Abstract
Soils provide essential ecosystem services for the existence of ecosystems and biodiversity. It is crucial to understand their quality through the evaluation of ecological processes. However, only some studies estimate the effectiveness of ecosystem restoration based on evaluating soil quality (SQ) indicators. This research evaluated the five most common land-use scenarios in Andean ecosystems within the Natural Reserve of the Civil Society (RNSC) “La Montaña Mágica” under natural forest, coffee plantation, badlands, and active and passive restoration. The main objective was to analyze the physical, chemical, and biological characteristics concerning land use, establish the baseline for SQ indicators in different land use activities, and determine the status of restoration systems for other land uses in the study site. ANOVA and Dunnett's test evaluated SQ parameters. In passive and active restoration, soil bulk density, porosity, and the number of individuals and families of macroinvertebrates were improved. The latter presented low pH and aluminum values but increased potassium compared to other soil uses. The restoration strategies favored changes in SQ indicators due to the contribution of organic carbon, a developed root system, and the recirculation of nutrients in the edaphic system. This study provided information on changes in SQ with soil usage as a practical tool to evaluate ecological restoration methods in natural areas of the eastern Colombian Andes.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Soil is a dynamic environment where biological, chemical, and physical components have complex interactions (Delgado and Gomez 2016) and are closely interrelated with the productivity and sustainability of the system. Most of its attributes directly or indirectly influence its quality (SQ) and biological production (De Deyn and Kooistra 2021; Ghorai et al. 2023). To effectively and sustainably improve soil quality and SOC sequestration, recommended practices should be region-specific and adapted to local pedoclimatic conditions (Amelung et al. 2020; Certini and Scalenghe 2023). Soil also provides and regulates ecosystem services that depend on formative factors, such as original parent material, relief, biota, weather, and climate, which affect it, as well as its management (Pereira et al. 2018; Vandermeer 2011; Certini and Scalenghe 2023). Drobnik et al. (2018), Diti et al. (2020), and Machado et al. (2019) propose to develop a soil quality index based on ecosystem services and their functions.
The conditions, distribution, and composition of plants respond to pH and nutrient availability (Bartelheimer and Poschlod 2016), and the integration of trees in productive systems improve SQ and nutrient availability; since increasing soil nutrient thresholds is a cost-effective and fast-efficient tool to assess SQ (Kisaka et al. 2023).
Soil services and their biota can sustain ecosystem biodiversity and support species heterogeneity and the presence of some unique species while providing genetic diversity several orders of magnitude greater than that found above ground (Parker 2010; Cooke et al. 2023). The abundance and diversity of the other organisms are generally positive for SQ and act as a buffer against climate change (Certini and Scalenghe 2023).
Soils are severely threatened, affecting yield and plant composition (Drobnik et al. 2018; Diti et al. 2020), and their rates of formation and recovery are far from balancing current rates of loss and degradation (Bindraban et al. 2012; Pulleman et al. 2012; Machado et al. 2019). Current practices of conservation and restoration of fragile ecosystems are an alternative to recover productivity through increased water and nutrient use efficiency, while the benefits that could be obtained from this restoration have rarely been evaluated (Zhao et al. 2005; Tittonell et al. 2012; Lewis et al. 2022). Such benefits are a renewed impetus to restore and return forests to their ecological function, protect species, sequester carbon, and secure livelihoods (Banin et al. 2022). The implementation of restoration programs in tropical environments restores soil biology and soil organic carbon and favors the early recovery of microbial communities (Bonner et al. 2020).
On the other hand, protected areas are a key conservation tool, although their effectiveness in maintaining biodiversity over time has yet to be quantified (Cooke et al. 2023). Global efforts are needed to define sustainable practices applicable in specific agroecological zones and food landscapes (Rumpel et al. 2023). Betancur-Corredor et al. (2023), Delgado et al. (2019), Lewis et al. (2022), Martinez and Zinck (2004) and Martinez et al. (2020), underline the difficulty and necessity of restoring degraded soils in the Andes and tropical savannas and the possibility of using microorganisms, reactivation of biochemical fluxes, transformation of degraded areas into technosoils or through other biotechnological alternatives Barrios et al. (2006) proposed technical indicators of agroecosystem change for farmers to assess unobservable changes in soil properties before they lead to more severe and visible forms of degradation. The use and evaluation of physical, chemical, and biological soil indicators based on land use dynamics in Colombia have been implemented in grazing systems by Teutscherová et al. (2021), degraded areas, agricultural and forestry systems (Velazquez et al. 2007; Machado et al. 2019), and forest restoration (Lozano-Baez et al. 2021). However, an integrated SQ indicator in different use systems is still lacking. The main objective of the present study was to evaluate SQ as an indicator of natural restoration and soil remediation. It established a baseline for physical, chemical, and biological indicators of Soil Quality under the five most common use scenarios in Andean ecosystems.
Scope
To evaluate soil quality as an indicator of recovery and natural restoration processes in the Civil Society Natural Reserve “La Montaña Mágica-El Poleo” (MMEP) in Zapatoca, Santander, Colombian Andes. Second, to analyze the soil's physical, chemical, and biological characteristics to establish its quality according to the land uses.
Methodology
Soil quality status was determined from three types of analysis: chemical, physical and biological. Five coverages were sampled: secondary forest (SF), natural regeneration (NR), active restoration (AR), cropland (CL), and degraded soil (DS) at the Civil Society Nature Reserve (CSNR) “La Montaña Mágica-El Poleo” at Zapatoca (Santander), based on Aguilar-Garavito and Ramírez (2015), Arshad and Martin (2002), Instituto Geografico Agustin Codazzi (IGAC) et al. (2006) and Pérez (2010) methodologies.
