Abstract
This study provides comprehensive results on the growth, thyroid, liver, and gonadal condition, maturity, and sex ratio in red tilapia exposed to exogenous thyroxine and goitrogen. One-gram red tilapia fingerlings were reared in three groups: control, thyroxine (T4) (10 nM), and goitrogen (sodium perchlorate, 30 mg/L) for 30 days, followed by 30 days without treatment. After the first 30 days of treatment, liver enzymes and whole-body T4 and T3 were assessed. The gonadosomatic index (GSI), maturity stages and sex ratio were examined. Final body weight (FBW), weight gain (WG), specific growth rate (SGR), and survival rate were also measured. Results revealed insignificant differences among groups in FBW, WG, SGR, and GSI, but the hepatosomatic index (HSI) of the goitrogen group significantly increased. Exogenous thyroxine significantly increased whole-body T4. Liver enzymes were not affected. Thyroid follicles of the thyroxine-treated fish showed large thyroid follicles, whereas the goitrogen-treated fish showed degenerated and negligible thyroid follicles. Liver tissue of the thyroxine-treated fish group displayed a few fat vacuoles, while perchlorate-treated fish showed hydrophobic degenerated hepatocytes. At the end of 60 days, the thyroxine treatment significantly increased FBW, WG, SGR, female GSI, and male hepatosomatic index (HSI). Perchlorate and thyroxine exposure, respectively, skewed sex ratios toward males and females. Exogenous T4 enhanced the maturity stages of both females and males, while perchlorate exposure delayed their gonadal ripening. These results validated the significant role of T4 during early life of red tilapia as it could improve the growth and maturity, and affect its sex ratio, while T4 insufficiency could alter these factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tilapia is one of the chief fish species of warm water designed for aquaculture production (Charo-Karisa et al. 2006). The commercial production of tilapia has gained increasing expansion in many countries worldwide because it is able to change the conditions in pond farming, resistance to diseases, and great survival and growth rates (Onumah et al. 2010). Red tilapia is well reported as a hybrid fish species, which has resulted from a continuous selective breeding procedure. Malaysian red tilapia is thought to be resulting from the cross-breeding among male Nile tilapia (Oreochromis niloticus) and female Mozambique tilapia (Oreochromis mossambicus); nevertheless, the species genetic background is not well identified (Pongthana et al. 2010).
Early sexual maturity and undesirable reproduction of tilapia species is a well-documented problem. The environmental factors, age, and size are the major causes for tilapia sexual maturity (Vajargah 2021). Moreover, sexual maturity is achieved at smaller length and younger ages for females compared to males (Shalloof and Salama 2008; Khallaf et al. 2020). Tilapia in captivity reached sexual maturity in smaller size comparing to the wild (Suresh and Bhujel 2012). Also, tilapia that live in larger populations matured at larger sizes and older ages than smaller communities (Vajargah 2021). Female O. niloticus under captivity displayed maturity at a relatively smaller size of 8–14 cm (Suresh and Bhujel 2012; Shalloof and Salama 2008; Khallaf et al. 2020). For O. mossambicus, sexual maturity was reported to be ranged 6–28 cm (Fishbase Froese and Pauly 2023). The histology of gonads for hybrid red tilapia showed four developmental stages during the growth: immature, initial maturing, maturing, and partially spent (Chellappa et al. 2012).
Thyroid hormones (THs) have a marked influence on numerous processes that control growth, development, differentiation, body metabolism, and in governing fish osmoregulation activity (Blanton and Specker 2007; McMenamin and Parichy 2013; Peyghan et al. 2013; Deal and Volkoff 2020). During fish larval–juvenile transformation, several morphological, physiological, and ecological changes are controlled by THs (McMenamin and Parichy 2013), including participation in fish gastrointestinal progress as in summer flounder (Paralichthys dentatus) (Huang et al. 1998) and zebrafish, Danio rerio (Liu and Chan 2002), and larval growth initiation as in Solea solea (Ismail 2018). Maternally supplied THs play an essential role in larval growth earlier to the active embryonic thyroid function (Yamano 2005; Einarsdottir et al. 2006). The inoculation of Nile tilapia brooder with T4 indirectly improved larval growth, as an obvious rise in length, weight, and survival rate of larvae was reported after female brooder were injected (Khalil et al. 2011). Furthermore, T4 maternal injection upsurges the embryonic growth, development, and survival rate of sterlet sturgeon, Acipenser ruthenus, offspring (Abdollahpour et al. 2018). However, THs possibly will have no impact or adverse effects on embryo-larval growth and development, as explained in several of studies (Yamano 2005; Sharma and Patiño 2013; Brown et al. 2014).
Thyroid hormones have been shown to sustain reproductive progress, whereas THs have been displayed to stimulate all reproductive stages, including the early development of gonads, vitellogenesis, and maturation crucial to successful ovulation and spawning (Cyr and Eales 1988). T4 injection improved the spawning efficiency and reproductive success in sterlet sturgeon; the injection with 0.1 μg/BW T4 triggered the peak of hatching and increased egg diameter (Abdollahpour et al. 2018). In broodstock mud crab, Scylla serrata, the observed association between the thyroid and reproduction demonstrated a possible co-evolution of these two systems (Tovo-Neto et al. 2018). T4 supplementation displayed a significant effect in the acceleration of the mature ovarian (Iromo et al. 2015). In male fish, T3 administration increased the production of Sertoli cells, which induce spermatogenesis, and T3 activates androgen production through gonadotropin induction (Tovo-Neto et al. 2018).
The majority of teleosts do not display an organized solid thyroid gland; however, they have detached thyroid follicles initiated separately or in bands all over the basibranchial region (Hsu et al. 2014; Chalde and Miranda 2017). During the larval stages, some small follicles producing thyroid hormone were noticed and then the follicles grew progressively in size and number (Yamano 2005). The thyroid follicles are spherical arrangements made by epithelial cells with a lumen enclosed; the lumen is occupied by a colloid and works as a reservoir of thyroid hormone creation materials, precursors, and the produced hormone (Carr and Patiño 2011).
