Abstract
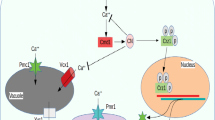
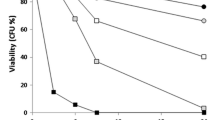
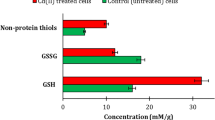
The toxic metal cadmium is linked to a series of degenerative disorders in humans, in which Cd-induced programmed cell death (apoptosis) may play a role. The yeast, Saccharomyces cerevisiae, provides a valuable model for elucidating apoptosis mechanisms, and this study extends that capability to Cd-induced apoptosis. We demonstrate that S. cerevisiae undergoes a glucose-dependent, programmed cell death in response to low cadmium concentrations, which is initiated within the first hour of Cd exposure. The response was associated with induction of the yeast caspase, Yca1p, and was abolished in a yca1Δ mutant. Cadmium-dependent apoptosis was also suppressed in a gsh1Δ mutant, indicating a requirement for glutathione. Other apoptotic markers, including sub-G1 DNA fragmentation and hyper-polarization of mitochondrial membranes, were also evident among Cd-exposed cells. These responses were not distributed uniformly throughout the cell population, but were restricted to a subset of cells. This apoptotic subpopulation also exhibited markedly elevated levels of intracellular reactive oxygen species (ROS). The heightened ROS levels alone were not sufficient to induce apoptosis. These findings highlight several new perspectives to the mechanism of Cd-dependent apoptosis and its phenotypic heterogeneity, while opening up future analyses to the power of the yeast model system.





Similar content being viewed by others
References
Duke RC, Ojcius DM, Young JD (1996) Cell suicide in health and disease. Sci Am 275:80–87
Shivapurkar N, Reddy J, Chaudhary PM, Gazdar AF (2003) Apoptosis and lung cancer: a review. J Cell Biochem 88:885–898
Hess EV (2002) Environmental chemicals and autoimmune disease: cause and effect. Toxicology 181–182:65–70
Phaneuf S, Leeuwenburgh C (2002) Cytochrome c release from mitochondria in the aging heart: a possible mechanism for apoptosis with age. Am J Physiol Regul Integr Comp Physiol 282:R423–R430
Brunet A, Sweeney LB, Sturgill JF et al (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303:2011–2015
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Lowe SW, Lin AW (2000) Apoptosis in cancer. Carcinogenesis 21:485–495
Madeo F, Frohlich E, Frohlich KU (1997) A yeast mutant showing diagnostic markers of early and late apoptosis. J Cell Biol 139:729–734
Reiter J, Herker E, Madeo F, Schmitt MJ (2005) Viral killer toxins induce caspase-mediated apoptosis in yeast. J Cell Biol 168:353–358
Gourlay CW, Du W, Ayscough KR (2006) Apoptosis in yeast—mechanisms and benefits to a unicellular organism. Mol Microbiol 62:1515–1521
Manon S, Chaudhuri B, Guerin M (1997) Release of cytochrome c and decrease of cytochrome c oxidase in Bax-expressing yeast cells, and prevention of these effects by coexpression of Bcl-xL. FEBS Lett 415:29–32
Ligr M, Madeo F, Frohlich E, Hilt W, Frohlich KU, Wolf DH (1998) Mammalian Bax triggers apoptotic changes in yeast. FEBS Lett 438:61–65
Blanchard F, Rusiniak ME, Sharma K et al (2002) Targeted destruction of DNA replication protein Cdc6 by cell death pathways in mammals and yeast. Mol Biol Cell 13:1536–1549
Chae HJ, Ke N, Kim HR et al (2003) Evolutionarily conserved cytoprotection provided by Bax Inhibitor-1 homologs from animals, plants, and yeast. Gene 323:101–113
Manon S (2004) Utilization of yeast to investigate the role of lipid oxidation in cell death. Antioxid Redox Signal 6:259–267