Study area
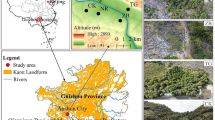
The CSNR is located in Zapatoca, Santander, Colombia (Fig. 1) in the buffer zone of the Serranía de los Yariguíes National Natural Park (06°50′05.4″ N latitude and 73°18′06.3″ W longitude). It is an 86.1 ha of premontane rainforest area between 1850 and 2300 m above sea level. Precipitation reaches 4000 mm y-1, with a bimodal regime (Espinal and Montenegro 1977). The mountains have steep to moderately steep slopes (slope > 50%) and moderate to severe erosion; soils derived from sandstones and shales are moderately acidic. The predominant soils have low pedogenetic development and, according to SSS (2022a) and WRB (2022), correspond to Typic Dystrudepts Sandy Loam Mixed Subactive Acid Hyperthermic (CAMBISOLS Endoleptic Dystric Arenic Loamic Ochric Raptic), Lithic Quartzipsamments Sandy Loam Mixed Subactive Acid Hyperthermic (REGOSOLS Epileptic Yermic Dystric Arenic Loamic Ochric) and Humic Psammentic Dystrudepts Sandy Loam Mixed Subactive Acid Hyperthermic (CAMBISOLS Dystric Arenic Loamic Ochric Raptic).
Experiment design
Soils were studied in secondary forest (SF), natural regeneration (NR), active restoration (AR), cropland (CL), and degraded soils (DS) with 25, 11, 6, 25, and 25 years of age, respectively. Sampling was carried out from October 2021 to February 2022 in five coverages with four replicates per coverage in composite samples (10 subsamples) after removing the surface layer of vegetation and leaf litter (sensu IGAC et al. 2006). Each sample was homogenized and packed in a polyethylene bag for transport to the laboratory (Aguilar-Garavito and Ramírez 2015).
Chemical, physical and biological properties were analyzed at the Soil Science Laboratory—National University of Colombia at Medellin Campus. The status of each cover was estimated through 19 selected soil quality indicators.
Chemical, physical and biological characterization
Chemical analysis
We determined the following parameters: pH, organic carbon (OC), nitrogen (N), phosphorus (P), aluminum (Al), calcium (Ca), magnesium (Mg) and potassium (K) and cation exchange capacity in meq/100 g soil (CEC) using the methodologies of SSS (2022b); the main methods are summarized in Table 1.
Physical análisis
Bulk density in (Bd), % porosity (Po), cm/hr infiltration (I), cm/hr percolation (Pe), penetration resistance (Pr), and texture (%) were determined (see Table 2).
Biological analysis
Macrofauna was collected in 1 m2 per sample, the litter layer was removed, and the surface layer (5 cm thick) was extracted, collected, and deposited on a 5 × 5 mm sieve for approximately three minutes (Krell et al. 2005). Each sample was mounted in a Winkler bag in the laboratory for 48 h. Macroinvertebrates were cleaned and stored in plastic containers with alcohol, individuals were morphologically described, and taxon assignment (family level) was based on morphological diagnosis (Krell et al. 2005) with a 30X Leica ES2 ® stereoscope. The result is expressed in the number of individuals per m2 (In) and families per m2 (Fn).
Statistical analysis
Statistical analyses were performed with R software. The Gaussian assumptions of normality and homoscedasticity were tested with the Shapiro–Wilk test (Shapiro.test) and the Levene test (Levene test) with a significance level of 0.01%. When these were not met, transformations were performed. A principal component analysis was performed to approximate the data's nature and a correlation matrix (corrplot) to know the relationships between parameters of different natures (physical, chemical, and biological). A one-way analysis of variance (ANOVA, aov) was applied to determine which hedges differed from Dunnet's test (DunnettTest). Both tests were analyzed at a significance level of 0.05% (null hypothesis in both cases: the means of the coverages are equal).
Results
The results of the nineteen variables assessed in the five analyzed coverages are presented in Table 3. The variation collected from PC1 and PC2 was 45.1% and 16.4%, respectively (Fig. 2). The grouping of the data in the principal component analysis showed two extreme land covers (SF and DS) that, given their characteristics, presented low standard variances. SF and DS coverages presented lower CL, AR, and NR variance. Bd, Al, and Pr had greater weight in PC1 positive towards DS, unlike pH, OC, N, Ca, In, and Fn, which had greater weight in PC1 negative towards SF. The variables with the greatest weight in PC1 were In, Fn, Ca, Mg, K, OC, N CEC, AL, and pH. In PC2, the variables with the greatest weight were K, texture, and Pr. The variance shows that CL, AR, and NR coverage is intermediate between DS and SF (Fig. 2).
Principal component analysis (PCA). Al: aluminum; Ca: calcium; K: potassium; Mg: magnesium; N: nitrogen; OC: organic carbon; P: phosphorus; pH; Clay; Sand; Silt; Bd: bulk density; Po: porosity; CEC: cation exchange capacity; Pr: penetration resistance; I: infiltration; Pe: percolation; Fn: families number; In: individuals number. Land use coverages were defined as cropland (CL), natural regeneration (NR), degraded soil (DS), active restoration (AR), and secondary forest (SF)
In the matrix of Fig. 3, it is possible to distinguish a trend of negative correlation in red tones between texture, Bd, Al, and Pr, especially with Ca, Mg, K, and pH. A positive correlation was evidenced between N and OC with In and Fn.
