Abstract
Homegarden (HG) agroforestry combines biological carbon (C) sequestration with biodiversity conservation outcomes. Although C stocks and species richness of HGs vary along elevational gradients and as a function of holding sizes, there is no consensus on the nature and magnitude of such variations. Field studies were conducted in the Western Ghats region of central Kerala, India (180 homesteads in 20 selected panchayats), to evaluate the effects of elevation (near sea level to 1938 m) and garden size (162–10,117 m2) on aboveground C stocks and floristic diversity. The C stocks (per unit area) of HGs (arborescent species) were highly variable (0.63–93.65 Mg ha–1), as garden management was highly individualistic and it exhibited a weak negative relationship with elevation. Likewise, there was a weak negative relationship between C stocks and garden size. Tree stocking levels (stems/garden) and species richness (species/garden) positively impacted total C stocks per garden. Floristic diversity was high in the study area (753 species) and included many rare and endangered species (43 IUCN Red-Listed species) making homegardens circa situm reservoirs of biodiversity. Elevation and holding size exerted a weak negative linear relationship on Simpson’s floristic diversity index, which ranged from 0.26 to 0.93 for the arboreal species. Homegardens, regardless of elevation or size, contribute to C sequestration and agrobiodiversity conservation and help achieve the UN Sustainable Development Goals (SDGs), particularly Climate Action (SDG-13) and conserving agrobiodiversity (SDG-15, Life on Land).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agroforestry systems (AFS) contribute significantly to the global carbon (C) cycle due to their high biomass production and C accumulation potentials (Nair et al. 2021). Homegarden agroforestry (Fig. 1) not only has a large potential for biological C sequestration in soil and biomass (Kumar and Kunhamu 2021) but also is an icon of circa situm biodiversity conservation (preserving planted and/or relic trees and wildings on farmlands where forests or woodlands of similar composition existed once; Dawson et al. 2013; Kumar 2011). Distributed predominantly in the tropical areas of Asia, Africa, Central America, the Caribbean, and the Pacific Islands (Nair et al. 2021), the homegardens (HGs) are rich in plant diversity and have been ranked first among all man-made agroecosystems in terms of species richness (Kumar and Nair 2006; Kabir and Webb 2008a). HGs provide food and nutritional security to the gardeners and represent Nature-based Solutions (NbS) for climate change mitigation and biodiversity conservation. Indeed, they provide an effective framework fundamental to C cycle regulation and agrobiodiversity conservation with enormous potential for achieving the United Nations Sustainable Development Goals (SDGs: Sharma et al. 2022).
Multispecies Kerala homegardens. Top: a large-sized garden on a rolling terrain, Middle: a small garden on the lowland plains and Bottom: a small garden on the edges of paddy fields. The coconut palms (Cocos nucifera) are a prominent feature of the Kerala homegardens and are grown in association with several annual and perennial species of different forms (Photo: BM Kumar)
Multiple factors, however, influence the C sequestration and biodiversity attributes of tropical HGs, making it difficult to discern the effects of a single factor. Elevation, size of the holdings, stand age, and management practices (e.g., the suite of species, silvicultural/agronomic techniques, and harvesting) are important in this respect (Nair et al. 2009, 2010). Increasing elevation, associated with a decline in temperature (Sundqvist et al. 2013), may reduce decomposition rates and enhance soil organic matter (SOC) accumulation. However, such low-temperature regimes are also associated with low radiation regimes and low nitrogen availability, signifying lower productivity levels (Grubb et al. 1963; Moser et al. 2011). The increasing mean annual temperature at lower elevations, conversely, may promote higher biomass productivity, but it is also possible that faster organic matter decomposition leads to lower SOC accumulation.
