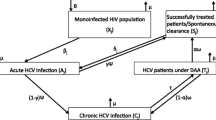
Abstract
Risk of HIV infection is high in Chinese MSM, with an annual HIV incidence ranging from 3.41 to 13.7/100 person-years. Tenofovir-based PrEP is effective in preventing HIV transmission in MSM. This study evaluates the epidemiological impact and cost-effectiveness of implementing PrEP in Chinese MSM over the next two decades. A compartmental model for HIV was used to forecast the impact of PrEP on number of infections, deaths, and disability-adjusted life years (DALY) averted. We also provide an estimate of the incremental cost-effectiveness ratio (ICER) and the cost per DALY averted of the intervention. Without PrEP, there will be 1.1–3.0 million new infections and 0.7–2.3 million HIV-related deaths in the next two decades. Moderate PrEP coverage (50%) would prevent 0.17–0.32 million new HIV infections. At Truvada’s current price in China, daily oral PrEP costs $46,813–52,008 per DALY averted and is not cost-effective; on-demand Truvada reduces ICER to $25,057–27,838 per DALY averted, marginally cost-effective; daily generic tenofovir-based regimens further reduce ICER to $3675–8963, wholly cost-effective. The cost of daily oral Truvada PrEP regimen would need to be reduced by half to achieve cost-effectiveness and realize the public health good of preventing hundreds of thousands of HIV infections among MSM in China.


Similar content being viewed by others
References
Fonner VA, Dalglish SL, Kennedy CE, Baggaley R, O’Reilly KR, Koechlin FM, et al. Effectiveness and safety of oral HIV preexposure prophylaxis for all populations. AIDS (London, England). 2016;30(12):1973–83.
Molina JM CC, Charreau I, et al., editor On demand PrEP with oral TDF/FTC in MSM: results of the ANRS Ipergay trial. In: Conference on retroviruses and opportunistic infections, Abstract 23LB; Seattle, Washington; 23–26 Feb 2015.
Global PrEP Landscape as of April 2018. 2018. https://www.prepwatch.org/wp-content/uploads/2018/04/PW_Global-Tracker_Apr2018.xlsx. Accessed 6 April 2018
Wilson C. Massive drop in London HIV rates may be due to internet drugs New Scientist. 2017 [updated January 9th, 2017; cited 2017 May 3rd]. https://www.newscientist.com/article/2117426-massive-drop-in-london-hiv-rates-may-be-due-to-internet-drugs/
Highleyman L. San Francisco reports new low in HIV infections and faster treatment, but disparities remain www.aidsmap.com2016. http://www.aidsmap.com/San-Francisco-reports-new-low-in-HIV-infections-and-faster-treatment-but-disparities-remain/page/3082266/. Accessed 5 Sept 2016
Smith AKJ. The Prep Effect—The Changing Landscape Of HIV Prevention In WA: Western Australia AIDS Council; 2018. http://waaids.com/item/765-the-prep-effect-the-changing-landscape-of-hiv-prevention-in-wa.html.
San Francisco Department of Public Health Population Health Division. HIV Epidemiology Annual Report 2017. San Francisco: San Francisco Department of Public Health Population Health Division; 2017.
AVAC. Regulatory status of Truvada for PrEP www.avac.org2018. https://www.avac.org/sites/default/files/infographics/truvada_status_april2018.jpg.
WHO. Guideline on when to start antiretroviral therapy and on pre-exposure prophylaxis for HIV. Geneva; 2015. p. 78.
Qin Q, Tang W, Ge L, Li D, Mahapatra T, Wang L, et al. Changing trend of HIV, Syphilis and Hepatitis C among men who have sex with men in China. Sci Rep. 2016;6:31081.
Xu JJ, Tang WM, Zou HC, Mahapatra T, Hu QH, Fu GF, et al. High HIV incidence epidemic among men who have sex with men in china: results from a multi-site cross-sectional study. Infect Dis Poverty. 2016;5(1):82.