Uren AG, O’Rourke K, Aravind LA et al (2000) Identification of paracaspases and metacaspases: two ancient families of caspase-like proteins, one of which plays a key role in MALT lymphoma. Mol Cell 6:961–967
Madeo F, Herker E, Maldener C et al (2002) A caspase-related protease regulates apoptosis in yeast. Mol Cell 9:911–917
Jin YH, Clark AB, Slebos RJ et al (2003) Cadmium is a mutagen that acts by inhibiting mismatch repair. Nat Genet 34:326–329
Avery AM, Willetts SA, Avery SV (2004) Genetic dissection of the phospholipid hydroperoxidase activity of yeast gpx3 reveals its functional importance. J Biol Chem 279:46652–46658
Howlett NG, Avery SV (1997) Relationship between cadmium sensitivity and degree of plasma membrane fatty acid unsaturation in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 48:539–545
Lee J, Godon C, Lagniel G et al (1999) Yap1 and Skn7 control two specialized oxidative stress response regulons in yeast. J Biol Chem 274:16040–16046
Vido K, Spector D, Lagniel G, Lopez S, Toledano MB, Labarre J (2001) A proteome analysis of the cadmium response in Saccharomyces cerevisiae. J Biol Chem 276:8469–8474
Filipic M, Fatur T, Vudrag M (2006) Molecular mechanisms of cadmium induced mutagenicity. Hum Exp Toxicol 25:67–77
Fortuniak A, Zadzinski R, Bilinski T, Bartosz G (1996) Glutathione depletion in the yeast Saccharomyces cerevisiae. Biochem Mol Biol Int 38:901–910
Stohs SJ, Bagchi D, Hassoun E, Bagchi M (2001) Oxidative mechanisms in the toxicity of chromium and cadmium ions. J Environ Pathol Toxicol Oncol 20:77–88
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal ions. Free Radic Biol Med 18:321–336
Avery SV (2006) Microbial cell individuality and the underlying sources of heterogeneity. Nat Rev Microbiol 4:577–587
Wang J, Liu W, Uno T, Tonozuka H, Mitsui K, Tsurugi K (2000) Cellular stress responses oscillate in synchronization with the ultradian oscillation of energy metabolism in the yeast Saccharomyces cerevisiae. FEMS Microbiol Lett 189:9–13
Wang J, Liu W, Mitsui K, Tsurugi K (2001) Evidence for the involvement of the GTS1 gene product in the regulation of biological rhythms in the continuous culture of the yeast Saccharomyces cerevisiae. FEBS Lett 489:81–86
Smith MC, Sumner ER, Avery SV (2007) Glutathione and Gts1p drive beneficial variability in the cadmium resistances of individual yeast cells. Mol Microbiol 66:699–712
Klevecz RR, Bolen J, Forrest G, Murray DB (2004) A genomewide oscillation in transcription gates DNA replication and cell cycle. Proc Natl Acad Sci USA 101:1200–1205
Tu BP, Kudlicki A, Rowicka M, McKnight SL (2005) Logic of the yeast metabolic cycle: temporal compartmentalization of cellular processes. Science 310:1152–1158
Murray DB, Beckmann M, Kitano H (2007) Regulation of yeast oscillatory dynamics. Proc Natl Acad Sci USA 104:2241–2246
Xu C, Johnson JE, Singh PK, Jones MM, Yan H, Carter CE (1996) In vivo studies of cadmium-induced apoptosis in testicular tissue of the rat and its modulation by a chelating agent. Toxicology 107:1–8
Aydin HH, Celik HA, Deveci R et al (2003) Characterization of the cellular response during apoptosis induction in cadmium-treated Hep G2 human hepatoma cells. Biol Trace Elem Res 95:139–153
Lee WK, Abouhamed M, Thevenod F (2006) Caspase-dependent and -independent pathways for cadmium-induced apoptosis in cultured kidney proximal tubule cells. Am J Physiol Renal Physiol 291:F823–F832
Yokouchi M, Hiramatsu N, Hayakawa K et al (2007) Atypical, bidirectional regulation of cadmium-induced apoptosis via distinct signaling of unfolded protein response. Cell Death Differ 14:1467–1474
