Abstract
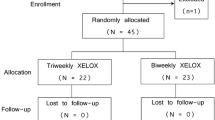
We studied the efficiency of combined chemotherapy with tomudex and xeloda preparations in patients with metastasizing colorectal cancer. The treatment (240 courses) was effective in 75% patients. Time median before progression was 6.3 months, mean durations of partial remission and stabilization were 7.8 months, total survival 15.5 months, total survival after effective treatment was 18.2 months. The most prevalent manifestations of III–IV degree toxicity were neutropenia, diarrhea, and asthenia. Other symptoms of toxicity (increased transaminase level, bilirubin, nausea, vomiting) were observed in less than 3% courses. Thus, treatment with tomudex and xeloda are effective and safe for outpatient chemotherapy.
Similar content being viewed by others
References
C. Twelves, Eur. J. Cancer, 38, Suppl. 2, 15–20 (2002).
G. Cocconi, Tumori, 83, Suppl., S72 (1997).
D. Cunningham, J. Zalcberg, U. Rath, et al., Ann. Oncol., 7, No. 9, 961–965 (1996).
R. Pazdur and M. Vincent, Am. J. Clin. Oncol., 21, 279–283 (1997).
P. M. Hoff, R. Ansari, G. Batist, et al., J. Clin. Oncol., 19, No. 8, 2282–2292 (2001).
S. Mayer, R. Hilger, P. Muller, et al., Eur. J. Cancer, 35, Suppl. 4, 74 (1999).
S. Vazquez, J. M. Vieitez, P. Garcia Alfonso, et al., Ann. Oncol., 11, Suppl. 14, 52 (2000).
Author information
Authors and Affiliations
Additional information
__________
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 145, No. 2, pp. 210–212, February, 2008
Rights and permissions
About this article
Cite this article
Vakhabova, J.V., Semenov, N.N., Dobrova, N.V. et al. Clinical study of combined use of tomudex (raltitrexed) and xeloda (capecitabine) as first-line treatment for patients with metastasizing colorectal cancer. Bull Exp Biol Med 145, 249–251 (2008). https://doi.org/10.1007/s10517-008-0062-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-008-0062-1