Experiments in the neotropical mountain forests of Latin America have revealed a negative linear relationship between elevation and standing stocks of aboveground biomass (Raich et al. 2006; Leuschner et al. 2007; Malhi et al. 2017), and a positive trend in respect of SOC (Kitayama and Aiba 2002; Raich et al. 2006; Girardin et al. 2010), with no significant net changes in total C stocks (Girardin et al. 2010; Selmants et al. 2014). Phillips et al. (2019), however, reported a unimodal distribution in the aboveground C stocks of the tropical mountain forests of Colombia with elevation (750–2800 m range), which peaked in the middle of the gradient and then decreased, with a similar trend reported for other tropical mountain forests (e.g., Marshall et al. 2012; Ensslin et al. 2015; Venter et al. 2017).
Although several studies have examined how C stocks of natural forest ecosystems change along an elevational gradient, only a few studies have evaluated the C stocks and species diversity variations of HGs along such a gradient. Birhane et al. (2020) reported that for southern Ethiopian HGs, total biomass and soil C stocks were significantly (p = 0.001) higher in the upper (1877–2000 m) elevation zone than in the mid (1754–1876 m) and lower (1630–1753 m) zones with 26% more C stock in the upper zone. Bargali and Bargali (2020) also showed that soil C stocks increased with increasing altitude (350–2000 m) for small HGs in the Central Himalayas, India; but for medium and large HGs, it increased up to the mid-altitude and decreased thereafter, signifying a complex interrelationship between C stocks, size of gardens, and altitudinal positions.
The association between biological C sequestration and plant diversity has become a subject of huge scientific interest in the discourse on climate change mitigation. Although this hypothesis on species diversity and net primary production (i.e., species-diverse systems are more productive) was tested for natural systems (Tilman et al. 1997), very little research has been conducted to validate these claims in an agroforestry setting. Saha et al. (2009) reported that the soil C stock was directly related to the plant diversity of HGs. Yet, the interrelationships between species richness and aboveground biomass C stocks were seldom evaluated for HGs. It was, therefore, hypothesized that an increase in the number of species may influence the accumulation of biomass C and as a result, HGs that have higher species diversity may have greater C sequestration potential (CSP). However, operational attributes such as the size of HGs and managerial interventions may have a complex effect on species richness and biomass productivity. Therefore, it was further hypothesized that although biomass C stocks of Kerala HGs may co-vary with species richness along an elevational gradient, operational attributes such as the size of the HGs and managerial interventions may have an overriding impact on species diversity and CSP. The specific objectives of the study were (1) to investigate how the aboveground C stocks and species diversity attributes of Kerala HGs varied along an elevational gradient, (2) to examine the differences in aboveground C stocks in HGs of varying sizes, and (3) to assess the interrelationships between the aboveground C stocks and species diversity/richness.
Materials and methods
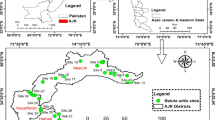
Field surveys were conducted in central Kerala, India, at four locations, namely, Thrissur, Ernakulum, Idukki, and Malappuram districts (Fig. 2). The study area falls within the broad biogeographic region of the Western Ghats, a "global biodiversity hotspot", and experiences a warm humid tropical climate. The principal soil types, number of households, and other details of the study locations are described in KSLUB (1995). HGs constitute the predominant land use activity in the study area (Kumar and Nair 2004; Kumar and Takeuchi 2009; Kumar 2011), which, however, are being cleared for various purposes, but offset by the expansion of younger, less diverse agroforests into paddy wetlands (Fox et al. 2017).
For estimating carbon stocks and species diversity of the HGs along an altitudinal gradient (near sea level to 1938 m elevation), 20 panchayats (the lowest level of local self-government) from the coastal plains to the High Ranges of Kerala were selected considering their elevations and ease of access (i.e., 11% of all panchayats in the designated districts). The survey was conducted during the period from February 2019 to March 2020 (until the onset of the Covid-19 pandemic). The contact details of the farmers and information on the approximate size of holdings and the general land-use pattern were obtained from the Krishibhavans (agricultural development offices) of the respective panchayats. The respondents were contacted and a detailed investigation of their homesteads was conducted, which included gathering details on the nature and kind of species, socioeconomic parameters, and farm management.