Zhao T, Shi J, Chen F, Ding L, Li X, Zhang Y. Study on incidence of new HIV infection among men who have sex with men in Nanyang. Chin J AIDS STD. 2013;19:39–42.
Zhang W, Xu JJ, Zou H, Zhang J, Wang N, Shang H. HIV incidence and associated risk factors in men who have sex with men in Mainland China: an updated systematic review and meta-analysis. Sex Health. 2016;13:373.
Zhang Y, Peng B, She Y, Liang H, Peng HB, Qian HZ, et al. Attitudes toward HIV pre-exposure prophylaxis among men who have sex with men in western China. AIDS Patient Care STDS. 2013;27(3):137–41.
Zhou F, Gao L, Li S, Li D, Zhang L, Fan W, et al. Willingness to accept HIV pre-exposure prophylaxis among Chinese men who have sex with men. PLoS ONE. 2012;7(3):e32329.
Meyers K, Wu Y, Qian H, Sandfort T, Huang X, Xu J, et al. Interest in long-acting injectable PrEP in a cohort of men who have sex with men in China. AIDS Behav. 2017;1–11.
Ding Y, Yan H, Ning Z, Cai X, Yang Y, Pan R, et al. Low willingness and actual uptake of pre-exposure prophylaxis for HIV-1 prevention among men who have sex with men in Shanghai, China. Biosci Trends. 2016;10(2):113–9.
Zhang Y, Peng B, She Y, Liang H, Peng H-B, Qian H-Z, et al. Attitudes toward HIV pre-exposure prophylaxis among men who have sex with men in western China. AIDS Patient Care STDs. 2013;27(3):137–41.
Wang Z, Lau JTF, Fang Y, Ip M, Gross DL. Prevalence of actual uptake and willingness to use pre-exposure prophylaxis to prevent HIV acquisition among men who have sex with men in Hong Kong, China. PLoS ONE. 2018;13(2):e0191671.
Jackson T, Huang A, Chen H, Gao X, Zhong X, Zhang Y. Cognitive, psychosocial, and sociodemographic predictors of willingness to use HIV pre-exposure prophylaxis among Chinese men who have sex with men. AIDS Behav. 2012;16(7):1853–61.
Desai K, Sansom SL, Ackers ML, Stewart SR, Hall HI, Hu DJ, et al. Modeling the impact of HIV chemoprophylaxis strategies among men who have sex with men in the United States: HIV infections prevented and cost-effectiveness. Aids. 2008;22(14):1829–39.
Paltiel AD, Freedberg KA, Scott CA, Schackman BR, Losina E, Wang B, et al. HIV preexposure prophylaxis in the United States: impact on lifetime infection risk, clinical outcomes, and cost-effectiveness. Clin Infect Dis. 2009;48(6):806–15.
Koppenhaver RT, Sorensen SW, Farnham PG, Sansom SL. The cost-effectiveness of pre-exposure prophylaxis in men who have sex with men in the United States: an epidemic model. JAIDS J Acquir Immune Defic Syndr. 2011;58(2):e51–2.
Gomez GB, Borquez A, Caceres CF, Segura ER, Grant RM, Garnett GP, et al. The potential impact of pre-exposure prophylaxis for HIV prevention among men who have sex with men and transwomen in Lima, Peru: a mathematical modelling study. PLoS Med. 2012;9(10):e1001323.
Juusola JL, Brandeau ML, Owens DK, Bendavid E. The Cost-effectiveness of preexposure prophylaxis for HIV prevention in men who have sex with men in the United States. Ann Intern Med. 2012;156(8):541–50.
Cremin I, Alsallaq R, Dybul M, Piot P, Garnett G, Hallett TB. The new role of antiretrovirals in combination HIV prevention: a mathematical modelling analysis. Aids. 2013;27(3):447–58.
Chen A, Dowdy DW. Clinical effectiveness and cost-effectiveness of HIV pre-exposure prophylaxis in men who have sex with men: risk calculators for real-world decision-making. PLoS ONE. 2014;9(10):e108742.