Correlation matrix. Correlation between physical, chemical, and biological soil variables. The color bar indicates the direction and intensity of the correlation. Colors in the blue band signify a positive correlation, and those in the red band indicate negative correlations. Al: aluminum; Ca: calcium; K: potassium; Mg: magnesium; N: nitrogen; OC: organic carbon; P: phosphorus; pH; Clay; Sand; Silt; Bd: bulk density; Po: porosity; CEC: cation exchange capacity; Pr: penetration resistance; I: infiltration; Pe: percolation; Fn: families number; In: individuals number
Analysis of variance tools and Dunnett's test
Dunnett's test showed no significant differences between active restoration (AR) and natural regeneration (NR) cover and secondary forest (SF). These treatments showed better soil conditions through the restoration processes. For AR, NR, and SF, Fn Bd and Pr had similar behavior. Active restoration (AR) did not differ significantly from the secondary forest (SF), while natural regeneration (NR) differed in pH, Al, and K concentration. AR showed better behavior than NR in these parameters. Crops (CL) did not show significant differences in pH, Al, or K concerning secondary forest (SF) associated with mechanical work or tillage on CL.
The pH (Fig. 4A) showed significant differences between SF, DS, and RN. Aluminum (Al) content (Fig. 4B) showed significant differences between SF, DS, and NR, and neither between AR and CL. An inverse relationship between pH and Al content was observed; cover crops with acid pH (< 5.0) also showed high Al levels (Fig. 4).
Analysis of variance between coverages, AR: active restoration, CL: cropland, DS: degraded soil, NR: natural regeneration, SF: secondary forest. A pH value (pH), p-value ANOVA: 0.000323 ***; B Aluminum (Al) content, p-value ANOVA: 7.84e-06 ***. The letters a and b in each analysis result from Dunnett's test, where all the land uses were compared with a control (SF). a: no significant difference; b: significant difference
Regarding organic carbon (OC) and nitrogen (N) content (Fig. 5A and B), SF showed significant differences concerning NR, DS, CL, and AR. Regarding potassium (K) content (Fig. 5C), SF did not show significant differences to CL and AR but did show significant differences with NR and DS.
Analysis of variance between coverages, AR: active restoration, CL: cropland, DS: degraded soil, NR: natural regeneration, SF: secondary forest. A Organic carbon (OC) content, p-value ANOVA: 9.86e-06 ***; B Nitrogen (N) content, p-value ANOVA: 4.19e-05 ***; C Potassium (K) content, p-value ANOVA: 0.000236 ***. Letters a and b in each analysis result from Dunnett’s test, where all covers are compared with a control (SF). a: no significant difference; b: significant difference. The levels of N and K had the same behavior as CICE
Bulk density (Bd) and penetration resistance (Pr) (Fig. 6A and B) showed significant differences between SF and DS and non-significant differences with NR, CL, and AR. Regarding the number of individuals (In) and families (Fn) of soil macroinvertebrates (Fig. 6C and D), SF presented significant differences to DS and CL and non-significant differences for NR and AR.
Analysis of variance between coverages, AR: active restoration, CL: cropland, DS: degraded soil, NR: natural regeneration, SF: secondary forest. A bulk density (Bd), p-value ANOVA: 0.00805 **; B penetration resistance (Pr), p-value ANOVA: 7.51e-05 ***; C number of individuals (In), p-value ANOVA: 0.0153 *; D number of families (Fn), p-value ANOVA: 0.011*. Letters a and b in each analysis result from Dunnett's test, where all covers are compared with a control (SF). a: no significant difference; b: significant difference
The increase observed in Bd for all canopies was the response to a decrease in the number of individuals (In) and families (Fn) of soil macroinvertebrates. The highest values of Bd and Pr and the lowest values of individuals (In) and number of families (Fn) of soil macroinvertebrates were found in DS. The SF and NR coverages presented the highest values of In and Fn. The NR coverage approached the SF conditions for all parameters and moved away from DS (Fig. 6).
The infiltration analysis (I) (Fig. 7A) showed significant differences between SF and the other land uses (AR, CL, and DS). Percolation (Pe) (Fig. 7B) in SF showed significant differences with AR, DS, and NR and no significant differences for CL. The SF cover presented the highest infiltration (I) and percolation (Pe) values. The lowest values of infiltration (I) were found in NR and those of percolation (Pe) in AR (Fig. 7).
Analysis of variance between coverages, AR: active restoration, CL: cropland, DS: degraded soil, NR: natural regeneration, SF: secondary forest. A infiltration (I), p-value ANOVA: 0.0937; B percolation (Pe), p-value ANOVA: 0.0515. The letters a and b in each analysis result from Dunnett's test, where all the coverages are compared with a control (SF). a: no significant difference; b: significant difference
Discussion
Bd, Al, and Pr are associated in the direction of SD. The increase in Al is associated with high acidity and limitation in developing vegetation and edaphic fauna (Forero 2011; Rahman and Upadhyaya 2021). In tropical soils, acidification is a natural process closely related to low base contents and soil impoverishment processes that affect soil fertility (Quinto-Mosquera et al. 2022). High Pr and Bd values are related to low soil quality and decreased macroinvertebrate populations (Capowiez et al. 2021; Lin et al. 2022), associated with a high degree of compaction that limits root growth and distribution and vegetation regeneration (Acton et al. 2011); at the same time affecting the porous soil system and air and water exchange. Basamba et al. (2006) stress the importance of biophysical measurements and bulk density and porosity as predictors of system performance and health. The pH, OC, N, Ca, In, and Fn parameters were associated in the direction of SF. In tropical forests, soil nutrient availability, organic matter content, and intense physicochemical changes in short periods condition vegetation development (Martínez et al. 2020; Quinto-Mosquera et al. 2022); at the same time, it has a direct effect on pH nutrient availability, SOM, CEC, and soil compaction (Betancur-Corredor et al. 2023). The variance in this analysis showed that the AR and NR covers are intermediate between DS and SF, suggesting the effectiveness of the restoration processes. The ecosystems were resilient and maintained fertility due to improved soil conditions (Chauveau 2015; Chapin et al. 2012).
