Abstract
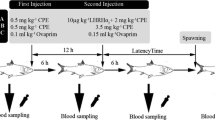
The tambaqui (Colossoma macropomum) is the main native species reared in aquaculture in Brazil and in its neighboring countries. Currently, the production of fries is based on the use of carp pituitary extract (CPE), which has intrinsic disadvantages such as impurity, the presence of unknown components, and discontinuity of supply. Three GnRH analogs (GnRHa) were tested to stimulate artificial reproduction in tambaqui. A total of 28 mature females were used in the following treatments: T1: synthetic salmon GnRHa—5.0 μg/kg combined with domperidone (2.5 mg/kg); T2: gonadorelin—60 μg/kg; T3: buserelin acetate—0.7 μg/kg; and T4: CPE—5.5 mg/kg. Spawning success under strip spawning, quantity and quality of the eggs were evaluated. The plasma concentrations of 17β-estradiol, 17α-hydroxyprogesterone, and 17α,20β-dihydroxy-4-pregnen-3-one were assessed before hormonal treatment and after stripping. All CPE-treated females released eggs via stripping, as occurred in 86% of the females treated with sGnRHa, 86% released eggs with gonadorelin, and 57% released eggs with buserelin. Degree-hour (DH) for strip spawning was higher in the GnRHa groups than for CPE (p ≤ 0.001). Total mass of the eggs, number of eggs/g of spawn, relative fecundity, fertilization index, and the rate of embryo survival were similar in all groups. There was a significant increase in steroid levels in all groups after receiving the hormones, including in the fish that did not release eggs under stripping. Although the commercial GnRHa did not stimulate the final maturation and/or ovulation in all females, they induced the ovulation of eggs in equal quality and quantity to the CPE, thus confirming the great potential of such drugs in the artificial reproduction of native neotropical fish.




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Acuña JJA, Rangel JLH (2009) Effects of hypophysial extract of common carp and the analog of the GnRH on the final maturation oocyte and the spawning of cachama negra (Colossoma macropomum). Rev Cient la Fac Ciencias Vet la Univ del Zulia
Aizen J, Hollander-Cohen L, Shpilman M, Levavi-Sivan B (2017) Biologically active recombinant carp LH as a spawning-inducing agent for carp. J Endocrinol 232:391–402. https://doi.org/10.1530/JOE-16-0435
Almeida FL, Lopes JS, Crescencio R et al (2016) Early puberty of farmed tambaqui (Colossoma macropomum): possible influence of male sexual maturation on harvest weight. Aquaculture 452:224–232. https://doi.org/10.1016/j.aquaculture.2015.10.031
Andrade ES, Carvalho AFS, Ferreira MR et al (2014) Indutores hormonais na reprodução artificial de curimba (Prochilodus lineatus). Rev Bras Reprodução Anim 38:230–236
Carolsfeld J, Ramos SM, Ormanezi R et al (1988) Analysis of protocols for application of an LHRH analog for induced final maturation and ovulation of female pacu (Piaractus mesopotamicus Holmberg 1887). Aquaculture 74:49–55. https://doi.org/10.1016/0044-8486(88)90085-3
Chaube R, Singh RK, Joy KP (2014) Effects of ovaprim, a commercial spawning inducer, on vasotocin and steroid hormone profiles in the catfish Heteropneustes fossilis: In vivo and in vitro studies. Gen Comp Endocrinol 195:190–200. https://doi.org/10.1016/j.ygcen.2013.11.009
Donaldson EM, Hunter GA (1983) Induced final maturation, ovulation, and spermiation in cultured fish. Fish Physiol 9:351–403. https://doi.org/10.1016/S1546-5098(08)60307-6
Drori S, Ofir M, Levavi-Sivan B, Yaron Z (1994) Spawning induction in common carp (Cyprinus carpio) using pituitary extract or GnRH superactive analogue combined with metoclopramide: analysis of hormone profile, progress of oocyte maturation and dependence on temperature. Aquaculture 119:393–407. https://doi.org/10.1016/0044-8486(94)90303-4
Dufour S, Sebert ME, Weltzien FA, Rousseau K, Pasqualini C (2010) Neuroendocrine control by dopamine of teleost reproduction. J Fish Biol 76:129–160. https://doi.org/10.1111/j.1095-8649.2009.02499.x