Three broad size categories of HGs viz., small (< 0.4 ha holding size), medium (0.4–0.8 ha holding size), and large (> 0.8 ha holding size) were included in the study (at least three gardens each per category and panchayath) and a total of 180 HGs with a total farm area of 190.18 ha was sampled. The extent of HGs within the total farm was ascertained from the gardeners, and it was occasionally cross-checked by field observations. In many small gardens, however, the boundaries of the HGs and the total farms were co-terminus with one another. Geographic coordinates and altitudes of each HG surveyed were noted using a Garmin eTrex 30 × GPS (USA). Mean monthly temperature and precipitation data for the study locations for the 1991–2021 period were downloaded from the Climate-data.org portal (Table 1; https://en.climate-data.org/asia/india/kerala-752/).
All arboreal species (trees and shrubs: ≧ 5 cm girth at breast height; 1.37 m, GBH) present in the HGs, including the border rows, were enumerated by measuring their GBH, bole height, and total height using a measuring tape and Haga Altimeter, respectively. For plantation species such as coconut (Cocos nucifera), rubber (Hevea brasiliensis), areca nut (Areca catechu), and nutmeg (Myristica fragrans), 10 random plants were measured, in addition to recording their total numbers as stated by the landowner. For bamboos, GBH of 10 random culms was measured and the number of culms per clump was counted, besides measuring clump height using a Haga Altimeter. Within each HG, three randomly selected 1 m × 1 m quadrats were also laid out, and the species-wise counts (number of plants within the quadrat) of the herbaceous plants (< 5 cm GBH) were recorded. Sample specimens of plant species that could not be identified in the field were collected for comparison with the herbaria collections maintained at Kerala Agricultural University.
Computations and statistical analysis of the data
The aboveground tree biomass was computed using the following allometric equations for individual trees and summed up to derive the garden-wise biomass estimates. However, species-specific allometric equations have not been developed for several dicot species. Therefore, a generalized allometric equation for the moist tropical forests (FAO 2004) was used in such cases.
-
Ailanthus triphysa: \(\mathrm{ln}B= -2.06622+2.120555\mathrm{ ln}DBH,\) where B = Aboveground biomass, DBH = Diameter at breast height (cm) (Kumar et al. 1998).
-
Artocarpus heterophyllus: \(\mathrm{In}B= -1.71891+2.251298\mathrm{ ln}DBH\), where B = Aboveground biomass, DBH = Diameter at breast height (cm) (Kumar et al. 1998).
-
Artocarpus hirsutus: \(\mathrm{ln}B = -3.06934+2.793477\mathrm{ ln}DBH\), where B = Aboveground biomass, DBH = Diameter at breast height (cm) (Kumar et al. 1998).
-
Emblica officinalis (syn. Phyllanthus emblica):
\(\mathrm{Ln}B=-2.70511+0.969805\mathrm{ ln}({DBH}^{2}H)\), where B = Aboveground biomass, DBH = Diameter at breast height (cm), H = Total height (m) (Kumar et al. 1998).
-
Hevea brasiliensis: \(B=0.002604 {G}^{2.7826}\), where B = Shoot weight in kg and G = girth in cm at 150 cm height (Shorrocks et al. 1965).
-
Tectona grandis: \({\mathrm{Log}}_{10}B = -0.815+2.382 {\mathrm{log}}_{10}DBH\), where B = Aboveground biomass, DBH = Diameter at breast height (cm) (Kraenzel et al. 2003).
-
All other dicot trees: \(Y = \mathrm{exp}\left\{-2.134+2.53 \mathrm{ln} \left(D\right)\right\},\) where Y = biomass per tree in kg, exp = exponential function, D = diameter at breast height in cm. This equation predicts the trunk and canopy biomass of moist (1500–4000 mm rainfall) forest trees (D < 80 cm) with reasonable precision (R2 = 0.97) (FAO 2004).
-
Coconut palms: \(Y= 5.5209x+89.355\), where Y = dry weight (kg); x = palm age (years) (Kumar and Russell, unpublished data, 2006).
-
All other palms (areca palm or Areca catechu L., palmyra palm or Borassus flabellifer L., etc.): \(Y=4.5+7.7 H\), where Y = biomass (kg) and H = stem height (m) (Brown 1997).
