Abstract
Genetic polymorphism in key metabolic genes plays a pivotal role in shaping phenotypes and adapting to varying environments. Polymorphism in the metabolic gene 6-phosphogluconate dehydrogenase (6Pgdh) in bulb mites, Rhizoglyphus robini is characterized by two alleles, S and F, that differ by a single amino acid substitution and correlate with male reproductive fitness. The S-bearing males demonstrate a reproductive advantage. Although the S allele rapidly fixes in laboratory settings, the persistence of polymorphic populations in the wild is noteworthy. This study examines the prevalence and stability of 6Pgdh polymorphism in natural populations across Poland, investigating potential environmental influences and seasonal variations. We found widespread 6Pgdh polymorphism in natural populations, with allele frequencies varying across locations and sampling dates but without clear geographical or seasonal clines. This widespread polymorphism and spatio-temporal variability may be attributed to population demography and gene flow between local populations. We found some correlation between soil properties, particularly cation content (Na, K, Ca, and Mg) and 6Pgdh allele frequencies, showcasing the connection between mite physiology and soil characteristics and highlighting the presence of environment-dependent balancing selection. We conducted experimental fitness assays to determine whether the allele providing the advantage in male–male competition has antagonistic effects on life-history traits and if these effects are temperature-dependent. We found that temperature does not differentially influence development time or juvenile survival in different 6Pgdh genotypes. This study reveals the relationship between genetic variation, environmental factors, and reproductive fitness in natural bulb mite populations, shedding light on the dynamic mechanisms governing 6Pgdh polymorphism.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Balancing selection is the process through which polymorphism within a population is actively maintained over generations. Such maintenance of genetic variation is particularly important in genes that govern key metabolic traits, as variation in these genes has the potential to significantly impact the organism’s fitness and chances of survival (Koshiba et al. 2020; Whitt et al. 2002). Despite the general conservation of metabolic genes (Mukherjee et al. 2018; Kapahi et al. 2010), the presence of functional differences and selective pressures acting on them suggest, in some cases, the action of balancing selection. Metabolic genes can affect multiple aspects of an organism’s physiology and many traits. Therefore, balancing selection on these genes is likely to take the form of antagonistic pleiotropy, where a gene affects two or more traits of an organism with opposite effects on fitness (Stearns 1998). In genes with multiple functional alleles under antagonistic pleiotropy, one of the alleles is beneficial for one trait while the other for another trait (oftentimes with additional detrimental effects on fitness), thus creating a tradeoff between different fitness components (Meyer and Zanger 1997; Kozyra et al. 2017; Di Bartolomeo et al. 2020). Antagonistic pleiotropy is already a recognized mechanism for the maintenance of polymorphism (Hedrick 1999).
This mechanism becomes even more intriguing when we consider the complex interplay of environmental factors (Brown and Kelly 2018; Mérot et al. 2020) where antagonistic pleiotropy might be present in some environments, but not in others. Different enzyme variants can be favored at different environments, resulting in selection on such enzyme variants and influencing the geographic patterns of genetic diversity. For example, in Drosophila serrata (Rusuwa et al. 2022), the polymorphism in a single gene influencing circular hydrocarbon profile (CHC) is maintained in warm and humid climates but not in hot and dry ones. This is because one of the alleles is associated with male reproductive advantage, but lowers female desiccation resistance, resulting a trade-off between male reproduction and female stress response (termed also sexually antagonistic pleiotropy) and driving balancing selection in specific climate conditions (Rusuwa et al. 2022). The influence of environmental heterogeneity on functional polymorphism in metabolic genes was shown by Kerwin et al (2015) on the gene involved in glucosinolate production in Arabidopsis thaliana, which showed that no allele consistently outperformed another in different environments, emphasizing the importance of environmental heterogeneity as an evolutionary force. Understanding the relationship between environmental factors and the functioning of enzyme variants is essential for gaining insight on how organisms adapt to their environment, utilize resources (Vieille and Zeikus 2001), defend against diseases (Hollman et al. 2016), and evolve (Rix et al. 2020).
This study focuses on the metabolic gene 6-Phosphogluconate dehydrogenase (6Pgdh), which is also a target of sexual selection in bulb mite, Rhizoglyphus robini. 6Pgdh is one of the key enzymes in the Pentose Phosphate Pathway (PPP), an alternative to glycolysis. It produces NADPH and ribose-5-phosphate used in the synthesis of fatty acids, sterols and nucleotides (Ge et al. 2020). PPP is also a source of amino acid and vitamin B6 precursors (Tambasco-Studart et al. 2005). The pathway is essential for energy metabolism of a cell and plays an important role in stress response to abiotic factors such as temperature, salinity, drought, etc. (Fahrendorf et al. 1995; Krüger et al. 2011; Hou et al. 2007). The expression levels and activities of PPP enzymes are associated with environmental factors (Watts and Lawrence 1990). Environmental heterogeneity has been shown to drive patterns of polymorphism in the 6Pgdh and some other genes involved in PPP in organisms ranging from invertebrates (González-Ruiz et al. 2023) to plants (Landi et al. 2021). However, to really understand how selection of genes involved in PPP is driven by environment, we need to connect geographical and temporal patterns of polymorphism with phenotypic differences between genotypes and environmental background of these differences.
6Pgdh coding sequence in bulb mites consists of 486 amino acids and is divided by four introns. While four SNPs have been found in the coding sequence (Skwierzyńska and Plesnar-Bielak 2018), only one is associated with amino acid substitution (arginine to methionine). The two alleles of 6Pgdh, S and F correlate with reproductive fitness of males. Males bearing the S allele gain higher reproductive success compared to males with the F allele (Konior et al. 2006; Łukasik et al. 2010), due to higher sperm production and copulation frequency (Skwierzyńska and Plesnar-Bielak 2018). Female fitness seems to be independent of the 6Pgdh genotype, but the S-bearing males reduce the fitness of their female partners, even though the exact mechanism behind it is unknown (Konior et al. 2006). As expected from the advantage in male competition, the S allele rapidly fixes in laboratory conditions, but surprisingly, the polymorphism seems to be maintained in some natural populations (Konior et al. 2006; Łukasik et al. 2010, personal observations). There is very limited knowledge regarding the ecological conditions linked to this polymorphism and we hypothesize the active maintenance of polymorphism of this metabolic gene is environment-dependent. Nevertheless, a systematic study on the patterns of 6Pgdh variation in natural bulb mite populations has not been conducted before so it is not known how common and stable over time the polymorphism is.