Perchlorate is one of the goitrogens that are commonly used to control the thyroid state by TH synthesis inhibition and is used as an experimental antithyroid action causing iodide uptake into the thyroid follicle blocking (Carr and Norris 2006; Mukhi et al. 2005; Schmidt et al. 2012). Thyroxine production suppression using perchlorate affects primordial sexual differentiation and germ cell development, causing masculinization in adult threespine stickleback (Petersen et al. 2016). Moreover, perchlorate exposure delayed male and female gonadal maturity, and shifted sex ratios toward males in threespine stickleback (Furin et al. 2015). Perchlorate exposure also affects epithelial cells of thyroid follicles by inducing hypertrophy, and higher doses cause colloid depletion and angiogenesis (Liu et al. 2006; Ismail 2018).
The relationship between THs and liver tasks has been reported in vertebrates; THs regulate liver metabolism, additionally lipid metabolism, and the activity of iodothyronine deiodinase (Malik and Hodgson 2002; Blanton and Specker 2007; Carr and Patiño 2011). Whereas the hepatic deiodinases contribute to the T3 synthesis regulation and metabolism, also it keeps the TH stability (Brown et al. 2014; Deal and Volkoff 2020). Thyroid dysfunction might disturb the liver occupation, and liver diseases affect the metabolism of THs (Malik and Hodgson 2002). Sodium perchlorate induced liver steatosis in the molly fish, Poecilia sphenops (Burcu et al. 2009), while T4 exposure induced steatosis in S. solea hepatocytes and sodium perchlorate prompted vacuolated liver tissue and deprivation of some hepatocytes (Ismail 2018). The liver of tilapia treated with T3 displayed down-regulation of numerous pathways associated with the metabolism of carbohydrates (Olvera et al. 2017).
Therefore, this work aimed to identify the significance of thyroid hormones on growth performance, thyroid follicle structure, liver development, and sexual maturity during the period of red tilapia gonadal progress. To accomplish this task, exogenous thyroid hormone (T4) and the thyroid activity disorder agent goitrogen (sodium perchlorate) were applied to the rearing water of tilapia fingerlings for 30 days of exposure. By the end of the exposure time, growth, thyroid follicle state, T4 and T4 whole-body content, and liver activity (histology and liver enzymes aspartate aminotransferase (AST), alanine transaminase (ALT), and alkaline phosphatase (ALP)) were evaluated. Moreover, the exposure time was followed by another 30 days of rearing to determine the effect of thyroid state on reproductive performance by determining the gonadosomatic index (GSI), sex ratio, and maturity advancement.
Materials and methods
Broodstock spawning
A total of 30 broodstock Florida red tilapia fish were obtained from El-Kilo-21 hatchery in west Alexandria and retained at the marine hatchery of the National Institute of Oceanography and Fisheries. Fish were kept in fiberglass tanks (2000 L) with flow-through seawater (salinity was 37 ± 2 ppt) for 3 days, then salinity gradually declined to reach 20 ppt. The water temperature was 26 ± 1 °C, with a stimulated natural photoperiod, and fish were fed an artificial pelleted diet with 35% crude protein. Fish brooder were carefully chosen with an average total weight of 70–220 g. The sex ratio was two females to one male. The hatching occurred 3 days after spawning, and the average weight of newly hatched larvae was 0.0055 g.
Experimental design
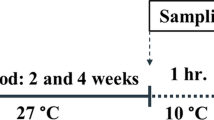
Red tilapia immature fingerlings at age 40 days after hatching were moved to glass aquaria to apply the treatments. The experiment was carried out in 9 glass aquaria containing 15 L of brackish water (20 ppt) and temperature of 26 ± 1 °C. Each glass aquarium was stocked with 50 fingerlings with an initial body weight of 1 ± 0.12 g and 1.77 ± 0.19 cm total length. Three treatment groups in triplicate were performed as follows: the first group with no chemical exposure was control, the second group was exposed to 10 nM/L thyroxine (T4), and the third group was exposed to 30 mg/L sodium perchlorate. Three replicates were applied for each treatment. The exposure time was 30 days followed by another 30 days without any exposure. By the end of the first 30 days, samples were collected (20 fish each aquarium) and weighed, total length measured, and stored at − 20 °C for whole-body T3 and T4 analysis and liver enzymes. Samples were also kept in 4% formal saline for thyroid follicle histology. By the end of the 60-day experimental time, fish samples were collected and sacrificed by extra-dose of MS222 anesthetic and weighed, length was measured, sex determined, GSI calculated, maturity evaluated, and gonads kept in formal saline.
Chemicals
The thyroxine (T4) used was under the commercial name Eltroxine (Aspen bad Oldesloe, GmbH Germany) in tablet form having 100 µg thyroxine each, which is equal to sodium thyroxine anhydrous salt 112 µg. The thyroxine exposure dose was selected as stated by Sharma and Patiño (2013). Sodium perchlorate monohydrate (NaClO4·H2O) (JPH7630; Wako, Japan) dose was selected according to Furin et al. (2015).
Enzyme extraction and measurement
After 30 days of exposure time, a random sample of liver was collected from fish to measure liver enzymes and then kept at − 20 °C until extraction. The enzymes aspartate aminotransferase (AST) (IU/L) and alanine aminotransferase (ALT) (IU/L) were analyzed along with Reitman and Frankel (1957), and the alkaline phosphatase (ALP) (IU/L) was performed according to Belfield and Goldberg (1971).