-
Bambusa spp. (culms): Log B = \(-1.2471+3.5238 \mathrm{log}D\), where B is the total dry weight (kg) and D is the culm diameter at breast height (cm) (Shanmughavel and Francis 1996).
Carbon sequestration was estimated based on biomass stocks, i.e., 50% of the total aboveground biomass stock was taken as C stock. Total C stock (per garden) was divided with the HG area, which includes the area used for herbaceous crop production in association with trees and the portions used for residential purposes, to derive the C stock per hectare.
Simpson’s diversity index (D) was calculated as \(D=1-\left[\sum\limits_{i=1}^{S}\left(\frac{n_{i}}{N}\right)^2\right]\), where s = total number of species, ni = number of individuals of species ‘i’, N = total number of individuals in the plot, and D is the diversity index (Simpson 1949) and Shannon index (H’) was computed as \(H^{\prime}=3.3219 \left({\rm{log_{10}}} \;N-\left({\frac{1}{N}}\right)\sum\limits_{i=1}^{S} n_{i}{\rm{log_{10}}} n_{i}\right),\) where H’ = Shannon index, s = total number of species, ni = number of individuals of species ‘i’, and N = total number of individuals in the plot (Shannon and Weaver 1949). In addition, Hmax, the maximum dispersion taking into account the number of species present (\(H_{\max } = 3.3219 \log_ {10}S\), where S is the total number of species) and equitability (E), a measure of homogeneity of relative diversity (\(E = H^{\prime } / H_{\max }\)), were computed. Species richness or accumulation (number of species) and stocking levels (number of stems per garden) of arboreal species (≧ 5 cm GBH) were derived from the HG survey data.
Linear regression equations linking aboveground C stocks (per hectare) with elevational positions and size of the holdings, tree stocking levels (stems per garden) and species richness of arboreal species with holding size, total C stocks (per garden) with stocking levels and species richness, and Simpson’s floristic diversity indexes of arboreal species with the size of holdings and elevational positions were fitted, using the "least squares" method and applying the principle of parsimony in MS-Excel. Since the R2 values of the fitted equations were modest, other equations (exponential, power, logarithmic, and polynomial) were also tried, which again did not improve the R2 values much. Analysis of variance was performed to test the levels of variability within the regression model.
Results
Aboveground C stocks
The aboveground C stock ranged from 0.63 to 93.65 Mg ha–1 for the HGs studied (Fig. 3A), implying great variability in the C sequestration potential of HGs. Average standing stocks of C panchayat-wise (for all holding sizes) were in the range of 6–58 Mg ha–1 (Table 1). The moderately high standard error values also highlight the high degree of variability in the C stocks within the panchayats. Elevational gradient generally had a negative linear relationship with C stocks (Fig. 3A). But the R2 value was low, indicating high “noise” levels in the dataset (F = 1.924; p = 0.167). The size of the HGs (Fig. 3B) also did not exert any robust influence on biomass C stocks. Although a higher C stocks in smaller HGs was postulated, the data presented in Fig. 3B, show only a weak negative trend (F = 2.32; p = 0.129). The size of gardens, however, was a major determinant of the number of stems of arboreal species per garden, which increased more or less linearly (F = 18.78; p < 0.00002) and the larger HGs generally had more stems per garden (Fig. 4A). The number of species also increased with the size of the garden (Fig. 4B) in a log-linear manner. The coefficients of determination for the prediction equations were, however, low. The wide variability among the HGs in species richness (3–38 species) and the number of stems per garden (21–1640) presumably may explain this lack of tight relationships among the variables. The number of stems per garden, in turn, determined the total C stocks per garden (Fig. 5A) and the linear function linking the variables explained the trend reasonably well with an R2 value of 47% (F = 154.92; p < 0.0001) signifying that large gardens with a greater number of stems will have higher total C stocks. The total C stock also increased with the number of species (Fig. 5B; R2 = 15%; F = 32.42; p < 0.0001). Aboveground C stocks per hectare, however, did not show a distinctive trend in respect of the number of stems or species per garden (data not presented).