Here, we study several natural bulb mite populations to look into the amount of polymorphism and its geographical variation across Poland. To start with, we aim to correlate 6Pgdh polymorphism levels with latitude and longitude, most important macro-climatic factors, as well as with local soil properties. Moreover, we investigate seasonal variation in 6Pgdh frequencies, by investigating frequency shifts between spring and autumn. Finally, we explore fitness differences between 6Pgdh genotypes at different temperatures. We test if the allele providing advantage in male-male competition has antagonistic effects on life history traits and if these effects are temperature-dependent, as earlier studies have suggested the effects of temperature on PPP (Kauffman et al. 1969) and on 6Pgdh allele frequencies (Plesnar-Bielak et al. 2020). So, we further complement our field study with experimental fitness assays at different temperatures. The expression of the S allele, that conveys reproductive advantage to males, might be associated with energetic costs that could result in reduced juvenile survival and/or longer development time for individuals bearing this allele.
Methods
Study species
The bulb mites, Rhizoglyphus robini (Acari: Acaridae) are common pests with cosmopolitan geographical distribution. They inhabit subterrain parts of Lilliaceae and other plants (Díaz et al. 2000). The life cycle of Rhizoglyphus robini consists of egg, larva, protonymph, tritonymph, and adult stages, with a facultative migratory stage of deutonymph, which develop from protonymphs when conditions are unfavorable (overcrowding, low food availability etc.). The bulb mites do not enter a diapause (Gerson et al. 1991), remaining active throughout the year. However, their activity might be reduced during colder months.
Rhizoglyphus robini is characterized by high promiscuity (Radwan and Siva-Jothy 1996) and is used as model species in sexual selection studies (e.g. Smallegange and Coulson 2011; Jarzebowska and Radwan 2010; Plesnar-Bielak et al. 2012; Łukasiewicz et al. 2017; Parrett et al. 2022). Both males and females mate multiply, with mating frequency depending on environmental conditions (Gerson and Thorens 1982). While male fitness increases with the number of copulations, multiple mating is associated with fitness cost to females, signifying sexual conflict (Tilszer et al. 2006). The amount of male harm has been shown to depend on a male’s 6Pgdh genotype, such that mating with males bearing the S allele is associated with higher cost than mating with a male lacking this allele (Konior et al. 2006). This effect might, at least to some extent, be caused by higher ability of the S-bearers to exert more frequent copulations of females (Skwierzyńska and Plesnar-Bielak 2018), but the actual physiological mechanism of female fitness reduction is not clear.
DNA extraction and 6Pgdh genotyping
DNA was extracted from individual mites. Each individual was placed in 1% chelex solution (40 μl) and was crushed. Then 3 μl of proteinase-K (EurX) was added, and the mixture was incubated in a thermocycler (10 min 94 °C, 15 min 75 °C).
The 6Pgdh genotyping was done using Real-Time PCR with fluorogenic TaqMan probes (Thermofisher Scientific) specific for the missense single nucleotide polymorphism determining the F and S alleles. The Bio-Rad CFX96 Real-Time PCR detection system was used for the genotyping. A TaqMan Genotyping Master Mix (Thermofisher Scientific) and Custom Genotyping Assay that included allele-specific primers and fluorescent probes were mixed in 10:1 ratio. 5.5 μl of such a mix and 4.5 μl of DNA were put in a 96 well plate for genotyping. PCR was performed in 41 cycles (15 s 95 °C, 1 min 60 °C).
The patterns of 6Pgdh polymorphism in the wild
Population sampling was carried out in Poland, which has a clear gradient of climatic and environmental conditions such as temperature, precipitation, air pressure, etc. from south-west to north-east (Błaś and Ojrzyńska 2024; Blazejczyk 2006) that might affect 6Pgdh frequencies. Moreover, there is some record of variation in the level of 6Pgdh polymorphism in Poland and substantial genetic diversity within populations with little structuring between populations in this region (Kolasa et al. unpublished; Boroń et al. unpublished; Przesmycka and Radwan 2023). Sampling was done between October 2021 and December 2022. The main sampling was done in late Spring/Summer (May, June) with some locations sampled also in Autumn (October, November) to see how stable 6Pgdh frequencies are across seasons.
Samples of bulbs of different plant species were collected from private gardens and botanical gardens across different regions in Poland (Table 1) and checked for the presence of mites. Between 2 and 6 plant bulbs together with soil samples (taken only during Spring sampling and for a subset of samples) were collected per location, depending on availability.
In the lab, bulb mites, if present, were transferred to plastic containers (diameter ≈ 2.5 cm) with plaster of Paris soaked with water (which are standard containers to keep large groups of mites). They were kept at 12 °C and fed powdered yeast ad libitum. Ca. 40 individuals from each sample (location, see Table 1) were genotyped within 2 months after collection to ensure that the individuals collected as juveniles reached adulthood.
Climate data
Climate data was obtained from the KNMI climate explorer website (https://climexp.knmi.nl). Daily values of mean surface temperature (in °C) and precipitation/rainfall (in mm/day) were obtained from the E-OBS database, with 0.25° regular grids. Daily climatic values were obtained for each location using their coordinates (from the grid they belonged to) for 60 days before the bulb collection date. The values were then averaged for each location (so that we obtained a mean for a 60-day long period before collection) and used for the analysis. Using the same procedure, we also calculated average surface temperatures and rainfall values for the 30 and 90-day periods before collection.
Soil analyses
Soil samples were collected from sampling points (10 sampling points) near the plant using teaspoons and ensuring that the points were within 1 m2 of the plant and were kept in 12 °C for analysis. The dry weight (DW) of the soil samples was determined by measuring mass loss (water) after soil samples dried at 105 ± 1 °C for 24 h. Next, the organic matter content (OM) in soil dry weight was determined as the mass loss on ignition at 550 ± 1 °C for 24 h. The water holding capacity (WHC), which is the amount of water that a given soil can hold without leaking, was measured by a standard gravimetric method after soil soaking for 24 h in net-ended plastic pipes immersed in water. The organic carbon (C), total nitrogen (N), and total sulfur (S) were analyzed by dry combustion of ca 10 mg milled soil samples with an elemental analyser (Vario El III, Elementar Analysensysteme GmbH). The soil pH was measured in air-dried subsamples (2 g) shaken in deionised water (1:10 w:v) for 1 h at 200 rpm (pH-meter with glass electrode).