Thyroid hormone extraction and measurement
Frozen fish samples of 1 g were homogenized in 1 mL of saline solution to prepare sample lysate. After that, T3 and T4 hormones were measured according to Cao et al. (2016). T3 and T4 were evaluated using the T3 ELISA kit (EIAab, NO E0453f, USA) and T4 ELISA kit (Biosource, NO MBS701162, USA), respectively, following the enclosed protocol.
Growth performance
Measured performance parameters included final body weight (FBW), weight gain (WG), specific growth rate (SGR%), survival rate (%), and total length (TL). The average BW (mg) of fish in each experimental group was dignified to the closest 0.01 mg on a microbalance. Weight gain (WG) was estimated from the following equation: WG (mg) = Wf − Wi, where Wf is the final body weight and Wi is the initial body weight.
The specific growth rate (SGR% day−1) was considered following the relation SGRw% day−1 = 100 × (ln Wf − lnWi)/t, where ln is the natural logarithm and t is the time by number of days, SGRw is the specific growth rate by weight, Wf is the final body weight (mg), and Wi is the initial body weight (mg) (Mourad et al. 2022).
GSI was estimated from the subsequent equation:
HSI was estimated from the following equation:
Histological examination
For the thyroid follicle examination, after 30 days of exposure time the fish heads were dissected and stored in formal saline (4%). Before starting tissue processing, samples were kept in EDTA for a week for declassification. Liver samples were also removed and stored in formal saline (4%) after 30 days of exposure. By the end of 60 days of experimental time, gonads were dissected from each fish to small pieces and then preserved in formal saline (4%). All samples were then washed in 70% ethyl alcohol, dehydrated by passing through an ethanol sequence, cleared and embedded in paraffin wax according to Assem et al. (2019), and sectioned for about 6 to 9 μm for gonads, 3 μm for liver samples, and 5 μm for frontal sections of the head region. The sections were stained with hematoxylin and eosin. Sections were microscopically inspected by a compound microscope and microphotographs were taken with a Leica digital camera.
Statistical analysis
Data are stated as mean ± standard error. Significant differences between various treatment groups and the control were verified via one-way analysis of variance. The significant changes probability was set at p < 0.05. The analysis was accomplished using the SPSS version 22.0 package (SPSS 1998).
Results
Growth performance after 30 days of treatments
The present results of growth (FBW, WG, SGR, and final total length) (Table 1) demonstrated insignificant variations for the thyroxine- and perchlorate-exposed groups compared to the results documented in the control fish group (p < 0.05), as presented in Table 1.
Gonadosomatic index after 30 days of treatments
The gonadal-gutted weight relationship, expressed as a percentage, was determined as the gonadosomatic index (GSI%), with insignificant differences for the thyroxine- and perchlorate-exposed groups compared to the results documented in the control fish group (p < 0.05), as illustrated in Fig. 1A.
Hepatosomatic index after 30 days of treatments
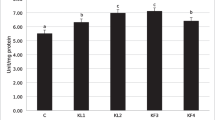
Figure 1B shows that the HSI% of sodium perchlorate group significantly increased to reach 3.59 ± 0.21 (p < 0.05). However, there were insignificant variations (p < 0.05) between the HSI% of the control and thyroxine groups (3.25 ± 0.33, 2.66 ± 0.1), respectively.
Thyroid hormone content (T4 and T3)
By the end of 30 days of exposure, the T3 hormone value of red tilapia displayed that sodium perchlorate group was significantly increased (p < 0.05) relative to thyroxine group (2.3 ± 0.29 and 1.4 ± 0.15, respectively), while insignificant variations were detected between perchlorate group and control group as well as thyroxine group and control group (Fig. 2A). The T4 whole-body content of the thyroxine-treated group significantly increased (p < 0.05) to reach 37.1 ± 9.02. However, there were insignificant differences between T4 content of the control and sodium perchlorate groups (Fig. 2B).
Liver enzymes
After 30 days of exposure, thyroxine-exposed group showed significantly decreased AST value (p < 0.05) relative to the control group, while insignificant variations were noticed among the control group and perchlorate group also for the thyroxine group and sodium perchlorate group, as shown in Fig. 3. There were no significant variations for ALT and ALP enzymes between the three experimental groups after 30 days of exposure, as illustrated in Fig. 3.
Thyroid follicle and liver histomorphology
After 30 days of exposure, the control group displayed numerous thyroid follicles of circular or oval shape with medium size (Fig. 4A). The follicles were lined with squamous epithelium cells (thyrocytes) (Fig. 4B). The thyroid follicles of the thyroxine-exposed group had many oval thyroid follicles with increasing size of some follicles comparing to the control, and thyrocytes showed normal size as control (Fig. 4C, D). The thyroid follicles of fish subjected to 30 mg/L sodium perchlorate had often irregular and difficult-to-define thyroid follicles of many small sizes, with the utmost thickening of the thyrocytes (hypertrophy) (Fig. 4E) and a mild colloid diminishing associated with follicular breakdown and large vacuoles were detected (Fig. 4F).
Photomicrograph of thyroid follicles and liver tissue of red tilapia after 30 days of treatment. A, B Control untreated group showing thyroid follicle (F), thyrocyte (T), colloid (c). C Thyroid follicles of thyroxine-treated fish group with enlarged thyroid follicles (*). D Thyroxine-treated fish group with magnification of large thyroid follicles. E, F 30 mg/L perchlorate-treated fish group with degenerated thyroid follicles (d), large vacuoles (V), very small thyroid follicles (red arrow), and hypertrophic thyrocytes (arrowheads). G Control untreated fish liver tissue showing normal hepatocytes (HC). H Liver of thyroxine-treated fish group, tissue with moderate vacuoles (V). I Liver tissue of 30 mg/L perchlorate-treated fish group with hydrophobic degenerated hepatocytes (Hd) and congestion of central veins (CV), H&E
We observed that there were no significant changes between the control liver tissue and thyroxine-exposed fish liver tissue, except the liver tissue of the thyroxine-exposed group showing normal hepatocytes with a few fat vacuoles (Fig. 4H). The sodium perchlorate group had hydrophobic degenerated hepatocytes and congestion of the central veins (Fig. 4I).