Floristic richness and diversity
A total of 753 plant species (313 herbs, 155 shrubs, and 285 trees; Table 2 and Appendix 1) were encountered in the HGs studied, which involved 43 endangered species (8 Near Threatened, 9 Endangered, 3 Critically Endangered, and 23 Vulnerable species) as per the IUCN Red List (IUCN 2022). Significantly, this included as many as 20 vulnerable trees, which are listed in Table 3 along with the other endangered species.
The number of stems and species per HG showed considerable variability among the studied gardens and their locations (Table 4), implying the interplay of holding size and management factors, besides locational attributes in determining stocking levels (number of stems per garden) and species richness of the HGs. For example, a high-altitude location such as Vattavada (near Munnar), where winter vegetable growing is the predominant occupation of the villagers, had the smallest suite of species and stocking levels (five species and 38 stems per garden respectively). Conversely, Panchayats like Madakkathara, Pallivasal, and Vandiperiyar located in vastly different altitudinal zones (Table 1) showed a greater number of species (24 to 26 per garden) and stems (≧ 5 cm GBH) per garden (308 to 644 stems), signifying the highly location-specific and unique nature of the HG systems.
The Simpson’s diversity index (D) of the sampled HG trees and shrubs (≧ 5 cm GBH; Table 4) ranged from 0.55 (Adimali) to 0.84 (Pariyaram) and the density-based Shannon index (H’) from 1.34 (Adimali) to 2.34 (Alagappanagar), with Hmax values of 1.472 (Vattavada) to 3.24 (Vandiperiyar), signifying moderately high species diversity. The moderate to high equitability or evenness index, 0.504 (Adimali) to 0.855 (Vattavada), indicates that the stems were more or less evenly distributed among the species. Neither holding size (Fig. 6A; F = NS) nor elevation (Fig. 6B; F = NS) revealed any distinct floristic diversity pattern for the arboreal species, but a faint negative correlation was visible for both parameters.
Discussion
Homegardens for climate action
Biological C sequestration, a key strategy for reducing atmospheric concentrations of CO2 and for mitigating global warming, is a central feature of HGs. A comparison of the data in Table 1 indicates that the aboveground biomass C stocks of HGs in central Kerala (arboreal species) ranged between 6 and 58 Mg ha–1. The high variability among the panchayats may be because of the interplay of a complex set of factors such as species composition of the gardens (Table 4), HG sizes which ranged from 162 to 10,117 m2, site characteristics, tree age, tree stocking or density, and tree/land management practices (Nair et al. 2010; Kumar 2011). Many of these parameters differed widely among the gardens and among the physiographic zones of Kerala (midlands, highlands, and coastal plains). Regardless of such variations, the aboveground standing C stocks were generally very high (> 30 Mg ha–1). Kumar (2011) in the previously mentioned study also reported average aboveground standing stocks of C in the range of 16–36 Mg ha−1 for trees in the size class above 20 cm GBH, which is broadly consistent with the present observations. The high biomass C stocks indicate the potential of HGs in climate change mitigation strategies (e.g., SDG-13; Sharma et al. 2022). For instance, in neighboring Sri Lanka, HGs figure prominently in the Intended Nationally Determined Contributions (INDCs) to the United Nations Framework Convention on Climate Change (UNFCCC) and in meeting the climate mitigation goals under UN REDD + Program to cut emissions from deforestation and forest degradation (Mattsson et al. 2018). Nonetheless, locations like Vattavada (Munnar), where winter vegetable growing is the predominant land use activity in the managed ecosystems, had low C stock values such as 6 Mg ha−1, which highlights the crucial role that land use management plays in determining biomass C stocks.
