Abstract
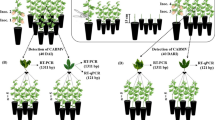
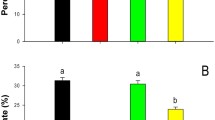
Citrus leprosis is the most important viral disease affecting citrus. The disease is caused predominantly by CiLV-C and is transmitted by Brevipalpus yothersi Baker mites. This study brings some insight into the colonization of B. yothersi in citrus [(Citrus × sinensis (L.) Osbeck (Rutaceae)] previously infested by viruliferous or non-viruliferous B. yothersi. It also assesses the putative role of shelters on the behavior of B. yothersi. Expression of PR1 and PR4 genes, markers of plant defense mechanisms, were evaluated by RT-qPCR to correlate the role of the plant hormonal changes during the tri-trophic virus-mite-plant interplay. A previous infestation with either non-viruliferous and viruliferous mites positively influenced oviposition and the number of adult individuals in the resulting populations. Mite populations were higher on branches that had received a previous mite infestation than branches that did not. There was an increase in the expression of PR4, a marker gene in the jasmonic acid (JA) pathway, in the treatment with non-viruliferous mites, indicating a response from the plant to their feeding. Conversely, an induced expression of PR1, a marker gene in the salicylic acid (SA) pathway, was observed mainly in the treatment with viruliferous mites, which suggests the activation of a plant response against the pathogen. The earlier mite infestation, as well as the presence of leprosis lesions and a gypsum mixture as artificial shelters, all fostered the growth of the B. yothersi populations after the second infestation, regardless of the presence or absence of CiLV-C. Furthermore, it is suggested that B. yothersi feeding actually induces the JA pathway in plants. At the same time, the CiLV-C represses the JA pathway and induces the SA pathway, which benefits the mite vector.





Similar content being viewed by others
References
Alberti G, Kitajima EW (2014) Anatomy and fine structure of Brevipalpus mites (Tenuipalpidae)-economically important plant-virus vectors. Zoologica 160:1–92
Andrade DJ, Falconi RS, Siqueira DS, Barbosa CL, Ferraudo AS, Oliveira CAL (2013) The influence of citrus rootstocks on the relationship between the mite Brevipalpus phoenicis and citrus leprosis disease. Pest Manag Sci 69:81–87. https://doi.org/10.1002/ps.3365
Andrade DJ, Lorençon JR, Siqueira DS, Novelli VM, Bassanezi RB (2018) Space–time variability of citrus leprosis as strategic planning for crop management. Pest Manag Sci 74:1798–1803. https://doi.org/10.1002/ps.4877
Arena GD, Ramos-González PL, Nunes MA, Ribeiro-Alves M, Camargo LE, Kitajima EW, Machado MA, Freitas-Astúa J (2016) Citrus leprosis virus C infection results in hypersensitive-like response, suppression of the JA/ET plant defense pathway and promotion of the colonization of its mite vector. Front Plant Sci 7:1757. https://doi.org/10.3389/fpls.2016.01757
Arena GD, Ramos-González PL, Rogerio LA, Ribeiro-Alves M, Casteel CL, Freitas-Astúa J, Machado MA (2018) Making a better home: modulation of plant defensive response by Brevipalpus mites. Front Plant Sci 9:1147. https://doi.org/10.3389/fpls.2018.01147
Arena GD, Ramos-González PL, Falk BW, Casteel CL, Freitas-Astúa J, Machado MA (2020) Plant immune system activation upon Citrus leprosis virus C infection is mimicked by the ectopic expression of the P61 viral protein. Front Plant Sci 11:1188. https://doi.org/10.3389/fpls.2020.01188
Bastianel M, Novelli VM, Kitajima EW, Kubo KS, Bassanezi RB, Machado MA, Freitas-Astúa J (2010) Citrus leprosis: centennial of an unusual mite–virus pathosystem. Plant Dis 94:284–292. https://doi.org/10.1094/PDIS-94-3-0284