Growth performance after 60 days
After 60 days of rearing, the FBW of red tilapia in the thyroxine group significantly increased to reach 9.28 ± 0.80 g (p < 0.05). However, there were insignificant changes between the FBW between the control and sodium perchlorate groups (7.72 ± 0.32 and 7.44 ± 0.36 g, respectively). Also, the WG of red tilapia in the thyroxine group significantly increased to reach 8.28 ± 0.80 g (p < 0.05). However, there were insignificant changes between the WG of the control and sodium perchlorate groups (6.72 ± 0.32 and 6.44 ± 0.36 g, respectively). SGRW% day−1 of red tilapia in the thyroxine group significantly increased to reach 3.61 ± 0.14 (p < 0.05). Insignificant variations were identified between the SGRW% day−1 of the control and sodium perchlorate groups (3.34 ± 0.07 and 3.29 ± 0.08, respectively). There were insignificant alterations (p < 0.05) in total length in the three groups: control, thyroxine, and sodium perchlorate (7.33 ± 0.1, 7.77 ± 0.21, and 7.33 ± 0.13 cm, respectively) and there were insignificant variations (p < 0.05) for survival rate in the three groups: control, thyroxine, and sodium perchlorate (69%, 67%, and 70%, respectively) as shown in Table 2.
Gonadosomatic index of male and female (%) after 60 days
At the end of the 60-day experiment, there were insignificant alterations (p < 0.05) in male GSI% of red tilapia in the three groups: control, thyroxine, and sodium perchlorate (0.15 ± 0.04, 0.26 ± 0.09, and 0.26 ± 0.06, respectively) as illustrated in Fig. 5A. The GSI% of female fish exposed to thyroxine was significantly greater than that of the perchlorate and control groups (p < 0.05) (2.19 ± 0.36, 1.38 ± 0.32, and 0.47 ± 0.07, respectively). The GSI% for the sodium perchlorate fish group was significantly less than the control and thyroxine groups (p < 0.05), as illustrated in Fig. 5B.
Male gonadosomatic indices (GSI%) (A), female gonadosomatic indices (GSI%) (B), hepatosomatic index (HSI%) (C), and sex ratio% (D) of red tilapia treated with 10 nM/L thyroxine and 30 mg/L sodium perchlorate at the end of the 60-day experiment. Mean ± SE, columns with unlike letters are significantly different
Hepatosomatic index of male and female after 60 days
After 60 experimental days, the HSI% male of the thyroxine group was significantly greater than that of the control and sodium perchlorate groups (p < 0.05) (6.03 ± 0.57, 4.30 ± 0.49, and 3.83 ± 0.22, respectively). However, insignificant differences (p < 0.05) were found between the male HSI% of the control and sodium perchlorate groups (Fig. 5C). There were insignificant differences in HSI% of female red tilapia in the three groups: control, thyroxine, and sodium perchlorate (4.36 ± 0.44, 4.3 ± 0.35, and 4.19 ± 0.22, respectively) (p < 0.05) (Fig. 5C).
Sex ratio (%) after 60 days
After 60 days of total experimental time, the male percentage of the sodium perchlorate group was higher than the control and thyroxine groups (46%, 42%, and 26%, respectively) and the thyroxine group was noted to have the lower percentage of males (Fig. 5D). The female sex ratio of the thyroxine-treatment group was greater than that of the control and sodium perchlorate groups (74%, 58%, and 45%, respectively), and perchlorate group recorded the lowest female ratio (Fig. 5D).
Maturity stages after 60 days
The maturity stages of male fish after 60 days of experimental time displayed that the perchlorate group had a higher percentage of mature stage than the control and thyroxine groups (73%, 70%, and 60%, respectively), while the thyroxine group displayed the highest percentage of nearly ripe stage (Fig. 6A). Ripe stage percentages were recorded in thyroxine and control groups representing 20% of each group, but the sodium perchlorate group did not display any ripe males (Fig. 6A). For female fish, the thyroxine group reported the lowest percentage of mature stage, whereas the control group had the highest percentage of mature females and the lowest number of nearly ripe females. The ripe stage percentages of 29% and 28.5% were recorded in the thyroxine and control groups, respectively, whereas it was not recorded for the sodium perchlorate group (Fig. 6B).
Gonadal histomorphology changes
The testicular tissue after 60 days of the experiment did not show noticeable changes due to thyroxine or perchlorate exposure (Fig. 7A–C). Testicular tissues of the control and treated groups showed a fully developed mature testis with nests of different spermatogenesis stages (Fig. 7A–C). However, the testicular tissues of perchlorate-exposed fish displayed less amount of spermatozoa comparing to those of control and thyroxine groups (Fig. 7C). The ovarian tissue of red tilapia after 30 days of experiment followed by another 30 days without any exposure showed that both control and thyroxine groups were in more advanced stages and had advanced stage of vitellogenesis and ripe oocytes (Fig. 7D, E).
Gonadal tissue of red tilapia after 30 days of experiment followed by another 30 days without any exposure. A Testis tissues of control group with fully developed mature testis with nests of spermatogonia (spg), primary spermatocytes (psc), secondary spermatocytes (ssc), spermatids (st), and filled with mature spermatozoa (Spz). B Testis tissues of 10 nM/L thyroxine-treated group with developed testes with different spermatogenesis stages served as control. C Testis tissues of 30 mg/L sodium perchlorate–treated group with different spermatogenesis stages and less amount of spermatozoa (*) from some lobules. D Ovarian tissues of control group with ripe stage. E Ovarian tissue of thyroxine group with ripe stage. F Ovarian tissue of perchlorate group with mature stage, cytoplasmic growth (cg), follicular epithelium (FE), primary oocyte (p) and advanced primary oocyte (pm), yolk granules (yg) and nucleus (N), ripe ova (*), and vacuoles (V). Hematoxylin–eosin stain
Ovarian sections of the control and thyroxine groups showed the advanced stage of vitellogenesis in the secondary and tertiary yolk stage, which grew in diameter. The oocyte wall consisted of a zona radiata and was enclosed with a follicular epithelial layer, and the ovaries also exhibited early stages of cytoplasmic growth (Fig. 7D, E).
