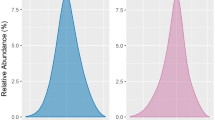
We compared anti-IIa activity of a heparin analogue and a reference product was carried out to confirm their biosimilarity. The experiment was based on the method of estimation of anti-IIa activity of a commercial sodium heparin preparation according to United States Pharmacopoeia. High similarity of the two medicinal heparin preparations by this parameter is shown. The method is recommended for the use in comparability studies.
Similar content being viewed by others
References
R. S. Aquino, M. S. Pereira, B. C. Vairo et al., Thromb. Haemost., 103, No. 5, 1005-1015 (2010).
Co-ordination Group for Mutual Recognition and Decentralised Procedures – Human. Overview of Biological Active Substances of Non-Recombinant Origin. [remote access: URL: www.hma.eu/fileadmin/dateien/Human_Medicines/CMD_h_/procedural_guidance/Compilation_Biological_Active_Substance_non-recombinant_origin.pdf]. Accessed date: August 20, 2013.
Council of Europe. European Pharmacopoeia, Strasbourg (2013).
European Medicines Agency. Guideline on Similar Biological Medicinal Products Containing Biotechnology-Derived Proteins as Active Substance: Quality Issues (EMEA/CHMP/ BWP/49348/2005). [remote access: URL: www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003953.pdf]. Accessed date: December 17, 2013.
European Medicines Agency. Similar Biological Medicinal Products (CHMP/437/04). [remote access: URL: www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003517.pdf]. Accessed date: December 17, 2013.
A. W. McMahon, R. G. Pratt, T. A. Hammad et al., Pharmacoepidemiol. Drug Saf., 19, No. 9, 921-923 (2010).
M. D. Rothmann, B. L. Wiens, I. S. F. Chan. Design and Analysis of Non-Inferiority Trials, Boca Raton (2012).
Scientifi c Opinion on BSE Risk in Bovine Intestines. EFSA Panel on Biological Hazards, EFSA J., No. 1317, 1-19 (2009).
United States Pharmacopeia and National Formulary (USP 36-NF 21). Rockville (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 158, No. 10, pp. 448-451, October, 2014
Rights and permissions
About this article
Cite this article
Mironov, A.N., Goryachev, D.V., Garvishina, E.V. et al. Specific Anti-IIa Activity is a Key Indicator of Safety and Efficacy in Validation of Biosimilarity of Unfractioned Heparin Preparations. Bull Exp Biol Med 158, 450–452 (2015). https://doi.org/10.1007/s10517-015-2782-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-015-2782-3