Abstract
In Germany, the knowledge about ticks infesting bats is limited, and is restricted only to a few studies, most of them dating back decades. To further improve our knowledge on ticks parasitising bats, healthy and sick bats in central Germany were examined for ticks. In total 519 larvae and one nymph of Carios vespertilionis were collected from nine bat species: Eptesicus nilssonii, Eptesicus serotinus, Myotis daubentonii, Myotis myotis, Nyctalus leisleri, Pipistrellus nathusii, Pipistrellus pygmaeus, Pipistrellus pipistrellus, and Vespertilio murinus. Either the presence of C. vespertilionis was new for some areas or it was confirmed in some federal states in central Germany. The infestation rate was mostly low (n = 1–5 larvae/bat). However, in two cases a high number of ticks was observed. The highest infestation of 97 C. vespertilionis larvae was recorded on one Parti-coloured bat (V. murinus).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ticks are obligate blood-feeding arthropods, with 950 living species grouped into three families: Ixodidae (726 species), Argasidae (223 species) and Nuttalliellidae (a single species) (Guglielmone et al. 2023; Mans et al. 2021). Argasidae (soft ticks) are more diverse in subtropical and tropical areas with more than 70 species being bat-specialists (Guglielmone et al. 2010). The family Argasidae is divided into two subfamilies: Argasinae and Ornithodorinae. Two soft tick species are known to regularly infest European bats: Carios vespertilionis is the most widespread species, whereas Secretargas transgariepinus is known from only a few locations in the central and western part of the Mediterranean Basin (Sandor et al. 2019). Carios vespertilionis, formerly known as Argas vespertilionis (Mans et al. 2021), is widely distributed between the Palearctic Region and South Africa. In total 42 host species were reported (Sándor et al. 2021). The main hosts are: common pipistrelle (Pipistrellus pipistrellus), Nathusius’s pipistrelle (P. nathusii), Kuhl’s pipistrelle (P. kuhlii), common noctule (Nyctalus noctula), whiskered bat (Myotis mystacinus), serotine bat (Eptesicus serotinus), and parti-coloured bat (Vespertilio murinus) (Sándor et al. 2021).
Carios vespertilionis (short-legged bat tick) and Argas reflexus (pigeon tick) are the only soft tick species in Germany and have been reported in many German Federal States. Carios vespertilionis has been reported for the first time in 1906 in Bremen-Vegesack, northern Germany, and is the only bat-associated soft tick (Voigt and Oudemanns 1906). There are only a few reports about its distribution in Walter (1992) summarised C. vespertilionis hosts (totally 12 bat species) in nine Federal States. In Bavaria (southern Germany), 11 bat species were reported as hosts for C. vespertilionis, with P. pipistrellus being the primary host (Rupp et al. 2004).
Carios vespertilionis parasitises bats in different types of transient colonies (e.g., maternal colonies) in all possible roost types, such as attics, burrows, hollow trees, and caves (Siuda et al. 2009). Carios vespertilionis can also attack humans and is considered ‘highly aggressive’ (Hoogstraal 1985; Estrada-Peña and Jongejan 1999). Jaenson et al. (1994) observed severe skin reactions with fever, ulceration, erythema, and edema on the legs and arms of two persons who had been bitten by C. vespertilionis nymphs, near Stockholm. This tick was reported as a vector for several pathogens such as viruses (e.g., tick-borne encephalitis virus, Issyk-Kul virus), bacteria (e.g., Coxiella burnetii, Ehrlichia, Rickettsia, and Borrelia species), and piroplasmids (Babesia spp.) (Sándor et al. 2021; Tompa et al. 2023).
In this study, we describe the distribution of C. vespertilionis in central Germany collected from bats caught with mist nets and sick or debilitated bats found on the ground.
Materials and methods
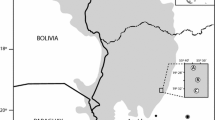
Ticks were collected from captured bats or bats delivered to animal rescue stations across six German federal states: Lower Saxony, Brandenburg, Saxony, North Rhine-Westphalia, Hesse, and Rhineland-Palatinate (Fig. 1). Ethical approval to catch bats had been issued by local Nature Conservation Authorities.