The AR and CL covers did not show significant differences with SF in pH, K, and Al contents, contrary to the NR and DS covers, which showed significant differences. These relations suggest an improvement of Bd under AR and NR. This response may be associated with higher root activity, organic matter dynamics, and higher pedogenetic activity (Basamba et al. 2006; Betancur-Corredor et al. 2023) associated with native forets. According to Quinto-Mosquera et al. (2022), nutrient balances in tropical forests are not always conditioned by the limiting factor but by elements in optimal amounts. Tejnecký et al. (2020) suggest that forest composition and management practices influence element dynamics, rhizosphere content, and biogeochemical behavior of ecosystems.
CL did not present significant differences in pH with SF due to manual liming in cultivation, which can cause alkalinity while soil pH increases (Fageria and Baligar 2008). However, due to leaching in tropical conditions, Ca, Mg, and other nutrients, added via amendments, quickly leave the system. For Schlesinger (1997), leaching dynamics are more active than nutrient release from parent materials under high rainfall conditions. In and Fn of AR and NR did not show significant differences with SF, in contrast to CL and DS, probably due to changes in pH. Such changes can influence soil communities (Griffiths et al. 2011), whose macroinvertebrates can respond rapidly in the recovering ecosystem (Schloter et al. 2017). They were, therefore, a good indicator of SQ because changes in their population dynamics were associated with physicochemical in the soil and use dynamics (Velásquez and Lavelle 2019). pH and available nutrients may be important for detecting initial changes in the ecosystem in the short term (Tibbett et al. 2019).
Bd and Pr in NR, AR, and CL were not significantly different from SF; however, DS did show significant differences from SF. The results for AR and NR suggest a reduction of Bd in restoration, coinciding with Jiao et al. (2011) and Neris et al. (2012). Decreased Bd indicates improvements in structure and porosity (Li and Shao 2006) and soil quality at different successional stages (Martinez and Zinck 2004). The Bd values in CL were due to the chemical and manual treatment performed on the cover. Accumulation of leaf litter in coffee plantations increases physical quality, and vegetative cover improves soil conditions (Da Rocha Junior et al. 2020). Bd values in SD were associated with degradation, structure and porosity losses, water behavior, and soil aeration (Acevedo and Silva 2003; León et al. 2013). A direct effect was seen in the availability and efficiency of nutrients in the cycle with plants, animals, and the atmosphere, which made them more susceptible to erosion (Magdoff and Van Es 2021).
SF presented high values of N and OC, resulting in significant differences compared to NR, AR, CL, and DS, results that were due to the slow accumulation of N. Even in developed forests, the N cycle, characteristic of a mature tropical forest, may not be reestablished (Amazonas et al. 2011; Davidson et al. 2007; Vitousek et al. 1989). The relationship between OC and forest age is not always direct and may only present changes in 20 years of development (Marin-Spiotta et al. 2009; Neumann-Cosel et al. 2011). The results of León et al. (2013), Martínez et al. (2020), and Betancur-Corredor et al. (2023) for neotropical conditions in Colombia showed reactivation of soil nutrients and biogeochemical cycles associated with litterfall and high nutrient returns of N.
Infiltration (I) in AR, CL, and DS did not show significant differences to SF, while NR presented significant differences in I. This response is associated with the parent materials; the different coverages studied come from sandstone rocks (IGAC 2011). Heilweil et al. (2007) conducted hydrological studies in soils developed from sandstone, which showed a high net infiltration rate, mineralogy, and texture, influencing the soil infiltration rate (Wakindiki et al. 2002). In the study, site infiltration had high variability; which will be affected by soil heterogeneity and management, organic matter content, and soil porosity (Helalia 1994; Connolly et al. 1997; Heilweil et al. 2007; Leite et al. 2018). The importance is related to the direct relationship between parent material, texture, and porosity behavior (Gray et al. 2016; Araujo et al. 2017; Bonfatti et al. 2020) and responds to previous land use, soil type, and vegetation (Helalia 1994; Zimmermann et al. 2006). Soil infiltration (I) in SF was associated with an improvement in soil physical structure related to a higher organic matter content (Franzluebbers 2002; Teutscherová et al. 2021), a more developed root system (Basamba et al. 2006; Pohl et al. 2009) and higher macroinvertebrate activity (high pedoturbation) in the soil (Colloff et al. 2010). AR, DS, and NR presented significant differences for Pe in SF and did not present significant differences with CL. Plowing processes and improved mechanization in CL may explain the Pe conditions (Acevedo and Silva 2003; Basamba et al. 2006).
Conclusions
Significant differences in soil quality (SQ) were related to land uses for the study site. The soil's physical, chemical, and biological properties varied among the five evaluated land uses, indicating that the use significantly affects the properties and behavior of soils. Active restoration (AR) and natural regeneration (NR) effectively recover soil quality for Bd, Pr, In, and Fn. AR is more effective than NR in reducing acidity and aluminum concentration and increasing K in the soil. The number of individuals and families of macroinvertebrates showed significant differences according to land use in short periods, which suggests that these parameters will be a good indicator of soil quality. Infiltration was variable at the surface and showed significant differences at depth, influenced by parent material and affected by soil characteristics and land use. Behavior did not change significantly for AR, NR, OC, N, Ca, Mg, and CEC. It is important to highlight the importance of the species adaptability to the environment and not to confuse soil fertility with exuberance; under tropical conditions, plant species have a close relationship with SQ. For future studies, it is recommended to deepen the evaluation of soil biological diversity and its role in the behavior of SQ indicators.
