Abstract
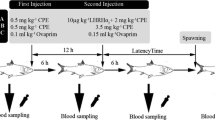
The use of recombinant gonadotropin-releasing hormone (rGnRH) has very rarely been tested in fish to promote spawning. This study evaluated the impact of recombinant chicken gonadotropin-releasing hormone (rcGnRH) with metoclopramide on the release of sex steroids and final maturation induction in goldfish (Carassius auratus) broodstock. For this purpose, goldfish broodstock was divided into four groups and treated with 0.9% NaCl with 20 mg/kg metoclopramide (Met) (C); 10 μg/kg body weight (BW) rcGnRH with 20 mg/kg metoclopramide (rcGn10); 15 μg/kg BW rcGnRH with 20 mg/kg metoclopramide (rcGn15); and 20 μg/kg BW rcGnRH with 20 mg/kg metoclopramide (rcGn20). The capability of the rcGnRH for eliciting biological response was tested in vivo by evaluating the changes of 17β estradiol (E2), testosterone (T), and 17α, 20β-dihydroxy-4-pregnen-3-one (DHP) and the induced spawning. Blood samples were obtained at 0 h, 12 h, and 24 h after injection. The rcGn10, rcGn15, and rcGn20 treatments induced lower E2 concentration, especially 24 h post-injection. T levels were significantly higher in rcGn10, rcGn15, and rcGn20 treatments 12 h post-injection than at 0 h and then decreased at 24 h post-injection. Furthermore, the rcGnRH tested significantly enhanced DHP secretion in rcGn10, rcGn15, and rcGn20 treatments 12 h post-injection before a decline at 24 h post-injection. No significant difference between the sampling times was found in the C treatment for the 3 sex steroids tested. The results also displayed that rcGnRH at 10–20 µg/kg of body weight can trigger spawning with the highest speed and efficiency of spawning at 20 µg/kg. The obtained results represent a possible strategy for enhancing the artificial reproduction and ovulation of broodstock fish by rGnRH and further support the use of recombinant hormones to promote reproduction in aquaculture.






Similar content being viewed by others
Data availability
The data and materials that support the findings of this study are available from the corresponding author upon reasonable request.
References
Aizen J, Hollander-Cohen L, Shpilman M, Levavi-Sivan B (2017) Biologically active recombinant carp LH as a spawning-inducing agent for carp. J Endocrinol 232:391–402. https://doi.org/10.1530/JOE-16-0435
Aizen J, Kobayashi M, Selicharova I, Sohn YC, Yoshizaki G, Levavi-Sivan B (2012) Steroidogenic response of carp ovaries to piscine FSH and LH depends on the reproductive phase. Gen Comp Endocronol 178: 28–36. https://doi.org/10.1016/j.ygcen.2012.04.002
Andersen O, Klungland H (1993) The salmon GnRH encoding gene in teleost fish. Int Rev Cytol 147:165–191. https://doi.org/10.1016/S0074-7696(08)60768-8
Barannikova IA (1999) Sex steroids in the serum of Caspian sturgeons and their specific cytosol binding in brain and gonads during the migratory cycle. J Appl Ichthyol 15:193–195. https://doi.org/10.1111/j.1439-0426.1999.tb00232.x
Bayunova L, Canario AV, Semenkova T, Dyubin V, Sverdlova O, Trenkler I, Barannikova I (2006) Sex steroids and cortisol levels in the blood of stellate sturgeon (Acipenser stellatus Pallas) during final maturation induced by LH‐RH‐analogue. J Appl Ichthyol 22:334–339
Brzuska E (2001) Artificial spawning of herbivorous fish: use of an LHRH-a to induce ovulation in grass carp Ctenopharyngodon idella (Valenciennes) and silver carp Hypophthalmichthys molitrix (Valenciennes). Aquac Res 30:849–856. https://doi.org/10.1046/j.1365-2109.1999.00413.x
Ceapa C, Williot P, MennF Le, Cuisset D (2002) Plasma sex steroids and vitellogenin levels in stellate sturgeon (Acipenser stellatus Pallas) during spawning migration in the Danube River. J Appl Ichthyol 18:391–396. https://doi.org/10.1046/j.1439-0426.2002.00370.x
Chekan JR, Koos JD, Zong C, Maksimov MO, Link AJ, Nair SK (2016) Structure of the lasso peptide Iso peptidase identifies a topology for processing threaded substrates. J Am Chem Soc 138:16452–16458. https://doi.org/10.1021/jacs.6b10389