Bat ticks were collected from June to September 2021 and in August 2022, in six federal states in central Germany. Ticks were collected from bats caught with mist nets, and sick or debilitated bats coddled up in two volunteer bat rescue stations in Fulda city, eastern Hesse from 2002 to 2009 and in the urban region of Hanover, Lower Saxony, during summer 2022 and early 2023.
The bats were searched for ticks by brushing their fur with a small soft brush (to reduce the risk of possible infections of the handling person). The ticks were removed using fine-tipped tweezers and preserved in microtubes containing 70% ethanol, labelled according to each host. The ticks were identified using morphological characters according to Feider (1965).
Results
In total 519 C. vespertilionis larvae and one nymph were collected from 47 bats (Tables 1 and 2). Those bats belonged to nine species: Eptesicus nilssonii, E. serotinus, Myotis daubentonii, M. myotis, Nyctalus leisleri, Pipistrellus nathusii, P. pygmaeus, P. pipistrellus, and Vespertilio murinus. In the current study, P. pipistrellus was the dominant bat species from which C. vespertilionis larvae were collected. Generally, a few C. vespertilionis larvae were collected from bats, except for two cases: a P. nathusii was infested with 72 larvae and a V. murinus was infested with 97 larvae (Tables 1 and 2).
Discussion
In the Palearctic Region, bats are generally infested with a low number of ticks (Rupp et al. 2004; Hornok et al. 2017). In the UK, only 26 bats from the 7,606 submitted bats were infested with C. vespertilionis larvae (Lv et al. 2018). The distribution of C. vespertilionis in central Germany is known (Hutter et al. 2005) and results show that most of the examined bats did not carry any ticks or were only infested with a few ticks (n = 1–5 C. vespertilionis/bat). However, a few bats carried > 10 ticks. Nine P. pipistrellus were infested with 11, 13, 16, 28, 29 (2x), 32, 37, and 38 C. vespertilionis larvae, respectively. One P. nathusii bat was infested with 72 C. vespertilionis larvae. The highest number of larvae (n = 97) was collected from one V. murinus.
A study carried out in Bavaria (southern Germany) showed that generally, 1–2% of the bats is infested with ticks and the number of C. vespertilionis larvae found on 11 bat species averaged between 2 and 10 ticks (Rupp et al. 2004). There are few reports about bats infested with an uncommonly high number of ticks. Among 51 P. pipistrellus examined in Thuringia (Germany) in 2001/2002, three carried 12, 13, and 27 larvae of C. vespertilionis, respectively (Heddergott 2004). In total 231 C. vespertilionis larvae were collected from a young E. nilssonii in Lower Saxony (Germany), which was severely debilitated and died not long after. This was the highest number of tick larvae found on a bat in Germany (Walter and Rackow 2007).
It is unclear if sick bats are more likely to be infested by ticks or if high tick infestation causes the bats to become unwell. In the present study, the bat infested with 32 ticks appeared very weak, whereas the bat infested with 72 ticks appeared healthy. Ectoparasites such as ticks can cause short- and long-term health issues. Ticks may directly affect bats by sucking their blood, especially when highly infested. The direct impact associated with C. vespertilionis infestation are minor wounds in the bat’s skin evoked by tick bites that may cause physiological stress and inflammatory responses (del Cacho et al. 1994). Besides that, viruses and bacteria transmitted during the blood meal may have harmful effects on the bats’ health. Studies investigating C. vespertilionis (larvae, nymphs, and adults) collected from a wooden bat box harbouring P. pygmaeus in Sweden found 24% (22 of 92) of the C. vespertilionis specimens to be Borrelia-positive, with Borrelia sp. CPB1 (Jaenson and Wilhelmsson 2021), a spirochete found before in the UK (Evans et al. 2009) and in France (Socolovschi et al. 2012). Tompa et al. (2023) identified Rickettsia species, genetically closely related to Rickettsia parkeri, Rickettsia conorii, Rickettsia slovaca, Rickettsia sibirica subsp. mongolotimonae, Rickettsia rickettsii, and an uncultured Rickettsia sp. Several of these species are considered pathogenic to humans, nevertheless most of them are not usually circulating in European tick species. The Sweden studies from Jaenson and Wilhelmsson (2021) and Tompa et al. (2023) (using the same set of samples) concluded that further investigations are needed to confirm C. vespertilionis’ vector and/or reservoir capacity for the various pathogens. The aim of the current study was to gather more information about the distribution of C. vespertilionis on bats in Germany. Therefore, no studies on pathogens were planned and conducted. In the light of the Swedish results, future studies also have to focus on bacterial pathogens in bat soft tick to elucidate their potential to support the circulation of zoonotic pathogens.