The total element concentrations, that is phosphorus (P), calcium (Ca), potassium (K), magnesium (Mg), manganese (Mn), and sodium (Na) in each soil sample were determined after wet digestion of ca 0.5 g of DW in 10 ml of SupraPure-concentrated HNO3 and HClO4 (7:1 v/v) (Sigma-Aldrich). A flow injection analyser (FIA compact, MLE, Radebeul, Germany) was used to determine the P content. The total concentrations of the other elements were measured using atomic absorption spectrometry (AAS) with a flame nebulizer (Perkin-Elmer, AAnalyst200, Waltham, Massachusetts, USA). The accuracy of the mineralization process was determined using blank samples as well as standard certified material (CRM025-050, Sandy Loam 8, RT Corp.). Each analysis was performed in two subsamples from each soil sample, and the data were averaged and expressed based on the dry weight of the soil.
Laboratory population
For the life-history fitness experiments, we used a population enriched in the F allele that was established from a field population obtained in July 2020 from Łazany (49.9476, 20.1535) near Kraków. Several dozens of individuals collected from an onion were placed in a common container with powder yeast that served as food. Such obtained population was kept at 8 °C, with the exception of a 1-week period after we finished collecting individuals, when it was moved to 24 °C to let juvenile individuals develop so that population would expand. The F-increased population was created in spring 2021, when the F allele frequency in the source population was about 0.23. To do it, we randomly paired virgin females and males from a source population. After the pairs mated and females laid eggs, both parents were genotyped. Eight offspring from pairs with parents having at least 2 copies of F allele (either both parents FS, or one FF and one SS, or one FF and one SF, or both parents FF) were transferred as larvae/protonymphs to a common container to establish a population with increased F allele frequency. We used two containers (with offspring from the same parental pairs moved to both of them) that established two subpopulations that were mixed and divided again after ca. 2 months. The population was let to expand freely for ca. 2 months at 24 °C (which corresponds to 3–4 mite generations), before it was moved to 12 °C to elongate generation time, slowing down population’s evolution and the loss of the F allele. At all these stages the population was kept at > 90% humidity and constant darkness, with powdered yeast provided ad libitum as a food source.
Development time
Development time of the individuals with different genotypes was measured at three temperatures, 24 °C (standard temperature in which the laboratory populations are reared), 12 °C (average yearly ground temperature at 5–10 cm depth in Poland) and 8 °C (low temperature relevant to colder months of sampling) with three replicates per temperature (see Fig. 1). Per each replicate, ten females were randomly selected from the F-increased population were kept in containers for 24 h to lay eggs. After the females were removed, the containers with the eggs were placed to experimental temperatures. The eggs were allowed to develop. When they reached the stage of tritonymph (last juvenile stage), they were checked every 24 h for emerging adults. Adults that emerged were taken out from the containers and date of emergence and sex were noted. Then, the individuals were genotyped for 6Pgdh. The checks continued until all the adults emerged.
Juvenile survival differences between genotypes
Juvenile survival differences were also tested at 24 °C, 12 °C and 8 °C. For the assay, 50 females were put in a common container (five replicates per temperature) and allowed to lay eggs at 24 °C for 4 days, after which they were removed (see Fig. 1). The containers were then transferred to their respective experimental temperatures (24 °C, 12 °C and 8 °C). After all the adults emerged, around 40 individuals from each replicate were genotyped for the 6Pgdh. We calculated the frequencies of the F allele at each temperature and used them as a proxy of juvenile survival differences between genotypes. If juvenile survival of individuals with different alleles is temperature-independent, we expect that allele frequencies in adults do not differ between temperatures. A higher frequency of a certain allele at a given temperature, indicates higher survival of the individuals bearing this allele.
Statistical analysis
6Pgdh genotype frequencies in field samples were tested for Hardy–Weinberg equilibrium with likelihood ratio test implemented in Hardy–Weinberg package in R (Graffelman and Weir 2016). The frequencies from the samples collected in spring (when most of the samples were collected) were checked for their relationship with latitude and longitude of the location to look for geographical cline 6Pgdh polymorphism. We applied a quasibinomial model accounting for overdispersion (using glm function in R v3.6.1) with a vector of S and F allele counts at each location as a response variable and latitude and longitude as independent variables. For plotting the data points on the map of Poland, QGIS (v3.34.0-Prizren) was used along with the map shape file obtained from GADM data (v4.1).
Both mean temperature and precipitation levels were highly correlated (r22 = 0.57, p < 0.01 for a 60-day-long period). They were analyzed in separate quasibinomial models with a vector of S and F allele counts from each location as the response variable and the climatic variable (mean surface temperate or precipitation) as the predictor variable. The models were rerun for data averaged for 30 and 90 days. Since the results were qualitatively the same, we present only the analyses for 60 days.
To test for a correlation between 6Pgdh frequencies and soil characteristics, we first summarized soil parameters with Principal Component Analysis. Then, we ran a generalized linear model with a vector of S and F allele counts at each location as a response variable and PC1 and PC2 as independent variables. Again, quasibinomial distribution was used to account for overdispersion in our data.
To check how genotype affects development time at different temperatures, we used a linear mixed model fit with the number of days taken for development (transformed with square root) as the response variable and with genotype and temperature (factor) as the dependent variables and population ID as random factor. We also checked to see if the effect of sex of the individuals was important to the model using AIC scores, but the effect of sex did not improve the model and the conclusions remained unchanged and hence the effect of sex was removed from the main model. The function lmer was used for the analysis in R (the package lmertest, lme4, v1.1-26).
The allele frequencies of the individuals that survived to adulthood at each temperature were obtained from the juvenile survival experiment. To analyze the data, a binomial model was used with a vector of S and F allele counts in each replicate as the response variable and temperature as the dependent variable using the glm function in R (glm2 package, v1.2.1).