References
Acevedo E, Silva P (2003). Sistema de Labranza y Productividad de los Suelos. In: Agronomía de la cero labranza. Universidad de Chile. Serie Ciencias Agronómicas. 10, 21–35
Acton PM, Fox JF, Campbell JE, Jones AL, Rowe H, Martin D, Bryson S (2011) Role of soil health in maintaining environmental sustainability of surface coal mining. Environ Sci Technol 45(23):10265–10272. https://doi.org/10.1021/ES202764Q/SUPPL_FILE/ES202764Q_SI_001.PDF
Amazonas NT, Martinelli LA, de Piccolo MC, Rodríguez RR (2011) Nitrogen dynamics during ecosystem development in tropical forest restoration. Forest Ecol Manag 262(8):1551–1557. https://doi.org/10.1016/J.FORECO.2011.07.003
Amelung W, Bossio D, de Vries W, Kögel-Knabner I, Lehmann J et al (2020) Towards a global-scale soil climate mitigation strategy. Nat Commun. https://doi.org/10.1038/s41467-020-18887-7
Araujo MA, Zinn YL, Lal R (2017) Soil parent material, texture and oxide contents have little effect on soil organic carbon retention in tropical highlands. Geoderma 300:1–10. https://doi.org/10.1016/J.GEODERMA.2017.04.006
Arshad MA, Martin S (2002) Identifying critical limits for soil quality indicators in agro-ecosystems. Agr Ecosyst Environ 88:153–160. https://doi.org/10.1016/S0167-8809(01)00252-3
Banin LF et al (2022) The road to recovery: a synthesis of outcomes from ecosystem restoration in tropical and sub-tropical Asian forests. Phil Trans R Soc B378:20210090. https://doi.org/10.1098/rstb.2021.0090
Barrios E, Delve RJ, Bekunda M, Mowo J, Agunda J, Ramisch J, Trejo MT, Thomas RJ (2006) Indicators of soil quality: a South-South development of a methodological guide for linking local and technical knowledge. Geoderma 135:248–259. https://doi.org/10.1016/j.geoderma.2005.12.007
Bartelheimer M, Poschlod P (2016) Functional characterizations of Ellenberg indicator values—a review on ecophysiological determinants. Funct Ecol 30(4):506–516. https://doi.org/10.1111/1365-2435.12531
Basamba TA, Amézquita E, Singh BR, Rao IM (2006) Effects of tillage systems on soil physical properties, root distribution and maize yield on a Colombian acid-savanna Oxisol. Acta Agriculturae Scandinavica B Soil Plant Sci 56(4):255–262. https://doi.org/10.1080/09064710500297690
Betancur-Corredor B, Kamau JW, Perez-Jimenez JS, Loaiza-Usuga JC, Cardona-Gallo SA, Denich M, Borgemeister C (2023) Small-scale spatial variability of Technosol properties in a chronosequence of reclamation of dredged sediment landfills. Land Degradation & Development. https://doi.org/10.22541/au.164733180.09654559/v1
Bindraban PS, van der Velde M, Ye L, van den Berg M, Materechera S, Kiba DI, Tamene L, Ragnarsdóttir KV, Jongschaap R, Hoogmoed M, Hoogmoed W, van Beek C, van Lynden G (2012) Assessing the impact of soil degradation on food production. Current Opin Environ Sustain 4(5):478–488. https://doi.org/10.1016/J.COSUST.2012.09.015
Bonfatti BR, Demattê JAM, Marques KPP, Poppiel RR, Rizzo R, de Mendes WS, Silvero NEQ, Safanelli JL (2020) Digital mapping of soil parent material in a heterogeneous tropical area. Geomorphology 367:107305. https://doi.org/10.1016/J.GEOMORPH.2020.107305
Bonner MTL, Allen DE, Brackin R, Smith TE, Lewis T, Shoo LP, Schmidt S (2020) Tropical rainforest restoration plantations are slow to restore the soil biological and organic carbon characteristics of old growth rainforest. Microb Ecol 79:432–442. https://doi.org/10.1007/s00248-019-01414-7
Capowiez Y, Sammartino S, Keller T, Bottinelli N (2021) Decreased burrowing activity of endogeic earthworms and effects on water infiltration in response to an increase in soil bulk density. Pedobiologia 85–86:150728. https://doi.org/10.1016/J.PEDOBI.2021.150728
Certini G, Scalenghe R (2023) The crucial interactions between climate and soil. Sci Total Environ 856(2):159–169. https://doi.org/10.1016/j.scitotenv.2022.159169
Chapin FS, Matson PA, Mooney HA, Vitousek PM (2012) Principles of terrestrial ecosystem ecology. 2nd edition. 1–529. https://doi.org/10.1007/978-1-4419-9504-9/COVER
Chauveau M (2015) Efecto del cambio de manejo de una pradera degradada sobre la resiliencia física de un suelo en la precordillera andina de la Araucanía Effect of management change of a degraded pasture on the soil physical resilience in the Andean mountains of the Araucanía region. Agro Sur 43(2):19–28. https://doi.org/10.4206/agrosur.2015.v43n2-04
Colloff MJ, Pullen KR, Cunningham SA (2010) Restoration of an ecosystem function to revegetation communities: the role of invertebrate macropores in enhancing soil water infiltration. Restor Ecol 18:65–72. https://doi.org/10.1111/J.1526-100X.2010.00667.X
Connolly RD, Freebairn DM, Bridge BJ (1997) Change in infiltration characteristics associated with cultivation history of soils in south-eastern Queensland. Aust J Soil Res 35(6):1341–1358. https://doi.org/10.1071/S97032
Cooke R, Mancini F, Boyd RJ, Evans KL, Shaw A, Webb TJ, Isaac NJB (2023) Protected areas support more species than unprotected areas in Great Britain, but lose them equally rapidly. Biol Cons 278:109884. https://doi.org/10.1016/j.biocon.2022.109884