During the present study, only larvae of C. vespertilionis were collected, except one nymph. Carios vespertilionis is frequently found in caves and its larvae are found on bats, with few exceptions. This can be explained by the long feeding time of larvae from a few days up to 14 or even 31 days. Nymphs and adult ticks, on the other hand, feed very quickly for only a short period of < 1 h (Walter and Kock 1985; Hoogstraal 1956; Walter 1992).
In a recent review (Sándor et al. 2021), 44 bat species were listed as hosts of soft ticks. Among them, C. vespertilionis had the most diverse host spectrum with 42 bat species in total. In the present study, C. vespertilionis larvae were found on a wide range of bat species, mainly on P. pipistrellus (common pipistrelle). These results support findings from previous studies where this bat species was identified as the main host in Germany. However, C. vespertilionis was also found on eight other bat species: N. leisleri (Leisler’s bat), E. nilssonii (northern bat), E. serotinus (serotine bat), M. daubentonii (Daubenton’s bat), M. myotis (greater mouse-eared bat), P. nathusii (Nathusius’s pipistrellus), P. pygmaeus (soprano pipistrelle) and V. murinus (parti-coloured bat). In Lv et al. (2018), of the nine bat species included in the study three species – P. pipistrellus, Plecotus auritus, and M. daubentoniid – carried C. vespertilionis larvae, with P. pipistrellus being the most infested. In Sweden, C. vespertilionis have been recorded from two bat species – E. nilssoni and P. pipistrellus – and from dog (Canis familiaris) (Jaenson et al. 1994), whereas in Norway, four bat species – M. mystacinus, M. daubentoni, P. pipistrellus, and E. nilssoni – are known as hosts of this tick (Mehl 1983).
In the latest overview about the distribution of C. vespertilionis in Germany (Rubel et al. 2023), no georeferenced data points were reported in Saxony and Saxony-Anhalt federal states, but one in Thuringia and several in Brandenburg and Mecklenburg-West Pomerania. Here, the presence of C. vespertilionis in eastern Saxony was documented, which fills the gap between central German Federal States and adjacent Poland, where this tick species has been reported (Siuda et al. 2009; Mierzyński et al. 2018).
The currently known distribution of C. vespertilionis in Germany has been confirmed for the following federal states: Lower Saxony (Scheffler and Hiller 2010), Brandenburg (Cornely and Schultz 1992), North Rhine-Westphalia, Hesse, and Rhineland-Palatinate (Walter 1992). When reporting C. vespertilionis for a specific location, the collection time should be considered, as some bats have a wide distribution area. Pipistrellus nathusii, for example, migrates long distances of up to 2,486 km in one direction (Vasenkov et al. 2022). Migrating bat species (e.g., N. leisleri, N. noctula, P. nathusii, V. murinus) can travel > 4000 km in total each year during their flights between their summer and winter habitats (Hutterer et al. 2005).
Regarding the winter activity of C. vespertilionis, there are only two reports on ticks found on bats. One was in January 1985 and the other in February 1987, both in Baden-Wuerttemberg, southern Germany (Walter 1992). On top of those reports a few specimens of C. vespertilionis were found in bat faeces. In the present study C. vespertilionis larvae were collected during the winter months (December, January, and February) in the federal states Hesse and Lower Saxony. Larvae, nymphs and adults were collected in summer (June, July, and August 2015 and 2018) using a water-filled tray placed bellow a wood bat box harbouring an estimated number of 250–500 adult females and young P. pygmaeus (Jaenson and Wilhelmsson 2021). In the UK, larvae and nymphs where collected from P. pipistrellus in May and July and in material from roost in house in July (Arthur 1963).
Conclusions
There are still considerable knowledge gaps regarding the presence of several tick species in Germany, especially concerning rare species, such as C. vespertilionis. This study supplies more detailed information for some federal states in central Germany, but additional studies are warranted on C. vespertilionis presence, density, and evaluation of public and veterinary risks.