Fakriadis I, Miccoli A, Karapanagiotis S et al (2020) Optimization of a GnRHa treatment for spawning commercially reared greater amberjack Seriola dumerili: dose response and extent of the reproductive season. Aquaculture 521. https://doi.org/10.1016/j.aquaculture.2020.735011
Felizardo VO, Murgas LDS, Andrade ES et al (2012) Effect of timing of hormonal induction on reproductive activity in lambari (Astyanax bimaculatus). Theriogenology 77:1570–1574. https://doi.org/10.1016/j.theriogenology.2011.11.025
Fernandez-Palacios H, Schuchardt D, Roo J et al (2014) Dose-dependent effect of a single GnRHa injection on the spawning of meagre (Argyrosomus regius) broodstock reared in captivity. Spanish J Agric Res 12:1038–1048. https://doi.org/10.5424/sjar/2014124-6276
Gardes L (2000) Induced spawning of red drum, Sciaenops ocellatus: use of multivariate and univariate analysis methods in the search for side effects of LH-RHa treatments and ovarian development state upon spawn quality. Aquat Living Resour 13:19–27. https://doi.org/10.1016/S0990-7440(00)00137-6
Hilsdorf AWS, Hallerman E, Valladão GMR et al (2022) The farming and husbandry of Colossoma macropomum: from Amazonian waters to sustainable production. Rev Aquac 14:993–1027. https://doi.org/10.1111/raq.12638
Ibarra-Castro L, Duncan NJ (2007) GnRHa-induced spawning of wild-caught spotted rose snapper Lutjanus guttatus. Aquaculture 272:737–746. https://doi.org/10.1016/j.aquaculture.2007.09.007
Kobayashi M, Aida K, Hanyu I (1987) Hormone changes during ovulation and effects of steroid hormones on plasma gonadotropin levels and ovulation in goldfish. Gen Comp Endocrinol 67:24–32. https://doi.org/10.1016/0016-6480(87)90201-2
Konzen-Freitas AR, de AJG, de AJS et al (2020) Tambaqui females (Colossoma macropomum) spawn after hormonal induction with buserelin acetate. Anim Reprod Sci 221:106594. https://doi.org/10.1016/j.anireprosci.2020.106594
Le Cren ED (1951) The length-weight relationship and seasonal cycle in gonad weight and condition in the perch (Perca fluviatilis). J Anim Ecol 20:201–219. https://doi.org/10.2307/1540
Levavi-Sivan B, Bogerd J, Mañanós EL et al (2010) Perspectives on fish gonadotropins and their receptors. Gen Comp Endocrinol 165:412–437. https://doi.org/10.1016/j.ygcen.2009.07.019
Levavi-Sivan B, Safarian H, Rosenfeld H et al (2004) Regulation of gonadotropin-releasing hormone (GnRH)-receptor gene expression in tilapia: effect of GnRH and dopamine. Biol Reprod 70:1545–1551. https://doi.org/10.1095/biolreprod.103.021998
Levavi-Zermonsky B, Yaron Z (1986) Changes in gonadotropin and ovarian steroids associated with oocytes maturation during spawning induction in the carp. Gen Comp Endocrinol 62. https://doi.org/10.1016/0016-6480(86)90097-3
Lubzens E, Young G, Bobe J, Cerdà J (2010) Oogenesis in teleosts: how fish eggs are formed. Gen Comp Endocrinol 165:367–389. https://doi.org/10.1016/j.ygcen.2009.05.022
Mañanós E (2002) Luteinizing hormone and sexual steroid plasma levels after treatment of European sea bass with sustained-release delivery systems for gonadotropin-releasing hormone analogue. J Fish Biol 60:328–339. https://doi.org/10.1006/jfbi.2001.1839
Miura C, Higashino T, Miura T (2007) A progestin and an estrogen regulate early stages of oogenesis in fish. Biol Reprod 77:822–828. https://doi.org/10.1095/biolreprod.107.061408
Muniz JASM, Catanho MTJA, Santos AJG (2008) Influência do fotoperíodo natural na reprodução induzida do Tambaqui Colossoma macropomum (CUVIER , 1818). Bol do Inst Pesca 34:205–211
Muñoz-Cueto JA, Zmora N, Paullada-Salmerón JA et al (2020) The gonadotropin-releasing hormones: lessons from fish. Gen Comp Endocrinol 291:113422. https://doi.org/10.1016/j.ygcen.2020.113422
Mylonas CC, Fostier A, Zanuy S (2010) Broodstock management and hormonal manipulations of fish reproduction. Gen Comp Endocrinol 165:516–534. https://doi.org/10.1016/j.ygcen.2009.03.007
