain and Thyroid Tumors with NTRK Gene Rearrangement. Bull Exp Biol Med 175, 78–85 (2023). https://doi.org/10.1007/s10517-023-05815-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05815-0