References
Arthur DR (1963) British ticks. Butterworth &Co, London, p 213
Cornely M, Schultz U (1992) On the tick fauna of Eastern Germany. Angew Parasitol 33:173–183 (in German)
del Cacho E, Estrada-Peña A, Sanchez A, Serre J (1994) Histological response of Eptesicus serotinus (Mammalia: Chiroptera) to Argas vespertilionis (Acari: Argasidae). J Wildl Dis 30:340–345
Estrada-Peña A, Jongejan F (1999) Ticks feeding on humans: a review of records on human-biting Ixodoidea with special reference to pathogen transmission. Exp Appl Acarol 23:685–715
Evans NJ, Bown K, Timofte D, Simpson VR, Birtles R (2009) Fatal borreliosis in bat caused by relapsing fever spirochete, United Kingdom. Emerg Infect Dis 15:1331–1333
Feider Z (1965) Fasc. 2 Acaromorpha, Suprafamilia Ixodoidea (Căpuse). Fauna Republicii Populare Române. Arachnida. Editura Academiei Republicii Populare Române, Bucuresti, p 406
Guglielmone AA, Robbins RG, Apanaskevich DA, Petney TN, Estrada-Peña A, Horak IG, Shao R, Barker SC (2010) The Argasidae, Ixodidae and Nuttalliellidae (Acari: Ixodida) of the world: a list of valid species names. Zootaxa 2528:1–28. https://doi.org/10.5281/zenodo.196488
Guglielmone AA, Robbins RG, Nava S (2023) Geographic distribution of the hard ticks (Acari: Ixodida: Ixodidae) of the world by countries and territories. Zootaxa 5251(1):001–274
Heddergott M (2004) Hoher Befall mit Zeckenlarven (Argas vespertilionis) in einer Kolonie der Zwergfledermaus (Pipistrellus pipistrellus). Nyctalus 9:410–411
Hoogstraal H (1956) Argas vespertilionis. In: African Ixodidea I. Ticks of the Sudan (with special references to Equatoria Province and white preliminary reviews of the genera Boophilus, Margaropus, and Hyalomma). Washington DC, Department of the Navy, Bureau of Medicine and Surgery pp 1101
Hoogstraal H (1985) Argasid and Nuttalliellid ticks as parasites and vectors. Adv Parasitol 24:135–238. https://doi.org/10.1016/S0065-308X(08)60563-1
Hornok S, Szöke K, Tu VT, Konschán J, Takács N, Sándor AD, Halajian A, Földvári G, Estók P, Plantard O, Epis S, Görföl T (2017) Mitochondrial gene heterogeneity of the bat soft tick Argas vespertilionis (Ixodidae: Argasidae) in the Palaearctic. Parasit Vectors 10:109
Hutterer R, Ivanova T, Meyer-Cords C, Rodrigues L (2005) Bat migrations in Europe. A review of banding data and literature. Naturschutz und biologische Vielfalt, vol 28. Federal Agency for Nature Conservation, Bonn, p 180
Jaenson TGT, Wilhelmsson P (2021) First record of a suspected human-pathogenic Borrelia species in populations of the bat tick Carios vespertilionis in Sweden. Microorganisms 9:1100
Jaenson TG, Tälleklint L, Lundqvist L, Olsen B, Chirico J, Mejlon H (1994) Geographical distribution, host associations, and vector roles of ticks (Acari: Ixodidae, Argasidae) in Sweden. J Med Entomol 31:240–256
Lv J, Fernández de Marco MDM, Goharriz H, Phipps LP, McElhinney LM, Hernández-Triana LM, Wu S, Lin X, Fooks AR, Johnson N (2018) Detection of tick-borne bacteria and babesia with zoonotic potential in Argas (Carios) vespertilionis (Latreille, 1802) ticks from british. bats Sci Rep 8:1865
Mans BJ, Kelava S, Pienaar R, Featherstone J, de Castro MH, Quetglas J, Reeves WK, Durden LA, Miller MM, Laverty TM, Shao R, Takano A, Kawabata H, Moustafa MAM, Nakao R, Matsuno K, Greay TL, Evasco KL, Barker D, Barker SC (2021) Nuclear (18S-28S rRNA) and mitochondrial genome markers of Carios (Carios) vespertilionis (Argasidae) support Carios Latreille, 1796 as a lineage embedded in the ornithodorinae: re-classification of the Carios sensu Klompen and Oliver (1993) clade into its respective subgenera. Ticks Tick Borne Dis 12:101688