The fish group exposed to perchlorate showed less advanced oocytes in which primary oocyte, advanced primary oocyte, and primary and secondary yolk stages were the main demonstrative oocyte stages (Fig. 7F).
Discussion and conclusion
In the present study, after 30 days of exposure, there were insignificant differences in growth performance (FBW, WG, SGR, and TL) of red tilapia in the three groups (control, thyroxine, and perchlorate). Consistent with our finding, Landines et al. (2010) showed that there was no expressive effect of T4 on growth, but it amended the survival of the piracanjuba progeny. However, Navarrete-Ramírez et al. (2011) reported that thyroxine directly or indirectly improved pike silverside (Chirostoma estor) larval growth, subsequently a marked rise in both length and weight of larvae because of thyroxine treatment. Moreover, the 30-day exposure to T4 or goitrogen displayed insignificant differences in GSI% of red tilapia. Moreover, exogenous T4 treatment can improve the growth performance of sterlet sturgeon Acipenser ruthenus (Abdollahpour et al. 2018). However, long-term exposure to T4 (up to 28 weeks) demonstrated a significant decline in GSI% in the dose-dependent mode (Ismail et al. 2017).
After 60 days of experiment, the growth performance (FBW, WG, and SGR) of red tilapia exposed to thyroxine for 30 days showed a significant increase comparing to the control. In agreement with our results, Khalil et al. (2011) showed that T4-treated Nile tilapia females for 35 days revealed significantly greater total body weight than those of the control. Moreover, exogenous T4 treatment can improve the growth performance of sterlet sturgeon (Abdollahpour et al. 2018). Conversely, there were insignificant modifications in TL and survival rate in the three groups. In agreement with our results, Navarrete-Ramírez et al. (2011) reported that T4 exposure of pike silverside juveniles did not affect fish survival at any tested dose. Thyroxine did not affect the survival rate of the pink zebra larvae Brachydanio reiro (Heraedi et al. 2018). However, the thyroxine injection of Nile tilapia brooder induced a significantly higher larval survival rate than the control (Khalil et al. 2011). In addition, T4 exposure can improve the survival rate of sterlet sturgeon (Abdollahpour et al. 2018).
In the present work, the influence of exogenous thyroxine or goitrogen on red tilapia liver was explained by significant elevation in HSI% of the perchlorate group; also the perchlorate-treatment group displayed hydrophobic degenerated hepatocytes and congestion of central veins. While liver enzymes did not show significant changes in ALT and ALP enzymes, the thyroxine group showed a significant decrease in AST value.
Our results displayed that the administration of exogenous thyroxine had no effect on the liver, whereas exogenous goitrogen induced alterations such as hydrophobic degeneration and enlarged the hepatocytes’ size. Contrasting to the current results, exogenous thyroxine induced lessening of lipids in the liver of Anabas testudineus (Varghese and Oommen 1999). Exogenous T4 improved lipid storage in hepatocytes in sole larvae (Ismail 2018). In comparison, Khalil et al. (2011) reported that for tilapia larvae attained from treated females with thyroxine after 4 dph, lipid storage was perceived in the liver corresponding with the start of feeding. Furthermore, alterations in the hepatocytes induced by administration of goitrogen perchlorate have been reported for some studied species, raising the ability of hepatocyte-accumulated lipid for zebrafish (Schmidt et al. 2012). In addition, liver histology displayed vacuoles among hepatocytes and deterioration in some cells, which can be a mark of stress or disturbance of hormones in Solea solea (Ismail 2018) and in rare minnow (Gobiocypris rarus) (Zhu et al. 2014).
In this work, whole-body T3 hormone value after 30 days of experiment was significantly greater for the perchlorate group than the thyroxine group, while whole-body T4 hormone of the thyroxine group significantly increased. Ismail (2018) reported that whole-body T4 displayed a significant decline in fish subjected to 100 mg/L perchlorate. Insignificant variations in whole-body T3 content were noticed for sole larvae exposed to goitrogen or thyroxine (Ismail 2018). Higher doses of perchlorate might deteriorate the T4 levels, as reported for zebrafish when a significant decline was only observed for higher doses of goitrogen (Schmidt et al. 2012). However, goitrogens augmented the whole-body T4 concentration in larvae of fathead minnow Pimephales promelas (Crane et al. 2005).
In the present study, the thyroid follicles of the thyroxine group increased in the follicle with more colloid material, indicating more activity in TH synthesis. The thyroid follicles of the perchlorate group had small-sized follicles with many small hypertrophy thyrocytes. Consistent to our results, Solea larvae displayed a marked increase in colloid size after thyroxine exposure, and perchlorate-exposed Solea larvae showed a significant increase in thyroid follicle cell height (Ismail 2018). The colloid area was reported to be more sensitive during larval development (Furin et al. 2015). Thyroxine production inhibition by goitrogen perchlorate has been confirmed to disrupt the structure of thyroid follicles in several studied fish species as for the fathead minnow (Crane et al. 2005), threespine stickleback (Petersen et al. 2015), and zebrafish (Sharma et al. 2016). The thyroid follicular cells’ height and the follicular morphology are well thought out to be sensitive and appropriate biomarkers of thyroid disruption (Schmidt and Braunbeck 2011).