Results
The patterns of 6Pgdh polymorphism in the wild
We found 6Pgdh polymorphism in a majority (15 out of 17) of populations (Fig. 2), with only two of them having just one allele (S). In polymorphic populations, F-allele frequencies varied from 0.026 to 0.60, with a mean frequency of 0.23 (SD = 0.19). Mean frequency was similar for the samples collected in Autumn (mean ± SD 0.191 ± 0.18) and Spring (0.243 ± 0.19). 6Pgdh allele frequencies varied a lot between Autumn and Spring, but the changes were not consistent in their direction, with F frequency increasing in Spring in some locations, but decreasing in others (Fig. 3). Similarly, samples taken from the same botanical garden substantially differed in allele frequencies, even in the same season. Eighteen samples were in Hardy–Weinberg equilibrium. In 6 samples (Table 1), we found deviation from Hardy–Weinberg equilibrium and observed heterozygosity was lower than expected in all these cases. We did not find evidence for geographical clines in 6Pgdh frequencies in Poland (longitude: effect estimate − 0.285, t = − 1.78, p = 0.089, latitude: effect estimate − 0.406, t = − 1.88, p = 0.075). Similarly, we did not find any influence of climatic variables in 6Pgdh allele frequencies (temperature: effect estimate 0.132, t = − 1.27, p = 0.219, precipitation: effect estimate − 0.402, t = − 0.62, p = 0.543). The data from the 90 days and 30 days interval provided with similar results (not shown).
The association of 6Pgdh polymorphism with soil properties
PC1 and PC2 explained 44.3 and 19.8% of the total variance in soil parameters, respectively (Fig. 4a). PC1 was mainly influenced by organic matter, S, N and C content (Fig. 4b). Higher PC1 values were also associated with lower pH. PC2 values were negatively correlated with Na, K, Ca and Mg content. We found a significant negative relationship between F allele frequencies and PC2 (t11 = − 2.99, p = 0.017), but not PC1 (t11 = 1.584, p = 0.152). (Fig. 4c).
a The results from the PCA showing the PC1 and PC2 axes explaining 44.3% and 19.8% of the variance in soil patterns respectively. b The inverse relationship of the second PC axis with the Na, Mg, K and Ca content of the soil. Units: S (%), P (%), organic matter (%), Na (mg kg−1), N (%), Mn, Mg, K, Ca (mg kg−1), C (%). All data are expressed per dry soil mass. c Negative relationship between PC2 and F-allele frequency
Laboratory fitness experiments
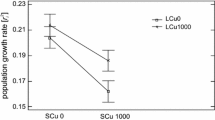
Development time increased with decreasing temperature (F2; 8 = 7521.85, p < 0.01). There was no difference in development time between genotypes (F2; 953 = 1.53, p = 0.22) or genotype by temperature interaction (F4; 954 = 1.33, p = 0.25) (Fig. 5). The effect of sex was also not significant (F1; 950 = 1.31, p = 0.22).
a Development time for bulb mites with different genotypes of the 6Pgdh allele at different temperatures. The boxplot shows the median along with the interquartile range. The points represent the spread of individual data points. b F-allele frequencies of the juveniles that survived at each temperature. The boxplot shows the median along with the interquartile range. The data points represent the F-frequency of the replicates
F allele frequencies did not differ between groups developed at different temperatures (effect estimate 0.002, z = 0.17, p = 0.86), indicating there is no temperature-dependent difference in juvenile survival between genotypes (Fig. 5).
Discussion
Genetic polymorphisms in key metabolic genes can potentially influence a wide range of phenotypic traits and may therefore be important for adaptive polymorphism. 6Pgdh is an example of such a gene with different allelic variants influencing fitness in vertebrates (Rivera et al. 2006; Chen et al. 2023), plants (Oostermeijer et al. 1995) and invertebrates (Begun and Aquardo 1994; Kilias and Alahiotis 1985), including bulb mites (Rhizoglyphus robini). The maintenance of such polymorphism under natural conditions is surprising, particularly in the case of bulb mites, given the strong reproductive advantage of the S allele leading to its rapid fixation in the laboratory. In some environments, however, reproductive advantage may be balanced by metabolic costs that could lead to a trade-off between sexual and non-sexual fitness, leading to the maintenance of stable polymorphism under these conditions (Robinson et al. 2006; Höglund et al. 1998). Our study investigates the abundance of 6Pgdh polymorphism in natural populations, explores potential environmental factors influencing these patterns in the wild, and examines potential trade-offs in laboratory settings.
We show that 6Pgdh polymorphism is indeed common in natural populations. The majority of screened populations were 6Pgdh-polymorphic, but the actual allele frequencies varied. The abundance of polymorphism aligns with certain observations reported in prior studies (Łukasik et al. 2010). For example, a natural population from Poland found by Konior et al. (2006) was polymorphic in respect to 6Pgdh with F-allele frequency of 0.11. Screening of ten populations in Poland by Skwierzyńska and Plesnar-Bielak (2018) found only one polymorphic population, suggesting 6Pgdh polymorphism to be rare. However, the search was aimed at finding a population of a relatively high polymorphism level to use in laboratory experiments. Hence, the sampling might have not been suitable for detecting moderate F frequencies, which were not uncommon in the current study.