Mylonas CC, Zohar Y (2001) Endocrine regulation and artificial induction of oocyte maturation and spermiation in basses of the genus Morone. Aquaculture 202:205–220. https://doi.org/10.1016/S0044-8486(01)00772-4
Nagahama Y (1997) 17α,20β-Dihydroxy-4-pregnen-3-one, a maturation-inducing hormone in fish oocytes: mechanisms of synthesis and action. Steroids 62:190–196. https://doi.org/10.1016/S0039-128X(96)00180-8
Nagahama Y, Yamashita M (2008) Regulation of oocyte maturation in fish. Dev Growth Differ 50:S195–S219. https://doi.org/10.1111/j.1440-169X.2008.01019.x
Ngamvongchon S, Pawaputanon O, Leelapatra W, Johnson WE (1988) Effectiveness of an LHRH analogue for the induced spawning of carp and catfish in Northeast Thailand. Aquaculture 74:35–40. https://doi.org/10.1016/0044-8486(88)90083-X
Nyuji M, Yamamoto I, Hamada K et al (2019) Effect of GnRHa on plasma levels of Fsh and Lh in the female greater amberjack Seriola dumerili. J Fish Biol 95:1350–1354. https://doi.org/10.1111/jfb.14137
Peng C, Gallin W, Blomqvist AG et al (1994) Gene expression in the Goldfish brain : and regulation by ovarian steroids. Endocrinology 134:1095–1113
Pereira TSB, Boscolo CNP, Moreira RG, Batlouni SR (2016) The use of mGnRHa provokes ovulation but not viable embryos in Leporinus macrocephalus. Aquac Int 25:515–529. https://doi.org/10.1007/s10499-016-0049-2
Pereira TSB, Boscolo CNP, Moreira RG, Batlouni SR (2018) Leporinus elongatus induced spawning using carp pituitary extract or mammalian GnRH analogue combined with dopamine receptor antagonists. Anim Reprod 15:64–70. https://doi.org/10.21451/1984-3143-2017-AR983
Peter RE, Lin HR, Van Der Kraak G (1988) Induced ovulation and spawning of cultured freshwater fish in China: advances in application of GnRH analogues and dopamine antagonists. Aquaculture. https://doi.org/10.1016/0044-8486(88)90080-4
Peter RE, Yu KL (1997) Neuroendocrine regulation of ovulation in fishes: basic and applied aspects. Rev Fish Biol Fish 7:173–197. https://doi.org/10.1023/A:1018431610220
Pires LB, Corrêa Filho RAC, Sanches EA et al (2018) Colossoma macropomum females can reproduce more than once in the same reproductive period. Anim Reprod Sci 196:138–142. https://doi.org/10.1016/j.anireprosci.2018.07.006
Prat F, Zanuy S, Carrillo M (2001) Effect of gonadotropin-releasing hormone analogue (GnRHa) and pimozide on plasma levels of sex steroids and ovarian development in sea bass (Dicentrarchus labrax L.). Aquaculture 198:325–338. https://doi.org/10.1016/S0044-8486(00)00600-1
Romagnoli S, Stelletta C, Milani C et al (2009) Clinical use of deslorelin for the control of reproduction in the bitch. Reprod Domest Anim 44:36–39. https://doi.org/10.1111/j.1439-0531.2009.01441.x
Rotili DA, Fornari DC, Zardo EL et al (2022) Sex steroid levels in females and males of Brycon orbignyanus throughout different juvenile and adult ages and during induction hormone in the mature females. Aquaculture 548:737695. https://doi.org/10.1016/j.aquaculture.2021.737695
Roubach R, Gomes LC, Leão Fonseca FA, Val AL (2005) Eugenol as an efficacious anaesthetic for tambaqui, Colossoma macropomum (Cuvier). Aquac Res 36:1056–1061. https://doi.org/10.1111/j.1365-2109.2005.01319.x
Scott BE, Marteinsdottir G, Begg GA et al (2006) Effects of population size/age structure, condition and temporal dynamics of spawning on reproductive output in Atlantic cod (Gadus morhua). Ecol Modell 191:383–415. https://doi.org/10.1016/j.ecolmodel.2005.05.015
Sharaf SM (2012) Effect of GnRHa, pimozide and Ovaprim on ovulation and plasma sex steroid hormones in African catfish Clarias gariepinus. Theriogenology 77:1709–1716. https://doi.org/10.1016/j.theriogenology.2011.12.019
Sink TD, Lochmann RT, Fecteau KA (2008) Validation, use, and disadvantages of enzyme-linked immunosorbent assay kits for detection of cortisol in channel catfish, largemouth bass, red pacu, and golden shiners. Fish Physiol Biochem 34:95–101. https://doi.org/10.1007/s10695-007-9150-9