Homegarden C stocks along the elevational gradient
Although elevation is generally considered an important determinant (negative) of biomass C stocks, elevation of HGs showed only a weak negative correlation in the present study (Fig. 3A). For natural ecosystems, many previous studies, however, have reported robust negative relationships between elevation and C stocks (Raich et al. 2006; Leuschner et al. 2007; Malhi et al. 2017). The underlying functional relationship between elevational positions and forest net primary productivity is usually explained by the strong correlation between elevation and mean annual temperature (Girardin et al. 2013) and/or precipitation seasonality (Cruz-Amo et al. 2020). The variations in mean monthly temperature values (range: 17.8–29.0 °C; Table 1) and precipitation seasonality (a general bimodal distribution pattern with the mean annual precipitation ranging from 1548 mm for Chakkupallam to 3410 mm for Vandiperiyar, about 20 km way; Table 1) for the present study locations, however, were not profound, implying that other resource gradients that co-vary with elevation, and more importantly, the management factors play a crucial role in this respect. Consistent with this, Birhane et al. (2020) attributed the greater C stocks in the high elevation zone to the higher frequency of the bigger diameter class individuals, owing to the exploitation of trees in the HGs at the mid and lower elevations. Furthermore, in the tropical mountain forests of Colombia, Phillips et al. (2019) reported a unimodal distribution of aboveground C stocks with elevation peaking in the middle of the gradient, implying a more complex interrelationship between elevation and biomass C stocks.
Carbon stocks as a function of holdings size
As documented by Kumar (2011) for the mid and lower elevation zones of central Kerala, the size of holdings may systematically vary the C stocks of the HG systems. However, the influence of holding size on the biological C sequestration process was modest in the present study (Fig. 3B). Kumar (2011) in the study on the aboveground C stocks of HGs for a subset of the sampled locations reported that C stocks (on a unit area basis) decreased in the order, small > medium > large size classes. Saha et al. (2009) also reported similar results for soil organic C stocks in the HGs of Thrissur, Kerala. The inclusion of a wider elevational belt for sampling in the present study (Table 1) and the disparate array of HG sizes, besides the interplay of many site factors and managerial interventions of variable intensity, may explain this weak relationship (Fig. 3B). The bottom line is that site factors, managerial interventions, and the structural attributes of HGs play a cardinal role in determining HG biomass C stocks than elevation per se or size of the holdings.
Large gardens with a greater number of stems per garden (Fig. 4A) had higher total C stocks (per garden), as expected (Fig. 5A). Species richness also exerted a favorable influence on total C stocks (Fig. 5B). High species assemblages in the HGs may promote higher net primary productivity as they harbor divergent species presumably with contrasting resource-utilization characteristics, in turn, promoting C sequestration, compared to less species-rich systems. Thompson et al. (2012) reported that the C stock in a system depends on both functional diversity and the diversity of woody species. Therefore, a positive relationship between plant species richness and C sequestration (Fig. 5B) is plausible, despite the modest R2 values of the fitted curve. Richards and Mendez (2014), however, argued that since AFS represents human-managed systems with substantial disturbance, greater species diversity may not be associated with increased standing stock of C, implying the crucial role of managerial interventions (species introduction, planting, harvesting, and other agronomic practices) in determining biomass C stocks. A more important driver of C sequestration than species richness, therefore, is the number of stems per garden or stocking levels (Fig. 5A).
Homegardens as a biodiversity conservation tool
With a total of 753 plant species (313 herbs, 155 shrubs, and 285 trees; Table 2), the species richness of the HGs in the study area was profound. Not only that, but the Kerala HGs also harbored many species that figure in the IUCN Red List. For instance, a total of 43 Threatened, Endangered, Critically Endangered, and Vulnerable plants were located in the study area, of which 14 were endemic to the Western Ghats (Table 3). Incidentally, Kerala is home to 5094 taxa of flowering plants (Sasidharan 2012), which includes 1709 taxa that are endemic to peninsular India (Nayar et al. 2008). In addition, the HGs contained several exotic plants as well (data not presented). According to Sasidharan (2012), the flowering plants of Kerala include 858 exotics; of which around 200 species have become naturalized. Furthermore, the farmers may focus on exotic or other fast-growing timber tree species because of economic benefits (Nath et al. 2016) or promotion by certain external agencies (Kabir and Webb 2008b). Consistent with this, Nath et al. (2016), evaluating the tree planting behavior of farmers of Kodagu district, Karnataka state (India), reported that exotic species such as Grevillea robusta are planted 5.4 times more often than native trees.
