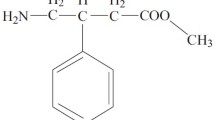
We studied pharmacokinetics of dilept after single cross-administration of tablets and substance of dilept in a dose of 40 mg to rabbits. The following pharmacokinetic parameters were calculated: maximum plasma concentration of dilept, time to maximum observed concentration, area under the pharmacokinetic curve, elimination half-life, and relative bioavailability. Dilept concentration in blood plasma was estimated using ultra-fast liquid chromatography with mass spectrometry detection. Relative bioavailability of dilept tablets was 93.46±28.91% of that for the substance.
Similar content being viewed by others
References
N. V. Arkhipenko, S. A. Appolonova, T. G. Sobolevskii, et al., Khim.-Farm. Zh., 43, No. 5, 53-56 (2009).
S. B. Seredenin, T. A. Voronina, T. A. Gudasheva, et al., Patent RF No. 2091390, Substituted Prolyl-Tyrosine with Psychotropic Activity, Byull. No. 27.
R. V. Shevchenko, A. A. Litvin, G. B. Kolyvanov, et al., Eksp. Klin. Farmakol., 76, No. 6, 34-37 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 157, No. 6, pp. 704-706, June, 2014
Rights and permissions
About this article
Cite this article
Shevchenko, R.V., Litvin, A.A., Kolyvanov, G.B. et al. Comparison of Pharmacokinetics and Relative Bioavailability of Tablets and Substance of New Dipeptide Neuroleptic Dilept. Bull Exp Biol Med 157, 735–737 (2014). https://doi.org/10.1007/s10517-014-2655-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-014-2655-1