6Pgdh frequencies varied between populations and sampling dates, but there was no seasonal or latitudinal pattern. Neither surface temperature nor precipitation/rainfall were able to explain the variation in 6Pgdh allele frequencies. The high variability of 6Pgdh allele frequencies is supported by previous observations in bulb mites. For example, the F frequency in a natural population was 0.34 in 2003, but it decreased below 0.05 at the same site a year later (Łukasik et al. 2010). The lack of geographic, macroclimatic or seasonal patterns suggests that perhaps factors other than temperature and rainfall may play a more significant role in shaping the frequencies of 6Pgdh alleles or that the effect of these variables may occur at a much finer scale. These fine scale effects might indeed be important; for example, plant cover may significantly affect shading and hence drastically affect both temperature and humidity (Procházka et al. 2011; Zhang et al. 2013). Similarly, since our sites were located in gardens, plant watering was likely to overwrite the effects of large-scale precipitation patterns. On the other hand, our results from the laboratory experiments do not support temperature’s role in shaping 6Pgdh frequencies in the bulb mite. We found no evidence that temperature differentially affects two life-history traits: development time and juvenile survival, in individuals with different 6Pgdh genotypes. This suggests that temperature does not have an effect on the allele fitness or the distribution of alleles on neither microhabitat level (life-history assays) nor macrohabitat scale (field study). Similarly, a prior laboratory investigation demonstrated that there is no reversal in F-allele fitness across temperatures, supporting our conclusion (Plesnar-Bielak et al. 2020). While the results from the laboratory studies suggest that temperature doesn’t contribute to the levels of 6Pgdh allele frequencies, we cannot rule out other factors as demonstrated in other species. For example, two studies in Drosophila (D. melanogaster and D. simulans) showed clear latitudinal clines of 6Pgdh allele frequencies in different regions across the world, likely associated with climatic conditions (Oakeshott et al. 1983; Begun and Aquadro 1994). Similarly, latitudinal clines in 6Pgdh frequencies have been associated with other selective factors such as water salinity in Atlantic killifish (Fundulus heteroclitus) (Powers et al. 1986). Stockwell and Mulvey (1998) also explicitly demonstrated that it was water salinity, and not temperature, that influenced the polymorphism levels in white sands pupfish, Cyprinodon tularosa (Stockwell and Mulvey 1998).
Our study found a significant effect of soil properties on the level of polymorphism, with higher amounts of cations (Na, K, Ca and Mg) in the soil corresponding to higher frequencies of the F allele. Bulb mites, being subterranean organisms reliant on soil, can experience direct or indirect effects of soil composition on their physiology. Soil properties can affect plant diversity and soil fauna indirectly (Kudureti et al. 2023). Indeed, evidence for soil properties differentially affecting fitness of multiple mite species have already been observed. Soil properties such as pH, nitrogen and carbon content, among other variables have been shown to affect community composition (Nielsen et al. 2010) and diversity (de Moraes et al. 2011) in oribatid mite group, suggesting they drive fitness differences at inter-species level. Our results suggest that soil properties can differentially affect mite fitness at the intra-species level too. The effects of soil could be mediated by factors like vegetation or soil microbial community composition (Li et al. 2023; Pineda et al. 2017). For example, host-microbiome interactions in bulb mites affect nutrition (Zindel et al. 2013), which in turn impacts fitness and traits like development rate and body size (Leigh and Smallegange 2014). Nutritional conditions can also influence fitness of different alleles of the same metabolic genes. It has been shown in Drosophila melanogaster, where diet quality affected fitness of the allelic variants of the “foraging” gene (Burns et al. 2012). In general, the patterns of 6Pgdh polymorphism could be associated with environmental quality, mediated by the relationship between soil properties and microbiome. However, resolving this issue would require more direct experimental verification.
Importantly, soil nutrients cannot solely explain the levels of 6Pgdh found in this study. The minerals or nutrients can explain the variation across space, but they cannot explain the variation across seasons. It’s because even though the primary nutrients (nitrogen, potassium, phosphorus) can vary between seasons (Hu et al. 2022), there is little evidence of such being the case for the other minor nutrients and minerals. The shifts in 6Pgdh levels might instead be a result of population demography and gene flow between local populations. Indeed, it has been suggested that bulbs are often colonized by small number of individuals or single gravid females, making founder effect an important determinant of genetic structuring. This could contribute to large differences and presumably erratic patterns of allele frequency changes in natural populations. Indeed, some of the population we have sampled were quite small, with high numbers of juvenile individuals, suggesting they had been founded recently. However, a recent field study found that colonization events are moderately common but, importantly, they do not seem to be associated with strong bottlenecks or founder effects (Przesmycka and Radwan 2023). A detailed study on the structure of genetic variation within and between bulb mite populations using genome-wide data would help to clarify this issue.
To conclude, the study shows that the 6Pgdh polymorphism is indeed common in bulb mites. We also found significant influence of soil properties on the polymorphism levels. Additionally, we found that the patterns of 6Pgdh polymorphism varied across locations and seasons, although there was no pattern to this change. Gene flow, driven by the migration of individuals between bulb mite populations, could be a crucial factor contributing to the observed variations in nature. In suspicion of a genotype-by-environment interaction for fitness, we looked at climatic variables such as temperature and precipitation, and their influence on the patterns of 6Pgdh, but we found no evidence of such. Similarly, the life-history assays performed in the lab did not provide any evidence of temperature influencing fitness of the allelic variants.