Schneider K, Gray RT, Wilson DP. A cost-effectiveness analysis of HIV preexposure prophylaxis for men who have sex with men in Australia. Clin Infect Dis. 2014;58(7):1027–34.
Drabo EF, Hay JW, Vardavas R, Wagner ZR, Sood N. A cost-effectiveness analysis of pre-exposure prophylaxis for the prevention of HIV among Los Angeles County men who have sex with men. Clin Infect Dis. 2016;63:ciw578.
Glaubius RL, Hood G, Penrose KJ, Parikh UM, Mellors JW, Bendavid E, et al. Cost-effectiveness of injectable preexposure prophylaxis for HIV prevention in South Africa. Clin Infect Dis. 2016;63:539.
Jenness SM, Goodreau SM, Rosenberg E, Beylerian EN, Hoover KW, Smith DK, et al. Impact of the centers for disease control’s HIV preexposure prophylaxis guidelines for men who have sex with men in the United States. J Infect Dis. 2016;214:1800.
MacFadden DR, Tan DH, Mishra S. Optimizing HIV pre-exposure prophylaxis implementation among men who have sex with men in a large urban centre: a dynamic modelling study. J Int AIDS Soc. 2016;19(1):20791.
Mitchell KM, Prudden HJ, Washington R, Isac S, Rajaram SP, Foss AM, et al. Potential impact of pre-exposure prophylaxis for female sex workers and men who have sex with men in Bangalore, India: a mathematical modelling study. J Int AIDS Soc. 2016;19(1):20942.
Nichols BE, Boucher CA, van der Valk M, Rijnders BJ, van de Vijver DA. Cost-effectiveness analysis of pre-exposure prophylaxis for HIV-1 prevention in the Netherlands: a mathematical modelling study. Lancet Infect Dis. 2016;16:1423.
Punyacharoensin N, Edmunds WJ, De Angelis D, Delpech V, Hart G, Elford J, et al. Effect of pre-exposure prophylaxis and combination HIV prevention for men who have sex with men in the UK: a mathematical modelling study. Lancet HIV. 2016;3(2):e94–104.
Ross EL, Cinti SK, Hutton DW. Implementation and operational research: a cost-effective, clinically actionable strategy for targeting HIV preexposure prophylaxis to high-risk men who have sex with men. J Acquir Immune Defic Syndr. 2016;72(3):e61–7.
Walensky RP, Jacobsen MM, Bekker L-G, Parker RA, Wood R, Resch SC, et al. Potential clinical and economic value of long-acting preexposure prophylaxis for South African women at high-risk for HIV infection. J Infect Dis. 2016;213(10):1523–31.
Cremin I, McKinnon L, Kimani J, Cherutich P, Gakii G, Muriuki F, et al. PrEP for key populations in combination HIV prevention in Narobi: a mathematical modelling study. Lancet HIV. 2017;4:e214.
Schackman BR, Eggman AA. Cost-effectiveness of pre-exposure prophylaxis for HIV: a review. Curr Opin HIV AIDS. 2012;7(6):587–92.
Adamson BJS, Carlson JJ, Kublin JG, Garrison LP. The potential cost-effectiveness of pre-exposure prophylaxis combined with HIV vaccines in the United States. Vaccines. 2017;5(2):13.
Zhang L, Phanuphak N, Henderson K, Nonenoy S, Srikaew S, Shattock AJ, et al. Scaling up of HIV treatment for men who have sex with men in Bangkok: a modelling and costing study. Lancet HIV. 2015;2(5):e200–7.
Eaton JW, Menzies NA, Stover J, Cambiano V, Chindelevitch L, Cori A, et al. Health benefits, costs, and cost-effectiveness of earlier eligibility for adult antiretroviral therapy and expanded treatment coverage: a combined analysis of 12 mathematical models. Lancet Global Health. 2014;2(1):e23–34.
Muessig KE, Tucker JD, Wang B-X, Chen X-S. HIV and syphilis among men who have sex with men in China: the time to act is now. Sex Transm Dis. 2010;37(4):214–6.
