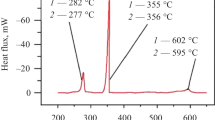
A eutectic mixture of lithium, sodium, and potassium fluorides is considered as a fuel salt for molten-salt reactors. For this reason, information about the solubility in it of fluorides of fissile materials and fission products is the most important when choosing a salt composition for a molten-salt reactor. In this work, the liquidus and solidus temperatures of the LiF–NaF–KF–CeF3 quasi-binary system are determined by thermal analysis and differential scanning calorimetry. The system has one eutectic point at CeF3 content 3 mol.% and temperature 445°C and three peritectic points – 7.5, 15, and 20.5 mol.% at 472, 512, and 600°C, respectively. The solubility of CeF3 in LiF–NaF–KF is 10 mol.% at 500°C and reaches 25 mol.% at 665°С.
Similar content being viewed by others
References
S. Chwaszczewski and B. Slowinski, “Transmutation of radioactive waste,” App. Energy, 75, No. 1–2, 87–96 (2003).
A. M. Degtyarev and L. I. Ponomarev, “Molten-salt reactor with a fast neutron spectrum based on LiF–NaF–KF,” At. Energ., 112, No. 6, 367–368 (2012).
E. O. Adamov, A. V. Dzhalavyan, and A. V. Lopatkin, “Conceptual provisions of the strategy for the development of future nuclear energy in Russia up to 2100,” ibid., 319–330.
K. Biss and B. Thomauske, “Shortening transmutation time by using the molten salt reactor,” Ann. Nucl. Energy, 83, 25–33 (2015).
L. I. Ponomarev, M. B. Seregin, A. P. Parshin, et al., “Choice of fuel salt for molten-salt reactor,” At. Energ., 115, No. 1, 6–11 (2013).
C. Bahri, W. Al-Aregi, M. Ruf, and A. Majid, “Characteristics of molten fl uoride salt system LiF–BeF2(FLiBe) and LiF–NaF–KF (FLiNaK) as coolant and fuel carrier in molten salt reactor (MSR),” AIP Conf. Proc. (2017), p. 1799.
L. I. Ponomarev, M. B. Seregin, A. A. Mikhalichenko, et al., “Substantiation of the choice of actinide fluoride imitators for investigating solubility in the fuel salt of molten-salt reactors,” At. Energ., 112, No. 6, 341–345 (2012).
A. A. Lizin, S. V. Tomilin, O. E. Gnevashov, et al., “Solubility of PuF3, AmF3, CeF3, NdF3 in LiF–NaF–KF melt,” At. Energ., 115, No. 1, 11–16 (2013).
P. N. Mushnikov and M. N. Gordeev, “Investigation of the solubility of CeF3 and NdF3 in salt melt LiF–NaF–KF,” 23rd Sci. Techn. Conf. NIO-5, VNIITF, Snezhinsk, March 27–29, 2012, pp. 322–325.
M. V. Volozhin, R. Ya. Zakirov, P. N. Mushnikov, et al., “Solubility of UF6, ThF4, and CeF3 LiF–NaF–KF melt,” At. Energ., 115, No. 1, 17–19 (2013).
M. B. Seregin, A. P. Parshin, A. Yu. Kuznetsov, “Solubility of UF4, ThF4, and CeF3 in LiF–NaF–KF melt,” Radiokhimiya, 53, No. 5, 416–418 (2011).
A. A. Lizin, S. V. Tomilin, O. E. Gnevashov, et al., “Solubility of UF4, ThF4 in LiF–NaF–KF mel,” At. Energ., 115, No. 1, 20–22 (2013).
P. N. Mushnikov, O. Yu. Tkacheva, V. I. et al., “Investigation of the quasi-binary phase diagram FLiNaK–NdF3,” Materials (Basel), 14, No. 21, 6428 (2021).
O. Beneš and R. Konings, “Actinide burner fuel: potential compositions based on the thermodynamics evaluation of M–PuF3 (M = Li, Na, K, Rb, Cs) and LaF3–PuF3 systems,” J. Nucl. Mater., 377, No. 3, 449–457 (2008).
G. Zong, Z. Zhang, J. Sun, and J. Xiao, “Preparation of high-purity molten FLiNaK salt by the hydrofl uorination process,” J. Fluor. Chem., 197, 134–141 (2017).
A. P. Apisarov, A. E. Dedyukhin, E. V. Nikolaeva, et al., “Liquidus temperatures of cryolite melts with low cryolite ratio,” Metall. Mater. Trans. B, 42, No. 1, 236–242 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Atomnaya Énergiya, Vol. 131, No. 5, pp. 260–264, November, 2021.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mushnikov, P.N., Tkacheva, O.Y., Kholkina, A.S. et al. Phase Diagram of the Quasibinary System LiF–NaF–KF–CeF3. At Energy 131, 263–267 (2022). https://doi.org/10.1007/s10512-022-00876-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10512-022-00876-2