In summary, soil properties can potentially explain the distribution of 6Pgdh alleles of bulb mites in the wild, but not the spatio-temporal variation. Perhaps there are other environmental factors contributing to this variation or perhaps it’s the result of gene flow between populations. Complementing this study with additional experiments to test the effects of different factors on more traits (related to reproductive success) may help us understand more about how selection works in nature and about environment dependent balancing selection in general.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Begun DJ, Aquadro CF (1994) Evolutionary inferences from DNA variation at the 6-phosphogluconate dehydrogenase locus in natural populations of Drosophila: selection and geographic differentiation. Genetics 136(1):155–171. https://doi.org/10.1093/genetics/136.1.155
Błaś M, Ojrzyńska H (2024) The climate of Poland. In: Migoń P, Jancewicz K (eds) Landscapes and landforms of Poland. Springer International Publishing, Cham, pp 33–51. https://doi.org/10.1007/978-3-031-45762-3_3
Błażejczyk K (2006) Climate and bioclimate of Poland. In: Degórski M (ed) Natural and human environment of Poland. A geographical review. Polish Academy of Sciences, Stanislaw Leszycki Institute of Geography and Spatial Organization, Polish Geographical Society, Warsaw, pp 31–48
Brown KE, Kelly JK (2018) Antagonistic pleiotropy can maintain fitness variation in annual plants. J Evol Biol 31(1):46–56. https://doi.org/10.1111/jeb.13192
Burns JG, Svetec N, Rowe L, Mery F, Dolan MJ, Boyce WT, Sokolowski MB (2012) Gene–environment interplay in Drosophila melanogaster: chronic food deprivation in early life affects adult exploratory and fitness traits. Proc Natl Acad Sci USA 109(supplement_2):17239–17244. https://doi.org/10.1073/pnas.1121265109
Chen C, Du P, Zhang Z, Bao D (2023) 6-Phosphogluconate dehydrogenase inhibition arrests growth and induces apoptosis in gastric cancer via AMPK activation and oxidative stress. Open Life Sci 18(1):20220514. https://doi.org/10.1515/biol-2022-0514
de Moraes J, Franklin E, de Morais JW, de Souza JLP (2011) Species diversity of edaphic mites (Acari: Oribatida) and effects of topography, soil properties and litter gradients on their qualitative and quantitative composition in 64 km2 of forest in Amazonia. Exp Appl Acarol 55(1):39–63. https://doi.org/10.1007/s10493-011-9451-7
Di Bartolomeo F, Malina C, Campbell K, Mormino M, Fuchs J, Vorontsov E, Gustafsson CM, Nielsen J (2020) Absolute yeast mitochondrial proteome quantification reveals trade-off between biosynthesis and energy generation during diauxic shift. Proc Natl Acad Sci USA 117(13):7524–7535. https://doi.org/10.1073/pnas.1918216117
Díaz A, Okabe K, Eckenrode CJ, Villani MG, Oconnor BM (2000) Biology, ecology, and management of the bulb mites of the genus Rhizoglyphus (Acari: Acaridae). Exp Appl Acarol 24(2):85–113. https://doi.org/10.1023/A:1006304300657
Fahrendorf T, Ni W, Shorrosh BS, Dixon RA (1995) Stress responses in alfalfa (Medicago sativa L.) XIX. Transcriptional activation of oxidative pentose phosphate pathway genes at the onset of the isoflavonoid phytoalexin response. Plant Mol Biol 28(5):885–900. https://doi.org/10.1007/BF00042073
Ge T, Yang J, Zhou S, Wang Y, Li Y, Tong X (2020) The role of the pentose phosphate pathway in diabetes and cancer. Front Endocrinol 11:365. https://doi.org/10.3389/fendo.2020.00365
Gerson U, Thorens D (1982) Mating frequency as an indication of food quality for the bulb mite, Rhizoglyphus robini Claparède. Int J Invertebr Reprod 5(4):201–206. https://doi.org/10.1080/01651269.1982.10553470
Gerson U, Cohen E, Capua S (1991) Bulb mite, Rhizoglyphus robini (Astigmata: Acaridae) as an experimental animal. Exp Appl Acarol 12(1):103–110. https://doi.org/10.1007/BF01204403
González-Ruiz R, Leyva-Carrillo L, Peregrino-Uriarte AB, Yepiz-Plascencia G (2023) The combination of hypoxia and high temperature affects heat shock, anaerobic metabolism, and pentose phosphate pathway key components responses in the white shrimp (Litopenaeus vannamei). Cell Stress Chaperones 28(5):493–509. https://doi.org/10.1007/s12192-022-01265-1
Graffelman J, Weir BS (2016) Testing for Hardy-Weinberg equilibrium at biallelic genetic markers on the X chromosome. Heredity 116(6):558–568. https://doi.org/10.1038/hdy.2016.20
Hedrick PW (1999) Antagonistic pleiotropy and genetic polymorphism: a perspective. Heredity 82(2):126–133. https://doi.org/10.1038/sj.hdy.6884400
Höglund J, Sheldon BC, Hoglund J (1998) The cost of reproduction and sexual selection. Oikos 83(3):478. https://doi.org/10.2307/3546675
Hollman A, Tchounwou P, Huang H-C (2016) The association between gene-environment interactions and diseases involving the human GST superfamily with SNP variants. Int J Environ Res Public Health 13(4):379. https://doi.org/10.3390/ijerph13040379
Hou F-Y, Huang J, Yu S-L, Zhang H-S (2007) The 6-phosphogluconate dehydrogenase genes are responsive to abiotic stresses in rice. J Integr Plant Biol 49(5):655–663. https://doi.org/10.1111/j.1744-7909.2007.00460.x
Hu B, Xie M, Li H, Zhao W, Hu J, Jiang Y, Ji W, Li S, Hong Y, Yang M, Optiz T, Shi Z (2022) Stoichiometry of soil carbon, nitrogen, and phosphorus in farmland soils in southern China: spatial pattern and related dominates. CATENA 217:106468. https://doi.org/10.1016/j.catena.2022.106468
Jarzebowska M, Radwan J (2010) Sexual selection counteracts extinction of small populations of the bulb mites. Evolution 64(5):1283–1289. https://doi.org/10.1111/j.1558-5646.2009.00905.x
Kapahi P, Chen D, Rogers AN, Katewa SD, Li PW-L, Thomas EL, Kockel L (2010) With TOR, less is more: a key role for the conserved nutrient-sensing TOR pathway in aging. Cell Metab 11(6):453–465. https://doi.org/10.1016/j.cmet.2010.05.001