Zhang L, Chow EP, Wilson DP. Distributions and trends in sexual behaviors and HIV incidence among men who have sex with men in China. BMC Public Health. 2012;12:546.
Chow EP, Wilson DP, Zhang L. Patterns of condom use among men who have sex with men in China: a systematic review and meta-analysis. AIDS Behav. 2012;16(3):653–63.
Cai Y, Wang Z, Lau JT, Li J, Ma T, Liu Y. Prevalence and associated factors of condomless receptive anal intercourse with male clients among transgender women sex workers in Shenyang, China. J Int AIDS Soc. 2016;19(3Suppl 2):20800.
Zou H, Zhang L, Chow EPF, Tang W, Wang Z. Testing for HIV/STIs in China: challenges, opportunities, and innovations. Biomed Res Int. 2017;2017:3.
Chow EPF, Wilson DP, Zhang L. The rate of HIV testing is increasing among men who have sex with men in China. HIV Med. 2012;13(5):255–63.
Zhang F, Dou Z, Ma Y, Zhang Y, Zhao Y, Zhao D, et al. Effect of earlier initiation of antiretroviral treatment and increased treatment coverage on HIV-related mortality in China: a national observational cohort study. Lancet Infect Dis. 2011;11(7):516–24.
Cohen, Gay CL. Treatment to prevent transmission of HIV-1. Clin Infect Dis. 2010;50(Supplement_3):S85–95.
Zhang L, Regan DG, Chow EPF, Gambhir M, Cornelisse V, Grulich A, et al. Neisseria gonorrhoeae transmission among men who have sex with men: an anatomical site-specific mathematical model evaluating the potential preventive impact of mouthwash. Sex Transm Dis. 2017;44(10):586–92.
Garnett GP, Mertz KJ, Finelli L, Levine WC, St Louis ME. The transmission dynamics of gonorrhoea: modelling the reported behaviour of infected patients from Newark, New Jersey. Philos Trans R Soc Lond B. 1999;354(1384):787–97.
Mills HL, Riley S. The spatial resolution of epidemic peaks. PLoS Comput Biol. 2014;10(4):e1003561.
van der Steen A, van Rosmalen J, Kroep S, van Hees F, Steyerberg EW, de Koning HJ, et al. Calibrating parameters for microsimulation disease models: a review and comparison of different goodness-of-fit criteria. Med Decis Mak. 2016;36(5):652–65.
Whitfield THF, John SA, Rendina HJ, Grov C, Parsons JT. Why i quit pre-exposure prophylaxis (PrEP)? A mixed-method study exploring reasons for PrEP discontinuation and potential re-initiation among gay and bisexual men. AIDS Behav. 2018. https://doi.org/10.1007/s10461-018-2045-1
Elsesser SA, Oldenburg CE, Biello KB, Mimiaga MJ, Safren SA, Egan JE, et al. Seasons of risk: anticipated behavior on vacation and interest in episodic antiretroviral pre-exposure prophylaxis (PrEP) among a large national sample of U.S. men who have sex with men (MSM). AIDS Behav. 2016;20(7):1400–7.
Carlo Hojilla J, Koester KA, Cohen SE, Buchbinder S, Ladzekpo D, Matheson T, et al. Sexual behavior, risk compensation, and HIV prevention strategies among participants in the San Francisco PrEP demonstration project: a qualitative analysis of counseling notes. AIDS Behav. 2016;20(7):1461–9.
Underhill K, Guthrie KM, Colleran C, Calabrese SK, Operario D, Mayer KH. Temporal fluctuations in behavior, perceived HIV risk, and willingness to use pre-exposure prophylaxis (PrEP). Arch Sex Behav. 2018.
Tan-Torres T, Edejer RB, Adam T, Hutubessy R, Acharya A, Evans DB, Murray CJL. WHO guide to cost-effectiveness analysis. Geneva; 2003.
WHO. GDP per capita (current US$) 2016. http://data.worldbank.org/indicator/NY.GDP.PCAP.CD?end=2015&locations=CN&start=1960&view=chart.