Drori S, Ofir M, Levavi-Sivan B, Yaron Z (1994) Spawning induction in common carp, Cyprinuscarpio, using pituitary extract or GnRHsuperactive anagoge combined with metoclopramide: analysis of profile, progress of oocyte maturation and dependence on temperature. Aquaculture 119:393–407. https://doi.org/10.1016/0044-8486(94)90303-4
Falahatkar B, Barzafshan H, Asadi M (2016) Effects of LHRH-A2 on sex steroids levels, stress indices, and some plasma biochemical parameters in female Sterlet sturgeon, Acipenser ruthenus, broodstock. Iran J Fish Sci 3:121–136 ((In Persian))
Falahatkar B, Poursaied S (2013) Effects of hormonal manipulation on stress responses in male and female broodstocks of pikeperch Sander lucioperca. Aquacultint 22:235–244. https://doi.org/10.1007/s10499-013-9678-x
Gothilf Y, Zohar Y (1991) Clearance of different forms of GnRH from the circulation of the gilthead seabream, Sparus aurata, in relation to their degradation and bioactivities. In: Scott, A.P., Sumpter, J.P., Kime, D.E. and Rolfe, M.S. (eds.), Reproductive Physiology of Fish. Fish Symposium 91, Sheffield, pp. 35–37
Kontermann RE (2005) Recombinant bispecific antibodies for cancer therapy. Acta Pharmacol Sin 26:1–9. https://doi.org/10.1111/j.1745-7254.2005.00008.x
Leatherland JF, Li M, Barkataki S (2010) Stressors, glucocorticoids and ovarian function in teleosts. J Fish Biol 76:86–111. https://doi.org/10.1111/j.1095-8649.2009.02514.x
Mahadevi SF, Cheryl A, Bhosale MM, Gopalakannan A, Gnanavel K (2018) Induced breeding of telescopic gold fish (Carassius auratus auratus) using synthetic hormone (WOVA-FH). J Entomol Zool Stu 6:1368–1373
Majumder S, Das S, Moulik SR, Mallick B, PalP MD (2015) G-protein coupled estrogen receptor (GPER) inhibits final oocyte maturation in common carp, Cyprinus carpio. Gen Comp Endocrinol 211:28–38. https://doi.org/10.1016/j.ygcen.2014.11.011
Milla S, Wang N, Kestemont MSNM, P, (2009) Corticosteroids: friends or foes of teleost fish reproduction? Comp Biochem Physiol 153A:242251. https://doi.org/10.1016/j.cbpa.2009.02.027
Mohammadzadeh S, Moradian F, Yeganeh S, Falahatkar B, Milla S (2020) Design, production and purification of a novel recombinant gonadotropin-releasing hormone associated peptide as a spawning inducing agent for fish. Protein Expr purif 166 (In press). https://doi.org/10.1016/j.pep.2019.105510
Mohammadzadeh S, Yeganeh S, Moradian F, Milla S, Falahatkar B (2020) Spawning induction in Sterlet sturgeon (Acipenser ruthenus) with recombinant GnRH: analysis of hormone profiles and spawning indices. Aquaculture 533 (in press). https://doi.org/10.1016/j.aquaculture.2020.736108
MojaziAmiri B, Maebayashi M, Hara A, Adachi S, Yamauchi K (1996) Ovarian development and serum sex steroid and vitellogenin profiles in the female cultured sturgeon hybrid, the bester. J Fish Biol 48:1164–1178. https://doi.org/10.1111/j.1095-8649.1996.tb01812.x
Munakata A, Kobayashi M (2010) Endocrine control of sex behavior in teleost fish. Gen Com Endocrinol 165:456–468. https://doi.org/10.1016/j.ygcen.2009.04.011
Mylonas C, Zohar Y (2001) Use of GnRHa-delivery systems for the control of reproduction in fish. Rev Fish Biol Fisher 10:463–491
Nagahama Y, Yamashita M (2008) Regulation of oocyte maturation in fish. Development, Growth & Differentiation 50:S195–S219
Nagahama Y, Hirose K, Suzuki K, Tamaokis B-I (1983) Relative in Vitro Effectiveness of 17a,20/3-Dihydroxy-4-pregnen-3-one and Other Pregnene Derivatives on Germinal Vesicle Breakdown in Oocytes of Ayu (Plecoglossus altivelis), Amago Salmon (Oncorhynchus rhodurus), Rainbow Trout (Salmo gairdneri), and Goldfish (Carassius auratus). Gen Comp Endocrinol 51:15–23. https://doi.org/10.1016/0016-6480(83)90092-8
Okubo K, Nagahama Y (2008) Structural and functional evolution of gonadotropinreleasing hormone in vertebrates. Acta Physiolo 193:3–15. https://doi.org/10.1111/j.1748-1716.2008.01832.x
Omori Y, Kon T (2019) Goldfish: an old and new model system to study vertebrate development, evolution and human disease. J Biochem 165:209–18. https://doi.org/10.1093/jb/mvy076
Ota KG, Abe G (2016) Goldfish morphology as a model for evolutionary developmental biology. Wiley Interdiscip Rev Dev Biol 5:272–95. https://doi.org/10.1002/wdev.224