Beers EH, Martinez-Rocha L, Talley RR, Dunley JE (2009) Lethal, sublethal, and behavioral effects of sulfur-containing products in bioassays of three species of orchard mites. J Econ Entomol 102:324–335. https://doi.org/10.1603/029.102.0143
Chabi-Jesus C, Ramos-González PL, Tassi AD, Guerra-Peraza O, Kitajima EW, Harakava R, Beserra JEA, Salaroli RB, Freitas-Astúa J (2018) Identification and characterization of citrus chlorotic spot virus, a new dichorhavirus associated with Citrus leprosis-like symptoms. Plant Dis 102:1588–1598. https://doi.org/10.1094/PDIS-09-17-1425-RE
Chabi-Jesus C, Ramos-González PL, Postclam-Barro M, Fontenele RS, Harakava R, Bassanezi RB, Freitas-Astúa J (2021) Molecular epidemiology of citrus leprosis virus C: a new viral lineage and phylodynamic of the main viral subpopulations in the Americas. Front Microbiol 29:12641252. https://doi.org/10.3389/fmicb.2021.641252
Chabi-Jesus C, Ramos-González PL, Tassi AD, Pereira LR, Bastianel M, Lau D, Canale MC, Harakava H, Novelli VM, Kitajima EW, Freitas-Astúa J (2023) Citrus bright spot virus: a new dichorhavirus, transmitted by Brevipalpus azores, causing citrus leprosis disease in Brazil. Plants 12:1371. https://doi.org/10.3390/plants12061371
Chiavegato LG (1986) Biologia do ácaro Brevipalpus phoenicis em citros. Pesqu Agropecu Bras 21:813–816
Cruz-Jaramillo JL, Ruiz-Medrano R, Rojas Morales L, López-Buenfil JA, Morales-Galván O, Chavarín-Palacio C, Ramírez-Pool JA, Xoconostle-Cázares B (2014) Characterization of a proposed Dichorhavirus associated with the citrus leprosis disease and analysis of the host response. Viruses 6:2602–2622. https://doi.org/10.3390/v6072602
de Lillo E, Freitas-Astúa J, Kitajima EW, Ramos-González PL, Simoni S, Tassi AD, Valenzano D (2021) Phytophagous mites transmitting plant viruses: update and perspectives. Entomol Gen 41:439–462. https://doi.org/10.1127/entomologia/2021/1283
Della Vechia JF, Bassanezi RB, Andrade DJ (2019) Physicochemical and biological compatibility of insecticide mixtures with acaricide in the management of Brevipalpus Yothersi. Syst Appl Acarol 24:1455–1464. https://doi.org/10.11158/saa.24.8.8
Eigenbrode SD, Bosque-Pérez NA, Davis TS (2018) Insect-borne plant pathogens and their vectors: ecology, evolution, and complex interactions. Annu Rev Entomol 63:169–191. https://doi.org/10.1146/annurev-ento-020117-043119
Freitas-Astúa J, Ramos-González PL, Arena GD, Tassi AD, Kitajima EW (2018) Brevipalpus-transmitted viruses: parallelism beyond a common vector or convergent evolution of distantly related pathogens? Curr Opin Virol 33:66–73. https://doi.org/10.1016/j.coviro.2018.07.010
Fundecitrus (2022) Final orange crop forecast update for the São Paulo and West-Southwest Minas Gerais citrus belt. 3rd. https://www.fundecitrus.com.br/pdf/pes_relatorios/0423_Final_Orange_Crop_Update.pdf Accessed 15 May 2023
Garcia A, Santamaria ME, Diaz I, Martinez M (2021) Disentangling transcriptional responses in plant defense against arthropod herbivores. Sci Rep. https://doi.org/10.1038/s41598-021-92468-6
Glas JJ, Alba JM, Simoni S, Villarroel CA, Stoops M, Schimmel BC, Schuurink RC, Sabelis MW, Kant MR (2014) Defense suppression benefits herbivores that have a monopoly on their feeding site but can backfire within natural communities. BMC Biol 12:1–14. https://doi.org/10.1186/s12915-014-0098-9
Green TR, Ryan CA (1972) Wound-induced proteinase inhibitor in plant leaves: a possible defense mechanism against insects. Science 175:776–777. https://doi.org/10.1126/science.175.4023.776
Haramoto FH (1966) Biology and control of Brevipalpus phoenicis (Geijskes) (Acarina: Tenuipalpidae). 105 f. Thesis-University of Hawaii, Honolulu
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8:R19. https://doi.org/10.1186/gb-2007-8-2-r19
Kant MR, Ament K, Sabelis MW, Haring MA, Schuurink RC (2004) Differential timing of spider mite-induced direct and indirect defenses in tomato plants. Plant Physiol 135:483–495. https://doi.org/10.1104/pp.103.038315