Kim SC, Cho MK, Kim SG (2003) Cadmium-induced non-apoptotic cell death mediated by oxidative stress under the condition of sulfhydryl deficiency. Toxicol Lett 144:325–336
Sancho P, Fernandez C, Yuste VJ et al (2006) Regulation of apoptosis/necrosis execution in cadmium-treated human promonocytic cells under different forms of oxidative stress. Apoptosis 11:673–686
Kim MS, Kim BJ, Woo HN et al (2000) Cadmium induces caspase-mediated cell death: suppression by Bcl-2. Toxicology 145:27–37
Shih CM, Wu JS, Ko WC et al (2003) Mitochondria-mediated caspase-independent apoptosis induced by cadmium in normal human lung cells. J Cell Biochem 89:335–347
Grant CM, MacIver FH, Dawes IW (1996) Glutathione is an essential metabolite required for resistance to oxidative stress in the yeast Saccharomyces cerevisiae. Curr Genet 29:511–515
Li ZS, Lu YP, Zhen RG, Szczypka M, Thiele DJ, Rea PA (1997) A new pathway for vacuolar cadmium sequestration in Saccharomyces cerevisiae: YCF1-catalyzed transport of bis(glutathionato)cadmium. Proc Natl Acad Sci USA 94:42–47
Shanmuganathan A, Avery SV, Willetts SA, Houghton JE (2004) Copper-induced oxidative stress in Saccharomyces cerevisiae targets enzymes of the glycolytic pathway. FEBS Lett 556:253–259
Brennan RJ, Schiestl RH (1996) Cadmium is an inducer of oxidative stress in yeast. Mutat Res 356:171–178
Chung J, Bachelder RE, Lipscomb EA, Shaw LM, Mercurio AM (2002) Integrin (alpha 6 beta 4) regulation of eIF-4E activity and VEGF translation: a survival mechanism for carcinoma cells. J Cell Biol 158:165–174
Kapitanovic S, Cacev T, Antica M et al (2006) Effect of indomethacin on E-cadherin and beta-catenin expression in HT-29 colon cancer cells. Exp Mol Pathol 80:91–96
Howlett NG, Avery SV (1999) Flow cytometric investigation of heterogeneous copper-sensitivity in asynchronously grown Saccharomyces cerevisiae. FEMS Microbiol Lett 176:379–386
Grabarek J, Amstad P, Darzynkiewicz Z (2002) Use of fluorescently labeled caspase inhibitors as affinity labels to detect activated caspases. Hum Cell 15:1–12
Madeo F, Frohlich E, Ligr M et al (1999) Oxygen stress: a regulator of apoptosis in yeast. J Cell Biol 145:757–767
Strouhal M, Kizek R, Vacek J, Trnkova L, Nemec M (2003) Electrochemical study of heavy metals and metallothionein in yeast Yarrowia lipolytica. Bioelectrochemistry 60:29–36
Madeo F, Herker E, Wissing S, Jungwirth H, Eisenberg T, Frohlich KU (2004) Apoptosis in yeast. Curr Opin Microbiol 7:655–660
Balzan R, Sapienza K, Galea DR, Vassallo N, Frey H, Bannister WH (2004) Aspirin commits yeast cells to apoptosis depending on carbon source. Microbiology 150:109–115
Wolbers F, Buijtenhuijs P, Haanen C, Vermes I (2004) Apoptotic cell death kinetics in vitro depend on the cell types and the inducers used. Apoptosis 9:385–392
Hauptmann P, Riel C, Kunz-Schughart LA, Frohlich KU, Madeo F, Lehle L (2006) Defects in N-glycosylation induce apoptosis in yeast. Mol Microbiol 59:765–778
Hall AG (1999) Review: the role of glutathione in the regulation of apoptosis. Eur J Clin Invest 29:238–245
Ludovico P, Rodrigues F, Almeida A, Silva MT, Barrientos A, Corte-Real M (2002) Cytochrome c release and mitochondria involvement in programmed cell death induced by acetic acid in Saccharomyces cerevisiae. Mol Biol Cell 13:2598–2606
Drakulic T, Temple MD, Guido R et al (2005) Involvement of oxidative stress response genes in redox homeostasis, the level of reactive oxygen species, and ageing in Saccharomyces cerevisiae. FEMS Yeast Res 5:1215–1228
Gross A, Pilcher K, Blachly-Dyson E et al (2000) Biochemical and genetic analysis of the mitochondrial response of yeast to BAX and BCL-X(L). Mol Cell Biol 20:3125–3136