Da Rocha Junior PR, Sturião WP, Nogueira NO, Passos RR, Donagemma GK, Passos Rangel OJ, Bhattarai R (2020) Soil quality indicators to evaluate environmental services at different landscape positions and land uses in the Atlantic Forest biome. Environ Sustain Indicat. https://doi.org/10.1016/J.INDIC.2020.100047
Davidson EA, de Carvalho CJR, Figueira AM, Ishida FY, Ometto JPHB, Nardoto GB, Sabá RT, Hayashi SN, Leal EC, Vieira ICG, Martinelli LA (2007) Recuperation of nitrogen cycling in Amazonian forests following agricultural abandonment. Nature 447(7147):995–998. https://doi.org/10.1038/nature05900
Delgado A, Gómez JA (2016) The soil. Physical, chemical and biological properties. In: Principles of agronomy for sustainable agriculture (pp 15–26). Springer, Cham. https://doi.org/10.1007/978-3-319-46116-8_2
Delgado A, Perez JC, Cardona Gallo S, Loaiza-Usuga JC (2019) Bioremediation (biostimulation and bioaugmentation) of oil spills contaminated soils under tropical humid forest. Carpathian J Earth Environ Sci. 14 (1), 165–170. https://doi.org/10.26471/cjees/2019/014/068
de Deyn GB, Kooistra L (2021) The role of soils in habitat creation, maintenance and restoration. https://doi.org/10.1098/rstb.2020.0170
Diti I, Legler SE, Caffi T, Rossi V, Canali G, Bosso A, Cancila E, Anelli S, Trioli G, Kleshcheva E, Gatti M, Poni S (2020) A new integrated approach for management of soil threats in the vineyard ecosystem. CATENA 195:104788. https://doi.org/10.1016/j.catena.2020.104788
Drobnik T, Greiner L, Keller A, Grêt-Regamey A (2018) Soil quality indicators—from soil functions to ecosystem services. Ecol Ind 94:151–169. https://doi.org/10.1016/J.ECOLIND.2018.06.052
Espinal TLS, Montenegro ME (1977) Zonas de vida o formaciones vegetales de Colombia. Memoria explicativa sobre el mapa ecológico de Colombia. Instituto Geográfico Agustín Codazzi, IGAC. Departamento Agrológico, Bogotá. 238p
Fageria NK, Baligar VC (2008) Chapter 7 Ameliorating soil acidity of tropical oxisols by liming for sustainable crop production. In: Advances in Agronomy (Vol. 99, pp. 345–399). Academic Press. https://doi.org/10.1016/S0065-2113(08)00407-0
Forero FE, Serrano-Cely PA, Balaguera-López WA (2011) El aluminio en el suelo y algunas estrategias de manejo. Ciencia Y Agricultura 7(1):19–28
Franzluebbers AJ (2002) Water infiltration and soil structure related to organic matter and its stratification with depth. Soil Tillage Res 66(2):197–205. https://doi.org/10.1016/S0167-1987(02)00027-2
Ghorai PS, Biswas S, Purakayastha TJ, Ahmed N, Das TK, Prasanna R, Gawade BH, Bhattacharyya K, Sinha K, Singh P, Das S (2023) Indicators of soil quality and crop productivity assessment at a long-term experiment site in the lower Indo-Gangetic plains. Soil Use Manag 39:503–520. https://doi.org/10.1111/sum.12847
Gray JM, Bishop TFA, Wilford JR (2016) Lithology and soil relationships for soil modelling and mapping. CATENA 147:429–440. https://doi.org/10.1016/J.CATENA.2016.07.045
Griffiths RI, Thomson BC, James P, Bell T, Bailey M, Whiteley AS (2011) The bacterial biogeography of British soils. Environ Microbiol 13(6):1642–1654. https://doi.org/10.1111/J.1462-2920.2011.02480.X
Heilweil V, McKinney T, Zhdanov M, Watt D (2007) Controls on the variability of net infiltration to desert sandstone. Water Resour Res 43:W07431. https://doi.org/10.1029/2006WR005113
Helalia AM (1994) The relation between soil infiltration and effective porosity in different soils. Agric Water Manag 24(1):39–47. https://doi.org/10.1016/0378-3774(93)90060-N
IGAC (2006) Métodos analíticos del laboratorio de suelos. In: Zamudio-Sánchez AM, Carrascal-Carrascal ML, Pulido-Roa CE, Gallardo JF, Gómez-Guzmán ID (eds) Instituto Geográfico Agustín Codazzi (IGAC). Bogotá, Colombia. 648p
IGAC (2011) Estudio general de suelos y zonificación de tierras de norte de Santander. Instituto Geográfico Agustín Codazzi (IGAC). Bogotá, Colombia
Jiao F, Wen ZM, An SS (2011) Changes in soil properties across a chronosequence of vegetation restoration on the Loess Plateau of China. CATENA 86(2):110–116. https://doi.org/10.1016/J.CATENA.2011.03.001
Kisaka O, Shisanya C, Cournac L, Manlay JR, Gitari H, Muriuki J (2023) Integrating no-tillage with agroforestry augments soil quality indicators in Kenya’s dry-land agroecosystems. Soil Tillage Res 227:105586. https://doi.org/10.1016/j.still.2022.105586
Krell F-T, Chung AYC, Deboise E, Eggleton P, Giusti A, Inward K, Krell-Westerwalbesloh S (2005) Quantitative extraction of macro-invertebrates from temperate and tropical leaf litter and soil: efficiency and time-dependent taxonomic biases of the Winkler extraction. Pedobiologia 49:175–186. https://doi.org/10.1016/j.pedobi.2004.10.005
Leite PAM, de Souza ES, dos Santos ES, Gomes RJ, Cantalice JR, Wilcox BP (2018) The influence of forest regrowth on soil hydraulic properties and erosion in a semiarid region of Brazil. Ecohydrology 11(3):e1910. https://doi.org/10.1002/ECO.1910