Kant MR, Jonckheere W, Knegt B, Lemos F, Liu J, Schimmel BCJ, Villarroel CA, Ataide LMS, Dermauw W, Glas JJ, Egas M, Janssen A, Van Leeuwen T, Schuurink RC, Sabelis MW, Alba JM (2015) Mechanisms and ecological consequences of plant defence induction and suppression in herbivore communities. Ann Bot 115:1015–1051. https://doi.org/10.1093/aob/mcv054
Klie M, Debener T (2011) Identification of superior reference genes for data normalisation of expression studies via quantitative PCR in hybrid roses (Rosa Hybrida). BMC Res Notes 28:4:518. https://doi.org/10.1186/1756-0500-4-518
Kubo KS, Novelli VM, Bastianel M, Locali-Fabris EC, Antonioli-Luizon R, Machado MA, Freitas-Astúa J (2011) Detection of Brevipalpus-transmitted viruses in their mite vectors by RT–PCR. Exp Appl Acarol 54:33–39. https://doi.org/10.1007/s10493-011-9425-9
Kunkel BN, Brooks DM (2002) Cross talk between signaling pathways in pathogen defense. Curr Opin Plant Biol 5:325–331. https://doi.org/10.1016/S1369-5266(02)00275-3
Mafra V, Kubo KS, Alves-Ferreira M, Ribeiro-Alves M, Stuart RM, Boava LP, Rodrigues CM, Machado MA (2012) Reference genes for accurate transcript normalization in citrus genotypes under different experimental conditions. PLoS ONE. https://doi.org/10.1371/journal.pone.0031263
Mafra V, Martins PK, Francisco CS, Ribeiro-Alves M, Freitas-Astúa J, Machado MA (2013) Candidatus Liberibacter americanus induces significant reprogramming of the transcriptome of the susceptible citrus genotype. BMC Genomics. https://doi.org/10.1186/1471-2164-14-247
Mineiro JLC, Sato ME (2019) Aspectos Comportamentais Da reprodução De Brevipalpus (Acari. Tenuipalpidae), em laboratório. O Biológico 81:1–13. https://doi.org/10.31368/1980-6221v81a10007
Mineiro JLC, Sato ME, Ochoa R, Beard J, Bauchan G (2018) Revisão taxonômica do ácaro Da leprose dos citros e sua distribuição no Brasil. Citrus Res Technol 39:1–11. https://doi.org/10.4322/crt.17147
Moreira RR, Machado FJ, Lanza FE, Trombin VG, Bassanezi RB, Miranda MP, Barbosa JC, Silva Junior GJ, Behlau F (2022) Impact of diseases and pests on premature fruit drop in sweet orange orchards in São Paulo state citrus belt, Brazil. Pest Manag Sci 78:2643–2656. https://doi.org/10.1002/ps.6894
Mysore KS, Ryu CM (2004) Nonhost resistance: how much do we know? Trends Plant Sci 9:97–104. https://doi.org/10.1016/j.tplants.2003.12.005
Pieterse CMJ, Van Der Does D, Zamioudis C, Leon-Reyes A, Van Wees SC (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Bi 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
Ramos-González PL, Chabi-Jesus C, Guerra-Peraza O, Tassi AD, Kitajima EW, Harakava R, Salaroli RB, Freitas-Astúa J (2017) Citrus leprosis virus N: a new Dichorhavirus causing citrus leprosis disease. Phytopathology 107:963–976. https://doi.org/10.1094/PHYTO-02-17-0042-R
Ramos-González PL, Chabi-Jesus C, Arena GD, Tassi AD, Kitajima EW, Freitas-Astúa J (2018) Citrus leprosis: a unique multietiologic disease. Citrus Americas 1:4–19
Rodrigues JCV, Kitajima EW, Childers CC, Chagas CM (2003) Citrus leprosis virus vectored by Brevipalpus Phoenicis (Acari: Tenuipalpidae) on citrus in Brazil. Exp Appl Acarol 30:161–179. https://doi.org/10.1023/b:appa.0000006547.76802.6e
Rodrigues VA, Bastianel MA, Kubo KA, Fadel AL, Nicolini F, Novelli VM, Freitas-Astúa J (2007) Desenvolvimento De um método para a otimização da transmissão experimental do vírus Da leprose dos citros. Laranja 28:29–38
Roy A, Choudhary N, Guillermo LM, Shao J, Govindarajulu A, Achor D, Wei G, Picton DD, Levy L, Nakhla MK, Hartung JS, Brlansky RH (2013) A novel virus of the genus Cilevirus causing symptoms similar to citrus leprosis. Phytopathology 103:488–500. https://doi.org/10.1094/PHYTO-07-12-0177-R