Poliakova D, Sokolikova B, Kolarov J, Sabova L (2002) The antiapoptotic protein Bcl-x(L) prevents the cytotoxic effect of Bax, but not Bax-induced formation of reactive oxygen species, in Kluyveromyces lactis. Microbiology 148:2789–2795
Wysocki R, Kron SJ (2004) Yeast cell death during DNA damage arrest is independent of caspase or reactive oxygen species. J Cell Biol 166:311–316
Wemmie JA, Steggerda SM, MoyeRowley WS (1997) The Saccharomyces cerevisiae AP-1 protein discriminates between oxidative stress elicited by the oxidants H2O2 and diamide. J Biol Chem 272:7908–7914
Fleury C, Mignotte B, Vayssiere JL (2002) Mitochondrial reactive oxygen species in cell death signaling. Biochimie 84:131–141
Brace JL, Vanderweele DJ, Rudin CM (2005) Svf1 inhibits reactive oxygen species generation and promotes survival under conditions of oxidative stress in Saccharomyces cerevisiae. Yeast 22:641–652
Waring P, Lambert D, Sjaarda A, Hurne A, Beaver J (1999) Increased cell surface exposure of phosphatidylserine on propidium iodide negative thymocytes undergoing death by necrosis. Cell Death Differ 6:624–637
Zhang Q, Chieu HK, Low CP, Zhang S, Heng CK, Yang H (2003) Schizosaccharomyces pombe cells deficient in triacylglycerols synthesis undergo apoptosis upon entry into the stationary phase. J Biol Chem 278:47145–47155
Silva RD, Sotoca R, Johansson B et al (2005) Hyperosmotic stress induces metacaspase- and mitochondria-dependent apoptosis in Saccharomyces cerevisiae. Mol Microbiol 58:824–834
Wheeler GL, Grant CM (2004) Regulation of redox homeostasis in the yeast Saccharomyces cerevisiae. Physiol Plant 120:12–20
Dormer UH, Westwater J, McLaren NF, Kent NA, Mellor J, Jamieson DJ (2000) Cadmium-inducible expression of the yeast GSH1 gene requires a functional sulfur-amino acid regulatory network. J Biol Chem 275:32611–32616
Fauchon M, Lagniel G, Aude JC et al (2002) Sulfur sparing in the yeast proteome in response to sulfur demand. Molecular Cell 9:713–723
Sugiyama K, Izawa S, Inoue Y (2000) The Yap1p-dependent induction of glutathione synthesis in heat shock response of Saccharomyces cerevisiae. J Biol Chem 275:15535–15540
Watjen W, Beyersmann D (2004) Cadmium-induced apoptosis in C6 glioma cells: influence of oxidative stress. Biometals 17:65–78
Flower TR, Chesnokova LS, Froelich CA, Dixon C, Witt SN (2005) Heat shock prevents alpha-synuclein-induced apoptosis in a yeast model of Parkinson’s disease. J Mol Biol 351:1081–1100
Murray DB, Engelen F, Lloyd D, Kuriyama H (1999) Involvement of glutathione in the regulation of respiratory oscillation during a continuous culture of Saccharomyces cerevisiae. Microbiology 145(Pt 10):2739–2745
Adams CA, Kuriyama H, Lloyd D, Murray DB (2003) The Gts1 protein stabilizes the autonomous oscillator in yeast. Yeast 20:463–470
Sohn HY, Kum EJ, Kwon GS, Jin I, Adams CA, Kuriyama H (2005) GLR1 plays an essential role in the homeodynamics of glutathione and the regulation of H2S production during respiratory oscillation of Saccharomyces cerevisiae. Biosci Biotechnol Biochem 69:2450–2454
Acknowledgments
This work was supported by a grant from the NIH (R01 GM57945). The support of the Georgia Research Alliance is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nargund, A.M., Avery, S.V. & Houghton, J.E. Cadmium induces a heterogeneous and caspase-dependent apoptotic response in Saccharomyces cerevisiae . Apoptosis 13, 811–821 (2008). https://doi.org/10.1007/s10495-008-0215-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-008-0215-8