Podhorec P, Socha M, Amma BL, Sokolowska M, Brzuska E, Milla S, Gosiewski G, Stejskal V, Simko M, Kouril J (2016) The effects of GnRHa with and without dopamine antagonist on reproductive hormone levels and ovum viability in tench Tinca tinca. Aquaculture 465:158–163. https://doi.org/10.1016/j.aquaculture.2016.09.012
PourhoseinSarameh S, Falahatkar B, AzariTakami G, Efatpanah I (2012) Effects of different photoperiods and handling stress on spawning and reproductive performance of pikeperch Sander lucioperca. Anim Reprod Sci 132:213–222. https://doi.org/10.1016/j.anireprosci.2012.05.011
Shokr EAM (2020) Effect of gonadotropin releasing hormone injection on physiological changes and reproductive hormones in Clarias gariepinus. Egypt J Aquat Biol Fish 24: 119–129. https://doi.org/10.21608/ejabf.2020.69364
Szlachcic A, Zakrzewska M, Otlewski J (2011) Longer action means better drug: tuning up protein therapeutics. Biotechnol Adv 29:436–441. https://doi.org/10.1016/j.biotechadv.2011.03.005
Targonska K, Kucharczyk K (2011) The application of hCG, CPH and Ovopel in successful artificial reproduction of goldfish (Carassius auratus auratus) under controlled conditions. Reprod Domestic Anim 46:651–655. https://doi.org/10.1111/j.1439-0531.2010.01723.x
Wen M, Feron R, Pan Q, Guguin J, Jouanno E, Herpin A, Klopp C, Cbau C, Zahm M, Parrinello H, Journot L, Burgess SM, Omori Y, Postlethwait JH, Schartl M, Guiguen Y (2020) Sex chromosome and sex locus characterization in gold fish, Carassius auratus (Linnaeus, 1758). BMC Genomics 21:552. https://doi.org/10.1186/s12864-020-06959-3
Xu J, Zhu Z, Duan P, Li W, Zhang Y, Wu J, Hu Z, Liu RSR, J, (2006) Cloning, expression, and purification of a highly immunogenic recombinant gonadotropin-releasing hormone (GnRH) chimeric peptide. Protein Expr Purify 50:163–170. https://doi.org/10.1016/j.pep.2006.08.016
Yaron Z, Bogomolnaya A, Drori S, Biton I, Aizen J, Levavi-Sivan KZ, B, (2009) Spawning induction in the carp: past experience and future prospects - review. Israeli J Aquacult 61:5–26
Zadmajid V, Imanpour MR, Soudagar M, Shabani A (2009) The effect of GnRHa, HCG and pituitary extract on biochemical parameters of seminal plasma in godfish (Carassius auratus). Iranian J Anim Res 22:333–342 (In Persian)
Zareski D, Fontaine P, Alix M, Blecha M, Broquard C, Milla KJ, S, (2019) Time of response to hormonal treatment but not the type of a spawning agent affects the reproductive effectiveness in domesticated pikeperch, Sander lucioperca. Aquaculture 503:527–536. https://doi.org/10.1016/j.aquaculture
Zhou LY, Wang DS, Kobayashi T, Yano A, Paul-Prasanth B, Suzuki A, Nagahama SF, Y, (2007) A novel type of P450c17 lacking the lyase activity is responsible for C21-steroid biosynthesis in the fish ovary and head kidney. Endocrinol 148:4282–4291. https://doi.org/10.1210/en.2007-0487
Zohar Y, Mylonas CC (2001) Endocrine manipulation of spawning in cultured fish: from hormones to genes. Aquaculture 197:99–136. https://doi.org/10.1016/S0044-8486(01)005841
Acknowledgements
The authors thank the Fisheries Labs, Sari Agricultural Sciences, and Natural Resources University for its support.
Funding
The Iran Biotechnology Development Council under project No. 970202 funded this work. Moreover, this research was supported by University of Zabol (Iran) with grant code (UOZ-GR.9718:51).
Author information
Authors and Affiliations
Contributions
Sedigheh Mohammadzadeh: conceptualization, methodology, software, data curation, and writing- original draft preparation; Sylvain Milla: reviewing and editing; Ehsan Ahmadifar: reviewing and editing; Masoumeh Karimi: software and editing; Mahmoud A.O. Dawood: reviewing and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The experiments of the current study were approved by the Faculty of Natural Resources, University of Zabol, Zabol, Iran.
The authors declare that they have consent to participate.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohammadzadeh, S., Milla, S., Ahmadifar, E. et al. Is the use of recombinant cGnRH may be a future alternative to control the fish spawning? Let us go with the goldfish example. Fish Physiol Biochem 47, 951–960 (2021). https://doi.org/10.1007/s10695-021-00953-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-00953-6