León JD, Casamitjana M, Osorio NW, Loaiza-Usuga JC (2013) Alluvial-gold-mining degraded soils reclamation using Acacia mangium plantations: an evaluation from biogeochemistry. Chapter 9. In: Plantations: biodiversity, carbon sequestration, and restoration. Nova Science Publishers, Hauppauge, NY. pp 155–176
Lewis K, de Barros FV, Moonlight PW, Hill TC, Oliveira RS, Schmidt IB, Sampaio AB, Pennington RT, Rowland L (2022) Identifying hotspots for ecosystem restoration across heterogeneous tropical savannah-dominated regions. Phil Trans R Soc B 378:20210075. https://doi.org/10.1098/rstb.2021.0075
Li YY, Shao MA (2006) Change of soil physical properties under long-term natural vegetation restoration in the Loess Plateau of China. J Arid Environ 64(1):77–96. https://doi.org/10.1016/J.JARIDENV.2005.04.005
Lin L, de Pue J, Vivanco AKM, van der Bolt F, Cornelis W (2022) Visual assessment of soil structural quality across soil textures and compaction levels—part I: examination of intact soil cores. Geoderma 426:116099. https://doi.org/10.1016/J.GEODERMA.2022.116099
Lozano-Baez SE, Domínguez-Haydar Y, Meli P, van Meerveld I, Vásquez Vásquez K, Castellini M (2021) Key gaps in soil monitoring during forest restoration in Colombia. Restor Ecol 29:e13391. https://doi.org/10.1111/rec.13391
Machado J, Villegas-Palacio C, Loaiza-Usuga JC, Castañeda D (2019) Soil natural capital vulnerability to environmental change. A regional scale approach for tropical soils in the Colombian Andes. Ecol Indicat 96(1):116–126
Magdoff F, Van Es H (2021) Building soils for better crops: ecological management for healthy soils. Sustainable Agriculture Network handbook series 10. Sustainable Agriculture Research & Education (Program). National Institute of Food and Agriculture (U.S.). U.S. Department of Agriculture (USDA). https://lccn.loc.gov/2021018006
Marin-Spiotta E, Silver WL, Swanston CW, Ostertag R (2009) Soil organic matter dynamics during 80 years of reforestation of tropical pastures. Glob Change Biol 15(6):1584–1597. https://doi.org/10.1111/J.1365-2486.2008.01805.X
Martínez LJ, Zinck JA (2004) Temporal variation of soil compaction and deterioration of soil quality in pasture areas of Colombian Amazonia. Soil Tillage Res 75:3–17. https://doi.org/10.1016/j.still.2002.12.001
Martínez JA, Loaiza-Usuga JC, Osorio NW, Correa G, Casamitjana-Causa M (2020) Leaflitter decomposition on diverse silvopastoral systems in a neotropical environment”. J Sustain For 39(7):710–729. https://doi.org/10.1080/10549811.2020.1723112
Neris J, Jiménez C, Fuentes J, Morillas G, Tejedor M (2012) Vegetation and land-use effects on soil properties and water infiltration of Andisols in Tenerife (Canary Islands, Spain). CATENA 98:55–62. https://doi.org/10.1016/J.CATENA.2012.06.006
Neumann-Cosel L, Zimmermann B, Hall JS, van Breugel M, Elsenbeer H (2011) Soil carbon dynamics under young tropical secondary forests on former pastures—a case study from Panama. For Ecol Manage 261(10):1625–1633. https://doi.org/10.1016/J.FORECO.2010.07.023
Parker SS (2010) Buried treasure: soil biodiversity and conservation. Biodivers Conserv 19(13):3743–3756. https://doi.org/10.1007/S10531-010-9924-8/METRICS
Pereira P, Bogunovic I, Muñoz-Rojas M, Brevik EC (2018) Soil ecosystem services, sustainability, valuation and management. Curr Opin Environ Sci Health 5:7–13. https://doi.org/10.1016/J.COESH.2017.12.003
Perez MA (2010) Sistema agroecológico rápido de evaluación de calidad de suelo y salud de cultivos. https://repository.agrosavia.co/handle/20.500.12324/11619
Pohl M, Alig D, Körner C, Rixen C (2009) Higher plant diversity enhances soil stability in disturbed alpine ecosystems. Plant Soil 324(1):91–102. https://doi.org/10.1007/S11104-009-9906-3/METRICS
Pulleman M, Creamer R, Hamer U, Helder J, Pelosi C, Peres G, Rutgers M (2012) Soil biodiversity, biological indicators and soil ecosystem services-an overview of European approaches. Current Opinion in Environmental Sustainability. http://www.fao.org/ag/AGL/agll/soilbiod/
Quinto-Mosquera H, Moreno-Hurtado FH, Quinto-Mosquera H, Moreno-Hurtado FH (2022) Influencia de los nutrientes del suelo sobre el crecimiento arbóreo en bosques del Pacífico colombiano. Colombia For 25(2):30–44. https://doi.org/10.14483/2256201X.18232
Rahman R, Upadhyaya H (2021) Aluminium toxicity and its tolerance in plant: a review. J Plant Biol 64(2):101–121. https://doi.org/10.1007/S12374-020-09280-4/METRICS
Aguilar-Garavito MW, Ramírez (2015) Monitoreo a procesos de restauración ecológica, aplicado a ecosistemas terrestres. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt (IAvH). Bogotá D.C., Colombia. 250 pp
Rumpel C, Amiraslani F, Bossio D et al (2023) Studies from global regions indicate promising avenues for maintaining and increasing soil organic carbon stocks. Reg Environ Change 23:8. https://doi.org/10.1007/s10113-022-02003-0
Schlesinger WH (1997) Biogeochemistry. An analysis of global change. 2nd Edition, Academic Press, San Diego, London, Boston, New York, Sydney, Tokyo, Toronto, 588 p