Mehl R (1983) The distribution and host relations of Norwegian ticks (Acari, Ixodides). Fauna Norv Ser B 30:46–51
Mierzyński L, Izdebska JN, Ciechanowska M (2018) New data on the distribution of Carios vespertilionis Latreille, 1802 (Ixodida, Argasidae) in bats (Chiroptera) from northern Poland. Ann Parasitiol 64:361–366
Rubel F, Zaenker S, Weigand A, Weber D, Chitimia-Dobler L, Kahl O (2023) Atlas of ticks (Acari:Argasidae, Ixodidae) in Germany: 1st data update. https://doi.org/10.1007/s10493-023-00784-5. Exp Appl Acarol
Rupp D, Zahn A, Ludwig P (2004) Actual records of bat ectoparasites in Bavaria (Germany). Spixiana 27:185–190
Sándor AD, Corduneanu A, Péter Á, Mihalca AD, Levente Barti L, Csősz I, Szőke K, Hornok S (2019) Bats and ticks: host selection and seasonality of bat–specialist ticks in eastern Europe. Parasit Vectors 12:605
Sándor AD, Mihalca AD, Domsa, Péter Á, Hornok A (2021) Argasid ticks of parlearctic bats: distribution, host selection, and zoonotic importance. Front Vet Sci 8:684737
Scheffler I, Hiller A (2010) Zur Ektoparasitenfauna der Fledermäuse in Niedersachsen: Neue Funde am Iberg bei Bad Grund. Nyctalus 15:309–317
Siuda K, Stanko M, Piksa K, Górz A (2009) Ticks (Acari: Ixodidae) parasitizing bats in Poland and Slovakia. Wiad Parazytologiczne 55:39–45
Socolovschi C, Kernif T, Raoult D, Parola P (2012) Borrelia, Rickettsia, and Ehrlichia species in bat ticks, France, 2010. Emerg Infect Dis 18:1966–1975
Tompa E, Jaenson TGT, Wilhelmsson P (2023) First records of possibly human pathogenic Rickettsia species in bat ticks, Carios vespertilionis, in Sweden. Microorganisms 11:357
Vasenkov D, Desmet J-F, Popov I, Sidorchuk N (2022) Bats can migrate farther than it was previously known: a new longest migration record by Nathusius’ pipistrelle Pipistrellus nathusii (Chiroptera: Vespertilionidae). Mammalia 86:524–526
Walter G (1992) Verbreitung und Biologie von Argas vespertilionis, Ixodes simplex und Ixodes ricinus (Ixodidae; Argasidae) bei Fledermäusen (Chiroptera) in der Bundesrepublik Deutschland. Myotis 30:123–132 (in German)
Walter G, Kock D (1985) Records of Ixodes vespertilionis, I. simplex and Argas vespertilionis (Ixodidae, Argasidae) from German bats (Chiroptera). Z Parasitenkd 71:107–111
Walter G, Rackow W (2007) Außergewöhnlich hoher Befall einer Nordfledermaus, Eptesicus nilssonii, mit der Lederzecke, Argas vespertilionis (Argasidae). Nyctalus 12:372–378 (in German)
Acknowledgements
We are thankful to Marika Fritzsche for English corrections. Saravanan Thangamani, SUNY Upstate Medical University, Syracuse, NY, for the helpfully reviewing and commenting on an earlier version of our manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
AF, SZ, JG, RK: feld study, tick collection, writing-review. CZ: map, writing-review. MB: writing-original draft, visualization, writing-review. LCD: conceptualization, tick determination, visualization, writing-review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
To avoid any misuse, the georeferenced data of the exact location where the bats were caught were not disclosed. Instead, the georeferenced data of the corresponding townships were reported.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fritzsche, A., Zaenker, S., Gottwald, J. et al. Distribution of the soft tick Carios vespertilionis in lowlands and low mountain regions of Germany. Exp Appl Acarol 91, 89–97 (2023). https://doi.org/10.1007/s10493-023-00822-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-023-00822-2