The effect of thyroxine exposure or diminishing of sexual maturity and reproductive performance was assessed in this study by the end of 60 days. In the present study, insignificant differences were acknowledged in male GSI% of red tilapia between the three groups, while the female GSI% of thyroxine group was significantly increased and the GSI% of perchlorate individuals was significantly lesser than the control (p < 0.05). Consistent with the present findings, the administration of T4 increased C. gariepinus female GSI% (Supriya et al 2005); also, T3 administration encouraged GSI% in male C. gariepinus fish (Lema et al. 2009). The suppression of T4 transfer into T3 encouraged GSI% reduction, while treatment with exogenous T3 caused GSI elevation in female rainbow trout (Cyr and Eales 1988). Contrary to our results, long-term T4 exposure (12–28 weeks) triggered a significant decrease in red tilapia female GSI% (Ismail et al. 2017).
In this study, the male percentage of the sodium perchlorate group was greater than that of the control and thyroxine groups. Perchlorate exposure skewed sex ratios toward males. On the other hand, the sex ratio female percentage of the thyroxine group exceeded that of the control and sodium perchlorate groups. Thyroxine exposure skewed sex ratios toward females. Consistent with our findings, the sex ratio was considerably biased toward males after exposing to perchlorate (Petersen et al. 2016; Furin et al. 2015). The environmental perchlorate acquaintance early in life might be interfering with reproductive progress during sexual differentiation in exposed vertebrates and perchlorate has effects on the progress of reproduction by considerably dropping the number of primordial germ cells in female stickleback (Petersen et al. 2016; Furin et al. 2015). Contrasting to our results, the zebrafish sex ratio was skewed toward ovaries in goitrogen-subjected fish and toward testes in fish subjected to thyroxine (Sharma and Patiño, 2013).
The gonadal maturity was also delayed after goitrogen exposure in the present findings; male and female ripe stage was not recorded in the perchlorate group, while T4 exposure induced the percentage of nearly ripe stage in males of the thyroxine group. Perchlorate exposure reduced the ratio of late stages of ovaries and testes (Petersen et al. 2016; Furin et al. 2015) and delayed maturity in stickleback (Petersen et al. 2015). There was a progressive association between THs and fish state of reproduction, where THs are commonly linked with the development of testes, growth, and maturation (Tovo-Neto et al. 2018). The supplementation of thyroxine (0.1 μg/BW) displayed a significant effect in the acceleration of ovarian maturation of mud crab females (Iromo et al. 2015). T3 upsurge the Sertoli cell capacity for production of Igf3, which is a boosting growth factor of spermatogenesis. Furthermore, T3 acts through Leydig cells to improve the induction of gonadotropin synthesis and discharge of androgens (Tovo-Neto et al. 2018).
In the current study, testicular tissue of the perchlorate group displayed fewer amounts of spermatozoa consisting with the ripe stage absence. Testis tissues of thyroxine-treated group with developed testes with different spermatogenesis stages served as control. Spermatogenesis was postponed in testes of goitrogen-treated zebrafish at 60 dpf related to control but was unchanged in testes of thyroxine-treated fish (Sharma and Patiño, 2013). Moreover, the administration of T3 in male C. gariepinus augmented the quantity of maturing spermatozoa (Lema et al. 2009). In female catfish, T4 administration boosted the ovarian follicular growth rate and the quantity of vitellogenic/postvitellogenic follicles (Supriya et al. 2005).
In conclusion, a 1-month thyroxine exposure during early life of red tilapia larvae significantly improved growth performance, the whole-body T4, and ovarian development and displayed larger thyroid follicles, whereas exogenous thyroxine induced a decline in liver enzyme AST without alterations in liver histomorphology. The use of goitrogen displayed alterations in the liver and thyroid follicle histology with no effect on the growth performance. Thyroxine exposure enhanced gonadal maturity, whereas thyroxine reduction by goitrogen exposure delayed gonadal maturity. The sex ratio was also affected by thyroxine upsurge or decline, exogenous T4 during the early life period skewed sex ratios toward females, whereas perchlorate exposure twisted sex ratios toward males.
Data availability
The data that support the findings of this study are available upon reasonable request.
References
Abdollahpour H, Falahatkar B, Efatpanah I, Meknatkhah B, Van Der Kraak G (2018) Influence of thyroxine on spawning performance and larval development of Sterlet sturgeon Acipenser ruthenus. Aquacul 497:134–139
Assem SS, Srour TM, Mourad MM, Mansour AT, Sharaf HR (2019) Oogenesis and spermatogenesis of the common sole, Solea solea, from the Mediterranean Sea, Abu Qir Bay, Alexandria, Egypt: biological, histological and ultrastructural approaches. Med Aquacul J 11:11–33
Belfield A, Goldberg DM (1971) Normal ranges and diagnostic value of serum 5′ nucleotidase and alkaline phosphatase activities in infancy. Arch Dis Child 46(250):842–846
Blanton ML, Specker JL (2007) The hypothalamic–pituitary–thyroid (HPT) axis in fish and its role in fish development and reproduction. Crit Rev Toxicol 37:97–115