Schloter M, Nannipieri P, Sørensen SJ, Dirk Van Elsas J (2017) Microbial indicators for soil quality. Biol Fertil Soils. https://doi.org/10.1007/s00374-017-1248-3
SSS (2022a) Keys to soil taxonomy, 13th edition. USDA natural resources conservation service. Soil Survey Staff (SSS)
SSS (2022b) Kellogg Soil Survey Laboratory methods manual. Soil Survey Investigations Report No. 42, Version 6.0. U.S. Department of Agriculture, Natural Resources Conservation Service. Soil Survey Staff (SSS)
Tejnecký V, Křížová P, Penížek V, Maňourová A, Sillam-Dussès D, Šobotník J, Akama PD, Lojka B, Němeček K, Borůvka L, Drábek. (2020) The influence of land-use on tropical soil chemical characteristics with emphasis on aluminium. J Inorg Biochem 204:110962. https://doi.org/10.1016/j.jinorgbio.2019.110962
Teutscherová N, Vázquez E, Sotelo M, Villegas D, Velásquez N, Baquero D, Pulleman M, Arango J (2021) Intensive short-duration rotational grazing is associated with improved soil quality within one year after establishment in Colombia. Appl Soil Ecol 159:103835. https://doi.org/10.1016/j.apsoil.2020.103835
Tibbett M, Gil-Martínez M, Fraser T, Green ID, Duddigan S, de Oliveira VH, Raulund-Rasmussen K, Sizmur T, Diaz A (2019) Long-term acidification of pH neutral grasslands affects soil biodiversity, fertility and function in a heathland restoration. CATENA 180:401–415. https://doi.org/10.1016/J.CATENA.2019.03.013
Tittonell P, Scopel E, Andrieu N, Posthumus H, Mapfumo P, Corbeels M, van Halsema GE, Lahmar R, Lugandu S, Rakotoarisoa J, Mtambanengwe F, Pound B, Chikowo R, Naudin K, Triomphe B, Mkomwa S (2012) ARTICLE IN PRESS G model agroecology-based aggradation-conservation agriculture (ABACO): targeting innovations to combat soil degradation and food insecurity in semi-arid Africa. Field Crops Res. https://doi.org/10.1016/j.fcr.2011.12.011
Vandermeer JH (2011) The ecology of agroecosystems. Jones and Barlett Publishers, Sudbury, p 387
Velasquez E, Lavelle P (2019) Soil macrofauna as an indicator for evaluating soil based ecosystem services in agricultural landscapes. Acta Oecologica 100:103446. https://doi.org/10.1016/J.ACTAO.2019.103446
Velasquez E, Lavelle P, Andrade M (2007) GISQ, a multifunctional indicator of soil quality. Soil Biol Biochem 39(12):3066–3080. https://doi.org/10.1016/j.soilbio.2007.06.013
Vitousek PM, Matson PA, van Cleve K (1989) Nitrogen availability and nitrification during succession: primary, secondary, and old-field seres. Plant Soil 115(2):229–239. https://doi.org/10.1007/BF02202591
Wakindiki IIC, Ben-Hur M (2002) Soil mineralogy and texture effects on crust micromorphology, infiltration, and erosion. Soil Sci Soc Am J 66:897–905. https://doi.org/10.2136/sssaj2002.8970
WRB (2022) World reference base for soil resources. International soil classification system for naming soils and creating legends for soil maps. 4th edition. International Union of Soil Sciences (IUSS), Vienna, Austria.
Zhao WZ, Xiao HL, Liu ZM, Li J (2005) Soil degradation and restoration as affected by land use change in the semiarid Bashang area, northern China. CATENA 59(2):173–186. https://doi.org/10.1016/J.CATENA.2004.06.004
Zimmermann B, Elsenbeer H, de Moraes JM (2006) The influence of land-use changes on soil hydraulic properties: implications for runoff generation. For Ecol Manage 222(1–3):29–38. https://doi.org/10.1016/J.FORECO.2005.10.070
Acknowledgements
We are grateful for the support of the Natural Reserve of the Civil Society “La Montaña Mágica-El Poleo,” the Zapatoca Natural Reserves Network, to Eng. Daniel Mauricio Díaz Rueda, Mr. Reynaldo Díaz Rueda, the Biol. Alexandra González Pereira and colleagues Santiago Caro Cardona, Prof. Dr. Guillermo Antonio Correa Londoño, Dr. Diana Lucía Correa Moreno, and the Research Group on Ecosystem Conservation and Restoration for their unconditional support in this project. Thank you all for making it possible.
Funding
Open Access funding provided by Colombia Consortium.
Author information
Authors and Affiliations
Contributions
All authors of this study made a significant and equivalent contribution to the design, execution, and analysis of the research data presented in this article.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliation or participation in any organization or entity with financial or non-financial interests in the topic or materials discussed in this manuscript. This study was funded by the project “Community Participation in Monitoring Ecological Restoration in Civil Society Nature Reserves at Zapatoca, Santander'', National University of Colombia at Medellín code 51952. The datasets generated and analyzed during this study are not publicly available but can be obtained from the corresponding author upon reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
González, O.F., Baquero-Echeverri, S., Caicedo-García, J.P. et al. Soil quality: an indicator of recovery in a nature reserve in the Colombian Andes. Agroforest Syst 98, 1153–1166 (2024). https://doi.org/10.1007/s10457-023-00951-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-023-00951-y