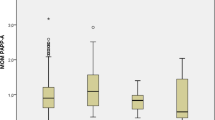
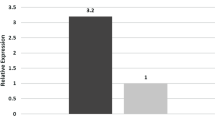
We studied regulation of the expression of placental growth factor (PlGF) that plays an important role in the trophoblast cells functions and reduced production of which by the placenta is associated with gestational complications. PlGF expression is regulated by transcription factors whose activity is controlled by sumoylation, which is also necessary for the formation of an adequate cellular response to hypoxia. Increased sumoylation and reduced expression of some miRNA targeted to transcription factors VEGF, GCM-1, and UBC9 conjugating SUMO with targets protein were detected in the placenta. Correlations were revealed between changes in the expression of miR-423-3p and miR-652-3p, the level of SUMO 1-4 and UBC9 in the placenta, reduced concentration of PlGF, and increased sFlt-1/PlGF ratio in the blood of pregnant women with early-onset preeclampsia, which attests to the presence of a regulatory mechanism along the axis of miR-652-3p/SUMO-2/3/4/UBC9/GCM-1/PlGF.
Similar content being viewed by others
References
Burton GJ, Redman CW, Roberts JM, Moffett A. Pre-eclampsia: pathophysiology and clinical implications. BMJ. 2019;366:l2381. https://doi.org/10.1136/bmj.l2381
Hung TH, Burton GJ. Hypoxia and reoxygenation: a possible mechanism for placental oxidative stress in preeclampsia. Taiwan. J. Obstet. Gynecol. 2006;45(3):189-200. https://doi.org/10.1016/S1028-4559(09)60224-2
Li S, Roberson MS. Dlx3 and GCM-1 functionally coordinate the regulation of placental growth factor in human trophoblast-derived cells. J. Cell. Physiol. 2017;232(10):2900-2914. https://doi.org/10.1002/jcp.25752
Gobble RM, Groesch KA, Chang M, Torry RJ, Torry DS. Differential regulation of human PlGF gene expression in trophoblast and nontrophoblast cells by oxygen tension. Placenta. 2009;30(10):869-875. https://doi.org/10.1016/j.placenta.2009.08.003
Caillon H, Tardif C, Dumontet E, Winer N, Masson D. Evaluation of sFlt-1/PlGF ratio for predicting and improving clinical management of pre-eclampsia: experience in a specialized perinatal care center. Ann. Lab. Med. 2018;38(2):95-101. https://doi.org/10.3343/alm.2018.38.2.95
Gill G. Post-translational modification by the small ubiquitin-related modifier SUMO has big effects on transcription factor activity. Curr. Opin. Genet. Dev. 2003;13(2):108-113. https://doi.org/10.1016/s0959-437x(03)00021-2
Chou CC, Chang C, Liu JH, Chen LF, Hsiao CD, Chen H. Small ubiquitin-like modifier modification regulates the DNA binding activity of glial cell missing Drosophila homolog a. J. Biol. Chem. 2007;282(37):27 239-27 249. https://doi.org/10.1074/jbc.M700351200
Chang HM, Yeh ETH. SUMO: from bench to bedside. Physiol. Rev. 2020;100(4):1599-1619. https://doi.org/10.1152/physrev.00025.2019
Baczyk D, Audette MC, Coyaud E, Raught B, Kingdom JC. Spatiotemporal distribution of small ubiquitin-like modifiers during human placental development and in response to oxidative and inflammatory stress. J. Physiol. 2018;596(9):1587-1600. https://doi.org/10.1113/JP275288
Serocki M, Bartoszewska S, Janaszak-Jasiecka A, Ochocka RJ, Collawn JF, Bartoszewski R. miRNAs regulate the HIF switch during hypoxia: a novel therapeutic target. Angiogenesis. 2018;21(2):183-202. https://doi.org/10.1007/s10456-018-9600-2
Zheng B, Bernier M, Zhang XH, Suzuki T, Nie CQ, Li YH, Zhang Y, Song LL, Shi HJ, Liu Y, Zheng CY, Wen JK. miR-200c-SUMOylated KLF4 feedback loop acts as a switch in transcriptional programs that control VSMC proliferation. J. Mol. Cell. Cardiol. 2015;82:201-212. https://doi.org/10.1016/j.yjmcc.2015.03.01
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25(4):402-408. https://doi.org/10.1006/meth.2001.1262
Baczyk D, Drewlo S, Kingdom JC. Emerging role of sumoylation in placental pathology. Placenta. 2013;34(7):606-612. https://doi.org/10.1016/j.placenta.2013.03.012
Gusar V, Timofeeva A, Chagovets V, Kan N, Vasilchenko O, Prozorovskaya K, Ivanets T, Sukhikh G. Preeclampsia: the interplay between oxygen-sensitive miRNAs and erythropoietin. J. Clin. Med. 2020;9(2):574. https://doi.org/10.3390/jcm9020574
Shi Z, Liu B, Li Y, Liu F, Yuan X, Wang Y. MicroRNA-652-3p promotes the proliferation and invasion of the trophoblast HTR-8/SVneo cell line by targeting homeobox A9 to modulate the expression of ephrin receptor B4. Clin. Exp. Pharmacol. Physiol. 2019;46(6):587-596. https://doi.org/10.1111/1440-1681.13080
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 3, pp. 200-204, September, 2022
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gusar, V.A., Timofeeva, A.V., Chagovets, V.V. et al. Regulation of the Placental Growth Factor Mediated by Sumoylation and Expression of miR-652-3p in Pregnant Women with Early-Onset Preeclampsia. Bull Exp Biol Med 174, 174–178 (2022). https://doi.org/10.1007/s10517-022-05668-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-022-05668-z