Brown CL, Urbinati EC, Weimin Zhang W, Brown SB, Michelle M (2014) Maternal thyroid and glucocorticoid hormone interactions in larval fish development, and their applications in aquaculture. Rev Fish Sci Aquacult 22:207–220
Burcu KT, Sema IU, Ozlem O (2009) The effects of sodium perchlorate on the liver of Molly Fish (Poecilia sphenops, Cyprinidae, Teleostei). Afr J Biotechnol 8(11):2640–2644
Cao C, Wang Q, Jiao F, Zhu G (2016) Impact of co-exposure with butachlor and triadimefon on thyroid endocrine system in larval zebrafish. Exp Toxicol Pathol 68(8):463–469
Carr JA, Norris DO (2006) The thyroid gland. In: Norris DO, Carr JA (eds) Endocrine disruption: biological bases for health effects in wildlife and humans. Oxford University Press, New York, pp 87–110
Carr JA, Patiño R (2011) The hypothalamo-pituitary-thyroid axis in teleosts and amphibians: endocrine disruption and its consequences to natural populations. Gen Comp Endocrinol 170:299–312
Chalde T, Miranda LA (2017) Pituitary–thyroid axis development during the larval–juvenile transition in the pejerrey Odontesthes bonariensis. J Fish Biol 91:818–834
Charo-Karisa H, Komen H, Rezk M, Ponzoni RW, Van Arendonk JAM, Bovenhuis H (2006) Heritability estimates and response to selection for growth of Nile tilapia (Oreochromis niloticus) in low-input earthen ponds. Aquacul 261:479–486
Chellappa S, de Medeiros APT, Ribeiro Freire MDS, Volpato GL (2012) Dynamics of territorial behaviour and gonad development in the hybrid red tilapia, Oreochromis niloticus x O. mossambicus (Osteichthyes: Cichlidae). Animal Biology Journal 3:5–19
Crane HM, Pickford DB, Hutchinson TH, Brown JA (2005) Effects of ammonium perchlorate on thyroid function in developing fathead minnows Pimephales promelas. Environ Health Perspect 113(4):396–401
Cyr DG, Eales JG (1988) Influence of thyroidal status on ovarian function in rainbow trout. Salmo Gairdneri J Exp Zool 248(1):81–87
Cyr DG, Eales JG (1996) Interrelationships between thyroidal and reproductive endocrine systems in fish. Rev Fish Biol Fish 6:165–200
Deal CK, Volkoff H (2020) The role of the thyroid axis in fish. Front Endocrinol 11:596585
Einarsdottir IE, Silva N, Power DM, Smaradottir H, Bjornsson BT (2006) Thyroid and pituitary gland development from hatching through metamorphosis of a teleost flatfish, the Atlantic halibut. Anato Embryol 211:47–60
Fishbase Froese R, Pauly D (eds) (2023) FishBase. World Wide Web electronic publication. www.fishbase.org, ( 02/2023 )
Furin CG, von Hippel FA, Postlethwait JH, Buck CL, Cresko WA, O’Hara TM (2015) Developmental timing of sodium perchlorate exposure alters angiogenesis, thyroid follicle proliferation and sexual maturation in stickleback. Gen Comp Endocrinol 219:24–35
Heraedi A, Prayitno SP, Yuniarti T (2018) The effect of different thyroxine hormone (T4) concentration on the growth, survival, and pigment development of pink zebra fish larvae (Brachydanio reiro). Omni-Akuatika 14(2):21–28
Hsu C, Tsai S, Shen SC, Wu SM (2014) Profiles of thyrotropin, thyroid hormones, follicular cells and type I deiodinase gene expression during ontogenetic development of tilapia larvae and juveniles. Fish Physiol Biochem 40:1587–1599
Huang L, Schreiber AM, Soffientino B, Bengtson DA, Specker JL (1998) Metamorphosis of summer flounder (Paralichthys dentatus): thyroid status and the timing of gastric gland formation. J Exp Zool 280:413–420
Iromo H, Zairin M Jr, Agus MS, Manalu W (2015) Supplementation doses thyroxine hormone of broodstock mud crab (Scylla serrata) during ovarian maturation. J Aquacul Res Develop 6(12):1000379
Ismail RF (2018) Involvement of thyroid hormone in the intestine and liver development of the common sole (Solea solea L.) larvae. Egypt J Aquat Biol Fisher 22(2):77–91
Ismail RF, Mourad MM, Negm RM, Assem SS (2017) Effect of prolonged exposure to thyroxine on growth, puberty timing and ovarian structure in female red tilapia (Oreochromis sp.). Egypt J Aquat Res 43(4):313–320
Khalil NA, Khalaf HM, Mostafa AM (2011) The effect of maternal thyroxine injection on growth, survival and development of the digestive system of Nile tilapia, Oreochromis niloticus, larvae. Adv Biosci Biotechnol 109:47–58
Khallaf EA, Alne-na-ei AA, El-messady F, Hanafy EH (2020) Effect of climate change on growth and reproduction of Nile tilapia (Oreochromis niloticus, L.) from Bahr Shebeen Canal, Delta of Egypt. Egyptian J Aquatic Biol Fish 24:483–509
Landines MA, Sanabria AI, Senhorini JA, Urbinati EC (2010) The influence of triiodothyronine (T3) on the early development of piracanjuba (Brycon orbignyanus). Fish Physiol Biochem 36(4):1291–1296
Lema SC, Dickey JT, Schultz IR, Swanson P (2009) Thyroid hormone regulation of mRNAs encoding thyrotropin beta-subunit, glycoprotein alpha-subunit, and thyroid hormone receptors alpha and beta in brain, pituitary gland, liver, and gonads of an adult teleost, Pimephales promelas. Endocrinol 202:43–54
Liu YW, Chan WK (2002) Thyroid hormones are important for embryonic to larval transitory phase in zebrafish. Differ 70:36–45
Liu FJ, Wang JS, Theodorakis CW (2006) Thyrotoxicity of sodium arsenate, sodium perchlorate, and their mixture in zebrafish Danio rerio. Environ Sci Technol 40(10):3429–3436
Malik R, Hodgson H (2002) The relationship between the thyroid gland and the liver. QJM-Int J Med 95:559–569
McMenamin SK, Parichy DM (2013) Metamorphosis in teleosts. Curr Topics Dev Biol 103:127–165