Kauffman FC, Brown JG, Passonneau JV, Lowry OH (1969) Effects of changes in brain metabolism on levels of pentose phosphate pathway intermediates. J Biol Chem 244(13):3647–3653. https://doi.org/10.1016/S0021-9258(18)83418-4
Kerwin R, Feusier J, Corwin J, Rubin M, Lin C, Muok A, Larson B, Li B, Joseph B, Francisco M, Copeland D, Weinig C, Kliebenstein DJ (2015) Natural genetic variation in Arabidopsis thaliana defense metabolism genes modulates field fitness. eLife 4:e05604. https://doi.org/10.7554/eLife.05604
Kilias G, Alahiotis SN (1985) Indirect thermal selection in Drosophila melanogaster and adaptive consequences. Theor Appl Genet 69–69(5–6):645–650. https://doi.org/10.1007/BF00251117
Konior M, Radwan J, Kołodziejczyk M, Keller L (2006) Strong association between a single gene and fertilization efficiency of males and fecundity of their mates in the bulb mite. Proc R Soc B Biol Sci 273(1584):309–314. https://doi.org/10.1098/rspb.2005.3302
Koshiba S, Motoike IN, Saigusa D, Inoue J, Aoki Y, Tadaka S, Shirota M, Katsuoka F, Tamiya G, Minegishi N, Fuse N, Kinoshita K, Yamamoto M (2020) Identification of critical genetic variants associated with metabolic phenotypes of the Japanese population. Commun Biol 3(1):662. https://doi.org/10.1038/s42003-020-01383-5
Kozyra M, Ingelman-Sundberg M, Lauschke VM (2017) Rare genetic variants in cellular transporters, metabolic enzymes, and nuclear receptors can be important determinants of interindividual differences in drug response. Genet Med 19(1):20–29. https://doi.org/10.1038/gim.2016.33
Krüger A, Grüning N-M, Wamelink MMC, Kerick M, Kirpy A, Parkhomchuk D, Bluemlein K, Schweiger M-R, Soldatov A, Lehrach H, Jakobs C, Ralser M (2011) The pentose phosphate pathway is a metabolic redox sensor and regulates transcription during the antioxidant response. Antioxid Redox Signal 15(2):311–324. https://doi.org/10.1089/ars.2010.3797
Kudureti A, Zhao S, Zhakyp D, Tian C (2023) Responses of soil fauna community under changing environmental conditions. J Arid Land 15(5):620–636. https://doi.org/10.1007/s40333-023-0009-4
Landi S, Capasso G, Esposito S (2021) Different G6PDH isoforms show specific roles in acclimation to cold stress at various growth stages of barley (Hordeum vulgare) and Arabidopsis thaliana. Plant Physiol Biochem 169:190–202. https://doi.org/10.1016/j.plaphy.2021.11.017
Leigh DM, Smallegange IM (2014) Effects of variation in nutrition on male morph development in the bulb mite Rhizoglyphus robini. Exp Appl Acarol 64(2):159–170. https://doi.org/10.1007/s10493-014-9822-y
Li G, Liu P, Zhao J, Su L, Zhao M, Jiang Z, Zhao Y, Yang X (2023) Correlation of microbiomes in “plant-insect-soil” ecosystem. Front Microbiol 14:1088532. https://doi.org/10.3389/fmicb.2023.1088532
Łukasiewicz A, Szubert-Kruszyńska A, Radwan J (2017) Kin selection promotes female productivity and cooperation between the sexes. Sci Adv 3(3):e1602262. https://doi.org/10.1126/sciadv.1602262
Łukasik P, Zygadło M, Radwan J (2010) The effect of a phosphogluconate dehydrogenase genotype on sperm competitiveness in the bulb mite, Rhizoglyphus robini. In: Sabelis MW, Bruin J (eds) Trends in acarology. Springer Netherlands, Dordrecht, pp 295–297. https://doi.org/10.1007/978-90-481-9837-5_47
Mérot C, Llaurens V, Normandeau E, Bernatchez L, Wellenreuther M (2020) Balancing selection via life-history trade-offs maintains an inversion polymorphism in a seaweed fly. Nat Commun 11(1):670. https://doi.org/10.1038/s41467-020-14479-7
Meyer UA, Zanger UM (1997) Molecular mechanisms of genetic polymorphisms of drug metabolism. Annu Rev Pharmacol Toxicol 37(1):269–296. https://doi.org/10.1146/annurev.pharmtox.37.1.269
Mukherjee D, Saha D, Acharya D, Mukherjee A, Chakraborty S, Ghosh TC (2018) The role of introns in the conservation of the metabolic genes of Arabidopsis thaliana. Genomics 110(5):310–317. https://doi.org/10.1016/j.ygeno.2017.12.003
Nielsen UN, Osler GHR, Campbell CD, Burslem DFRP, Van Der Wal R (2010) The influence of vegetation type, soil properties and precipitation on the composition of soil mite and microbial communities at the landscape scale: biogeography of mites and microbes. J Biogeogr 37(7):1317–1328. https://doi.org/10.1111/j.1365-2699.2010.02281.x
Oakeshott JG, Chambers GK, Gibson JB, Eanes WF, Willcocks DA (1983) Geographic variation in G6pd and Pgd allele frequencies in Drosophila melanogaster. Heredity 50(Pt 1):67–72. https://doi.org/10.1038/hdy.1983.7
Oostermeijer JGB, Van Eijck MW, Van Leeuwen NC, Den Nijs JCM (1995) Analysis of the relationship between allozyme heterozygosity and fitness in the rare Gentiana pneumonanthe L. J Evol Biol 8(6):739–759. https://doi.org/10.1046/j.1420-9101.1995.8060739.x
Parrett JM, Chmielewski S, Aydogdu E, Łukasiewicz A, Rombauts S, Szubert-Kruszyńska A, Babik W, Konczal M, Radwan J (2022) Genomic evidence that a sexually selected trait captures genome-wide variation and facilitates the purging of genetic load. Nat Ecol Evol 6(9):Article 9. https://doi.org/10.1038/s41559-022-01816-w
Pineda A, Kaplan I, Bezemer TM (2017) Steering soil microbiomes to suppress aboveground insect pests. Trends Plant Sci 22(9):770–778. https://doi.org/10.1016/j.tplants.2017.07.002
Plesnar-Bielak A, Skrzynecka AM, Prokop ZM, Radwan J (2012) Mating system affects population performance and extinction risk under environmental challenge. Proc R Soc B Biol Sci 279(1747):4661–4667. https://doi.org/10.1098/rspb.2012.1867
Plesnar-Bielak A, Skwierzyńska AM, Radwan J (2020) Sexual and ecological selection on a sexual conflict gene. J Evol Biol 33(10):1433–1439. https://doi.org/10.1111/jeb.13680
Powers DA, Ropson I, Brown DC, Beneden RV, Cashon R, Gonzalez-Villaseñor LI, DiMichele JA (1986) Genetic variation in Fundulus heteroclitus: geographic distribution. Am Zool 26(1):131–144