Souza FN, de Fatima Ferreira Martins E, RAC CF et al (2018) Ovopel® and carp pituitary extract for induction of reproduction in Colossoma macropomum females. Anim Reprod Sci 195:53–57. https://doi.org/10.1016/j.anireprosci.2018.05.005
Stoeckel JN (2000) A method for viewing the germinal vesicle in oocytes of commercial catfishes. N Am J Aquac 62:240–247. https://doi.org/10.1577/1548-8454(2000)062<0240:amfvtg>2.3.co;2
Vacher C, Ferrière F, Marmignon MH et al (2002) Dopamine D2 receptors and secretion of FSH and LH: role of sexual steroids on the pituitary of the female rainbow trout. Gen Comp Endocrinol 127:198–206. https://doi.org/10.1016/S0016-6480(02)00046-1
Van Der Kraak G, Donaldson EM, Chang JP (1986) Dopamine involvement in the regulation of gonadotropin secretion in coho salmon. Can J Zool 64:1245–1248. https://doi.org/10.1139/z86-185
Vazirzadeh A (2015) Profiles of sex steroids in wild-caught carp Cyprinus carpio carpio Linnaeus 1758 during ovulation induction by acute versus sustained delivery methods of different GnRHa analogues. Asian Fish Sci 28:26–36
Villacorta-Correa MA, Saint-Paul U (1999) Structural indexes and sexual maturity of tambaqui Colossoma macropomum (Cuvier, 1818) (Characiformes: Characidae) in central Amazon, Brazil. Rev Bras Biol 59:637–652. https://doi.org/10.1590/S0034-71081999000400013
Woynárovich A, Van Anrooy R (2019) Field guide to the culture of tambaqui Colossoma macropomum, Cuvier, 1816. FAO Fish Aquac Tech Pap 624:132
Zaniboni Filho E, Barbosa DC (1996) Priming hormone administration to induce spawning of some brazilian migratory fish. Rev Bras Biol 56:655–659
Zohar Y, Muñoz-Cueto JA, Elizur A, Kah O (2010) Neuroendocrinology of reproduction in teleost fish. Gen Comp Endocrinol 165:438–455. https://doi.org/10.1016/j.ygcen.2009.04.017
Zohar Y, Mylonas CC (2001) Endocrine manipulations of spawning in cultured fish: From hormones to genes. Aquaculture. https://doi.org/10.1016/S0044-8486(01)00584-1
Acknowledgements
We would like to express our gratitude to the staff of the Centro de Tecnologia, Treinamento e Produção em Aquicultura de Balbina (CTTPA), and to Secretaria Executiva Adjunta de Pesca e Aquicultura (Sepa) da Secretaria de Produção Rural do Estado do Amazonas (SEPROR/AM) for their support during the study.
Funding
This study was funded by the EMBRAPA (SEG 10.19.03.004.00.00), by Fundação de Amparo à Pesquisa no Amazonas (FAPEAM)–A mazonas Estratégico 004/2018–call no. 39886.UNI703.23148.10052018, and by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–CAPES (fellowship grant awarded to the first author).
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, investigation, and writing–original draft preparation: Rosilane Gomes Souza de Oliveira. Investigation and writing–reviewing: Gabriela Brambila de Souza. Conceptualization and methodology: Alexandre Nizio Maria. Investigation: Ronãn Alves de Freitas. Funding acquisition, supervision, conceptualization, methodology, data curation, and writing–reviewing and editing: Fernanda Loureiro de Almeida.
Corresponding author
Ethics declarations
Ethics approval
The authors assert that all procedures of this work comply with the Ethical Principles of Animal Experimentation, adopted by the Brazilian College of Animal Experimentation (COBEA), and were approved by the Ethics Committee on Animal Use (CEUA) at EMBRAPA Amazônia Ocidental (no. 05/2018, SEI 21158.003669/2018-77). The project has authorization for Access to Genetic Heritage under register no. A5784B5.
Consent for publication
This is not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Handling editor: Brian Austin
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oliveira, R.G.S., Souza, G.B., Maria, A.N. et al. Effects of GnRH analogs on strip spawning and steroid plasma levels of Colossoma macropomum. Aquacult Int 32, 1027–1040 (2024). https://doi.org/10.1007/s10499-023-01201-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01201-9