Mourad MM, Assem SS, Srour TM, Sharaf HR, Mansour AT (2022) Compatible first feeding for larval common sole (Solea solea): effect of rotifer and artemia scheme manipulation on growth performance and survival. Exam Mar Biol Oceanogr 5(1):EIMBO 000604
Mukhi S, Carr JA, Anderson TA, Patino R (2005) Novel biomarkers of perchlorate exposure in zebrafish. Environ Toxicol Chem 24:1107–1115
Navarrete-Ramírez P, Orozco A, Valverde-R C, Olvera A, Toledo-Cuevas EM, Ross LG, Martínez-Palacios CA (2011) Effects of thyroxine administration on the growth and survival of pike silverside (Chirostoma estor) juveniles. Aquacul Res 42(6):808–814
Olvera A, Martyniuk CJ, Buisine N, Jiménez-Jacinto V, Sanchez-Flores A, Sachs LM (2017) Differential transcriptome regulation by 3, 5–T2 and 3’, 3,5–T3 in brain and liver uncovers novel roles for thyroid hormones in tilapia. Sci Rep 7(1):15043
Onumah EE, Wessels S, Wildenhayn N, Brummer B, Schwark GH (2010) Stocking density and photoperiod manipulation in relation to Estradiol profile to enhance spawning activity in female Nile Tilapia. Turk J Fish Aquat Sci 10:463–470
Petersen AM, Dillon D, Bernhardt RA, Torunsky R, Postlethwait JH, von Hippel FA, Buck CL, Cresko WA (2015) Perchlorate disrupts embryonic androgen synthesis and reproductive development in threespine stickleback without changing whole-body levels of thyroid hormone. Gen Comp Endocrinol 210:130–144
Petersen AM, Earp NC, Redmond ME, Postlethwait JH, von Hippel FA, Buck CL, Cresko WA (2016) Perchlorate exposure reduces primordial germ cell number in female three spine stickleback. PLoS ONE 11(7):e0157792
Peyghan R, Enayati A, Sabzevarizadeh M (2013) Effect of salinity level on TSH and thyroid hormones of grass carp. Ctenophayngodon Idella Vet Res Forum 4(3):175–178
Pongthana N, Nguyen NH, Ponzoni RW (2010) Comparative performance of four red tilapia strains and their crosses in fresh- and saline water environments. Aquacul 308:S109–S114
Reitman S, Frankel S (1957) A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Amer J Clinic Pathol 28(1):56–63
Schmidt F, Braunbeck T (2011) Alterations along the hypothalamic-pituitary-thyroid axis of the zebrafish (Danio rerio) after exposure to propylthiouracil. J Thyroid Res 2011:376243
Schmidt F, Schnurr S, Wolf R, Braunbeck T (2012) Effects of the anti-thyroidal compound potassium-perchlorate on the thyroid system of the zebrafish. Aquat Toxicol 109:47–58
Shalloof KAS, Salama HMM (2008) Investigations on some aspects of reproductive biology in Oreochromis niloticus (Linnaeus, 1757) inhabited Abu-zabal Lake. Egypt Glob Vet 2(6):351–359
Sharma P, Patiño R (2013) Regulation of gonadal sex ratios and pubertal development by the thyroid endocrine system in zebrafish (Danio rerio). Gen Comp Endocrinol 184:1–9
Sharma P, Grabowski T, Patino R (2016) Thyroid endocrine disruption and external body morphology of zebrafish. Gen Comp Endocrinol 226:42–49
Supriya A, Raghuveer K, Swapna I, Rasheeda MK, Kobayashi T, Nagahama Y, Gupta AD, Majumdar KC, Senthilkumaran B (2005) Thyroid hormone modulation of ovarian recrudescence of air-breathing catfish (Clarias gariepinus). J Fish Physiol Biochem 31:267–270
Suresh V, Bhujel RC (2012) Tilapias. In: Lucas JS, Southgate PC (eds) Aquaculture: farming aquatic animals and plants. Wiley-Blackwell Publishing Company, United Kingdom, pp 338–364
Tovo-Neto A, da Silva RM, Habibi HR, Nóbrega RH (2018) Thyroid hormone actions on male reproductive system of teleost fish. Gen Comp Endocrinol 265:230–236
Vajargah MF (2021) A review of the physiology and biology of Nile tilapia (Oreochromis niloticus). J Aquac Mar Biol 10(5):244–246
Varghese S, Oommen OV (1999) Thyroid hormones regulate lipid metabolism in a teleost, Anabas testudineus (Bloch). Comp Biochem Physiol 124B:445–450
Yamano K (2005) The role of thyroid hormone in fish development with reference to aquaculture. Jpn Agric Res q 39(3):161–168
Zhu L, Li W, Zha J, Wang M, Yuan L, Wang Z (2014) Butachlor causes disruption of HPG and HPT axes in adult female rare minnow (Gobiocypris rarus). Chem Biol Interact 221:119–126
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
Conception and design of the manuscript: R.F.I., S.S.A., H.E.R.S., T.M.A.S. Data source: H.E.R.S. Analysis and interpretation of the data: R.F.I., H.E.R.S. Drafting the article: R.F.I., S.S.A., H.E.R.S. Revising the manuscript content: R.F.I., S.S.A., H.E.R.S., A.A.Z., T.M.A.S. Approval of the submitted manuscript: R.F.I., S.S.A., H.E.R.S., A.A.Z., T.M.A.S.
Corresponding author
Ethics declarations
Consent for publication
This manuscript has not been submitted to, nor is under review at, another journal or other publishing venue. The five authors agree to the submission to Aquaculture International and to nominate R.F.I. as corresponding author.
Ethics approval
The experimental setup and fish handling procedure were approved by the Research Ethical Committee of the National Institute of Oceanography and Fisheries, NIOF, Cairo, Egypt.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Brian Austin
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ismail, R.F., Assem, S.S., Sharaf, H.E.R. et al. The effect of thyroxine (T4) and goitrogen on growth, liver, thyroid, and gonadal development of red tilapia (O. mossambicus × O. urolepis hornorum). Aquacult Int 32, 1151–1168 (2024). https://doi.org/10.1007/s10499-023-01210-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01210-8