Jia Z, Mao Y, Zhang F, Ruan Y, Ma Y, Li J, et al. Antiretroviral therapy to prevent HIV transmission in serodiscordant couples in China (2003–11): a national observational cohort study. The Lancet. 2013;382(9899):1195–203.
MedsPaL Database. Patent Card for China 2004. http://www.medspal.org/patent/?uuid=c61535aa-76ad-4bdb-92c6-7445b24071b9.
Krakower D, Ware N, Mitty JA, Maloney K, Mayer KH. HIV providers’ perceived barriers and facilitators to implementing pre-exposure prophylaxis in care settings: a qualitative study. AIDS Behav. 2014;18(9):1712–21.
Marcus JL, Volk JE, Pinder J, Liu AY, Bacon O, Hare CB, et al. Successful implementation of HIV preexposure prophylaxis: lessons learned from three clinical settings. Curr HIV/AIDS Rep. 2016;13(2):116–24.
Liu AY, Cohen SE, Vittinghoff E, Anderson PL, Doblecki-Lewis S, Bacon O, et al. Preexposure prophylaxis for HIV infection integrated with municipal-and community-based sexual health services. JAMA Int Med. 2016;176(1):75–84.
Sun X, Lu F, Wu Z, Poundstone K, Zeng G, Xu P, et al. Evolution of information-driven HIV/AIDS policies in China. Int J Epidemiol 2010;39(suppl 2):ii4–ii13.
Lim SH, Christen CL, Marshal MP, Stall RD, Markovic N, Kim KH, et al. Middle-aged and older men who have sex with men exhibit multiple trajectories with respect to the number of sexual partners. AIDS Behav. 2012;16(3):590–8.
Baral SD, Poteat T, Strömdahl S, Wirtz AL, Guadamuz TE, Beyrer C. Worldwide burden of HIV in transgender women: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13(3):214–22.
Volk JE, Marcus JL, Phengrasamy T, Blechinger D, Nguyen DP, Follansbee S, et al. No new HIV infections with increasing use of HIV preexposure prophylaxis in a clinical practice setting. Clin Infect Dis. 2015;61(10):1601–3.
Acknowledgements
The authors wish to thank Dr. Junjie Xu (The First Affiliated Hospital, China Medical University in Shenyang), Dr. Xia Li (Yunnan AIDS Care Center in Kunming), Dr. Yinzhong Shen (Shanghai Public Health Clinical Center) and Dr. Junwei Su (First Affiliated Hospital, Zhejiang University, Hangzhou) for providing pricing information for routine screening tests and costs of ARV drugs. Finally, we thank the anonymous reviewer whose insightful comments were critical to shaping the final manuscript.
Funding
HW and XH contributions are supported by the Chinese Government 13th Five-Year Plan (2017ZX10201101-001-002), and the National Natural Science Foundation of China (No. 81701984). KM is supported by Grant # UL1TR001866 from the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health (NIH) Clinical and Translational Science Award (CTSA) program.
Author information
Authors and Affiliations
Contributions
LZ and KM designed the overall framework for the analysis; KM, YW, LZ, and PP conducted literature reviews and consulted expert opinion to generate assumptions for inputs into the model; LZ and PP created the mathematic model and did the analysis; XH, HW, and MM gave overall feedback to the inputs into model and provided critical insights for framing of the discussion. XM and NNS contributed in checking the key findings, generating supplementary figures and revising the manuscript. KM, YW, and LZ wrote the manuscript. All authors reviewed drafts and contributed to the overall final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
MM receives research support in the form of grants from GSK, ViiV, Merck, and Gilead. He is a consultant to Merck and GSK and participates in the speaker bureau for Gilead Sciences. KM receives research support from a grant from GSK. No other authors report any conflict of interest.
Research Involving Human and Animal Participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, L., Peng, P., Wu, Y. et al. Modelling the Epidemiological Impact and Cost-Effectiveness of PrEP for HIV Transmission in MSM in China. AIDS Behav 23, 523–533 (2019). https://doi.org/10.1007/s10461-018-2205-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-018-2205-3