Procházka J, Brom J, Št’astný J, Pecharová E (2011) The impact of vegetation cover on temperature and humidity properties in the reclaimed area of a brown coal dump. Int J Min Reclam Environ 25(4):350–366. https://doi.org/10.1080/17480930.2011.623830
Przesmycka K, Radwan J (2023) Small-scale genetic structure of populations of the bulb mite Rhizoglyphus robini. Exp Appl Acarol 90(3–4):219–226. https://doi.org/10.1007/s10493-023-00807-1
Radwan J, Siva-jothy MT (1996) The function of post-insemination mate association in the bulb mite, Rhizoglyphus robini. Anim Behav 52(4):651–657. https://doi.org/10.1006/anbe.1996.0209
Rivera PC, Gardenal CN, Chiaraviglio M (2006) Sex-biased dispersal and high levels of gene flow among local populations in the argentine boa constrictor, Boa constrictor occidentalis. Austral Ecol 31(8):948–955. https://doi.org/10.1111/j.1442-9993.2006.01661.x
Rix G, Watkins-Dulaney EJ, Almhjell PJ, Boville CE, Arnold FH, Liu CC (2020) Scalable continuous evolution for the generation of diverse enzyme variants encompassing promiscuous activities. Nat Commun 11(1):5644. https://doi.org/10.1038/s41467-020-19539-6
Robinson MR, Pilkington JG, Clutton-Brock TH, Pemberton JM, Kruuk LEB (2006) Live fast, die young: trade-offs between fitness components and sexually antagonistic selection on weaponry in Soay sheep. Evolution 60(10):2168–2181. https://doi.org/10.1111/j.0014-3820.2006.tb01854.x
Rusuwa BB, Chung H, Allen SL, Frentiu FD, Chenoweth SF (2022) Natural variation at a single gene generates sexual antagonism across fitness components in Drosophila. Curr Biol 32(14):3161-3169.e7. https://doi.org/10.1016/j.cub.2022.05.038
Skwierzyńska AM, Plesnar-Bielak A (2018) Proximate mechanisms of the differences in reproductive success of males bearing different alleles of Pgdh—a gene involved in a sexual conflict in bulb mite. J Evol Biol 31(5):657–664. https://doi.org/10.1111/jeb.13250
Smallegange IM, Coulson T (2011) The stochastic demography of two coexisting male morphs. Ecology 92(3):755–764. https://doi.org/10.1890/09-2069.1
Stearns SC (1998) The evolution of life histories. Oxford University Press, Oxford. https://doi.org/10.1093/oso/9780198577416.001.0001
Stockwell CA, Mulvey M (1998) Phosphogluconate dehydrogenase polymorphism and salinity in the white sands pupfish. Evolution 52(6):1856. https://doi.org/10.2307/2411357
Tambasco-Studart M, Titiz O, Raschle T, Forster G, Amrhein N, Fitzpatrick TB (2005) Vitamin B6 biosynthesis in higher plants. Proc Natl Acad Sci USA 102(38):13687–13692. https://doi.org/10.1073/pnas.0506228102
Tilszer M, Antoszczyk K, Ssaek N, Zajaoc E, Radwan J (2006) Evolution under relaxed sexual conflict in the bulb mite Rhizoglyphus robini. Evolution 60(9):1868–1873. https://doi.org/10.1111/j.0014-3820.2006.tb00530.x
Vieille C, Zeikus GJ (2001) Hyperthermophilic enzymes: sources, uses, and molecular mechanisms for thermostability. Microbiol Mol Biol Rev 65(1):1–43. https://doi.org/10.1128/MMBR.65.1.1-43.2001
Watts SA, Lawrence JM (1990) The effect of reproductive state, temperature, and salinity on DNA and RNA levels and activities of metabolic enzymes of the Pyloric Ceca in the Sea Star Luidia clathrata (Say). Physiol Zool 63(6):1196–1215. https://doi.org/10.1086/physzool.63.6.30152640
Whitt SR, Wilson LM, Tenaillon MI, Gaut BS, Buckler ES (2002) Genetic diversity and selection in the maize starch pathway. Proc Natl Acad Sci USA 99(20):12959–12962. https://doi.org/10.1073/pnas.202476999
Zhang Z, Lv Y, Pan H (2013) Cooling and humidifying effect of plant communities in subtropical urban parks. Urban For Urban Green 12(3):323–329. https://doi.org/10.1016/j.ufug.2013.03.010
Zindel R, Ofek M, Minz D, Palevsky E, Zchori-Fein E, Aebi A (2013) The role of the bacterial community in the nutritional ecology of the bulb mite Rhizoglyphus robini (Acari: Astigmata: Acaridae). FASEB J 27(4):1488–1497. https://doi.org/10.1096/fj.12-216242
Acknowledgements
We thank the following people and institutions from providing us with plant material for sampling mites: Krystyna Pietrasik, Aleksandra Walczyńska, Marzena Marszałek, Ligia Kuriańska, Aneta Grząbka (“PZD Okręg Poznań” garden), Botanical Garden of Adam Mickiewicz University in Poznań, Joanna Łupkowska and Marian Ptak (“ROD Omex” garden), Ewa Majewska (Polish Association of Allotment Gardeners, Opole District), Sebastian Chmielewski, Marta Labocha-Derkowska, Michał Czarnecki (The Botanical Garden of Maria Currie-Skłodowska University in Lublin), University of Warsaw Botanic Garden, Krzysztof Kapała (Botanic Garden of the Jagiellonian University), Magdalena Pardon (Silesian Botanical Garden). We are also grateful to Genomics and Experimental Evolution Group members (Jagiellonian University) for their comments on the previous version of the manuscript. The study was funded by a grant from Polish National Science Centre Grant No UMO-2019/33/B/NZ8/02442 awarded to Agata Plesnar-Bielak.
Funding
The study was funded by a grant from Polish National Science Centre Grant No UMO-2019/33/B/NZ8/02442 awarded to Agata Plesnar-Bielak.
Author information
Authors and Affiliations
Contributions
Agata Plesnar-Bielak and Pranav Unnikrishnan designed the study, Pranav Unnikrishnan, Szymon Grzesik, and Magdalena Trojańska performed the experiments, Agata Plesnar-Bielak collected material in the field, Beata Klimek performed soil analyses, Pranav Unnikrishnan and Agata Plesnar-Bielak analysed the data, Pranav Unnikrishnan and Agata Plesnar-Bielak wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Unnikrishnan, P., Grzesik, S., Trojańska, M. et al. 6Pgdh polymorphism in wild bulb mite populations: prevalence, environmental correlates and life history trade-offs. Exp Appl Acarol 93, 115–132 (2024). https://doi.org/10.1007/s10493-024-00909-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-024-00909-4