Roy A, Hartung JS, Schneider WL, Shao J, Leon G, Melzer MJ, Beard JJ, Otero-Colina G, Bauchan GR, Brlansky RH (2015) Role vending: complex relationships between viruses, hosts, and vectors related to Citrus Leprosis, and emerging disease. Phytopathology 105:1013–1025. https://doi.org/10.1094/PHYTO-12-14-0375-FI
Sarmento RA, Lemos F, Bleeker PM, Schuurink RC, Pallini A, Oliveira MGA, Lima ER, Kant M, Sabelis MW, Janssen A (2011) A herbivore that manipulates plant defence. Ecol Lett 14:229–236. https://doi.org/10.1111/j.1461-0248.2010.01575.x
SAS Institute (2015) SAS/IML® 14.1 User`s Guide. SAS Institute, Cary
Schmiesing A, Emonet A, Gouhier-Darimont C, Reymond P (2016) Arabidopsis MYC transcription factors are the target of hormonal salicylic acid/jasmonic acid cross talk in response to Pieris brassicae egg extract. Plant Physiol 170:2432–2443. https://doi.org/10.1104/pp.16.00031
Stark JD, Banks JE (2003) Population-level effects of pesticides and other toxicants on arthropods. Annu Rev Entomol 48:505–519. https://doi.org/10.1146/annurev.ento.48.091801.112621
Tang X, Wang X, Huang Y, Ma L, Jiang X, Rao MJ, Xu Y, Yin P, Yuan M, Deng X, Xu Q (2021) Natural variations of TFIIAγ gene and LOB1 promoter contribute to citrus canker disease resistance in Atalantia buxifolia. PLoS Genet. https://doi.org/10.1371/journal.pgen.1009316
Tassi AD, Garita-Salazar LC, Amorim L, Novelli VM, Freitas-Astúa J, Childers CC, Kitajima EW (2017) Virus–vector relationship in the citrus leprosis pathosystem. Exp Appl Acarol 71:227–241. https://doi.org/10.1007/s10493-017-0123-0
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Regul 19:195–216. https://doi.org/10.1007/s003440000026
Wu J, Baldwin IT (2010) New insights into plant responses to the attack from insect herbivores. Annual Rev Genet 44:1–24. https://doi.org/10.1146/annurev-genet-102209-163500
Zhang PJ, Li WD, Huang F, Zhang JM, Xu FC, Lu YB (2013) Feeding by whiteflies suppresses downstream jasmonic acid signaling by eliciting salicylic acid signaling. J Chem Ecol 39:612–619. https://doi.org/10.1007/s10886-013-0283-2
Zhao S, Fernald RD (2005) Comprehensive algorithm for quantitative real-time polymerase chain reaction. J Comput Biol 12:1047–1064. https://doi.org/10.1089/cmb.2005.12.1047
Zhao Y, Luo J, Xu S, Wang W, Liu T, Han C, Chen Y, Kong L (2016) Selection of reference genes for gene expression normalization in Peucedanum praeruptorum Dunn under abiotic stresses, hormone treatments and different tissues. PLoS ONE. https://doi.org/10.1371/journal.pone.0152356
Zhu J, Zhang L, Li W, Han S, Yang W, Liwang Q (2013) Reference gene selection for quantitative real-time PCR normalization in Caragana Intermedia under different abiotic stress conditions. PLoS One. https://doi.org/10.1371/journal.pone.0053196
Acknowledgements
The present study was accomplished with the support of the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Financing Code 001. The authors thank the National Council for Scientific and Technological Development (CNPq) for granting the research productivity scholarship (RBB, JFA and DJA) and the Fund for Citrus Protection (Fundecitrus) for support.
Author information
Authors and Affiliations
Contributions
ABPK, JFDV, TES, RBB, PLR-G, JF-A and DJA: wrote the main manuscript text. RBB, JF-A, PLR-G and DJA: Project administration, Investigation and Conceptualization. ABPK and JFDV: Investigation, and Formal analysis.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kapp, A.B.P., Vechia, J.F.D., Sinico, T.E. et al. Brevipalpus yothersi Baker (Tenuipalpidae) development in sweet orange plants is influenced by previous mite infestation and the presence of shelters. Exp Appl Acarol (2024). https://doi.org/10.1007/s10493-024-00903-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10493-024-00903-w