The Simpson’s and Shannon’s diversity indexes, the two popular diversity indexes used in this study, for the arboreal species generally followed a similar trend (Table 4). The diversity indexes were also moderately high. The natural forest ecosystems, however, had substantially higher diversity indexes (unpublished data). Holding size (Fig. 6A) and elevational gradient (Fig. 6B) exerted only a modest negative influence on Simpson’s floristic diversity attributes. Earlier, Kumar (2011) hypothesized that smaller gardens may have higher species richness (unit area basis) than larger gardens under identical ecological conditions (e.g., mid and lower-elevation HGs). For a wide range of altitudinal positions and holding sizes, however, the variations in site characteristics and managerial interventions may overshadow such effects, and as a result, the generalization that small HGs may have higher species richness than larger gardens is unlikely to hold (Fig. 6A). Likewise, the notion that farmers may grow/plant more species at lower elevations (e.g., Birhane et al. 2020) resulting in greater species diversity at lower elevations was also not true for the HGs of central Kerala (Fig. 6B). Furthermore, the high evenness indexes (Table 4) indicate that individuals are evenly distributed across species, with no discernible influence from location and, inter alia, elevation.
Clearly, HGs provide a potentially valuable conservation tool that can offset the erosion of farmland species diversity. Traditionally, the concept of “Biodiversity hotspots” has been the “mantra” for conservation efforts, which, however, leaves out vast stretches of the globe with considerable biological diversity “in the cold”—the so-called biodiversity “coldspots” (Kareiva and Marvier 2003; Nair et al. 2021). Conservation, in the future, will need to rely increasingly on these smallholder-farm circa situm approaches such as AFS and HGs (Bhagwat et al. 2008), perhaps transitioning to lower and less valuable (from a conservation perspective) farmland tree species diversity over time (Dawson et al. 2013; Nair et al. 2021).
Although harbingers of biodiversity and aligned with SDG-15, HGs have received little or no attention from conservationists in the past. Indeed, there are no policy prescriptions to promote HGs in Kerala and elsewhere. Clearly, there is a need for incentivizing the farmers to conserve these biodiverse land use systems by repaying the cost of maintaining native trees on farms, as exemplified in the Payment for Ecosystem Services (PES) schemes (Ferraro and Kiss 2002; Pascual et al. 2014).
Conclusions and way forward
This study evaluated aboveground C stocks and floristic diversity attributes of HGs of varying ecological characteristics along an elevational gradient from the coastal plains to the High Ranges of Kerala and, inter alia, examined the association between tree stocking levels and species richness with biomass C stocks and garden sizes. Aboveground biomass C stocks of Kerala HGs showed a modest negative correlation with elevation. Although a robust relationship was expected, this was not evident owing to the interplay of a wide array of locational and management factors. Indeed, garden management was highly individualistic in the study area (author’s observations), making any generalization on this vis a vis elevation, difficult. As expected, C stocks (per ha) and holding sizes showed a negative trend, albeit weak, implying greater biomass accumulation on a unit area basis in the smaller gardens, presumably because of more intense land management including higher stand density (stems per ha), than large gardens. But large gardens, by default, had greater stocking levels (stems per garden) and higher species richness and hence higher total C stocks (per garden) than small gardens. High species assemblages presumably have promoted higher net primary productivity as they harbored divergent species with seemingly strong resource-utilization characteristics. Both stocking levels (stems per garden) and species richness (species per garden) showed a positive trend with biomass C stocks, but holding size and elevation exerted a weak negative linear relationship on Simpson’s floristic diversity index.
The generally high floristic diversity levels observed in the study area, which includes many rare and endangered species (43 IUCN Red-Listed species), imply that HGs provide a potentially valuable conservation tool, i.e., a circa situm reservoir of biodiversity that can be employed for offsetting land-use pressure and improving rural livelihoods by providing a range of goods and services. The diverse range of timber and non-timber produce-yielding species in the HGs may also alleviate the resource-use pressure on natural forests. The potential of tree-covered but intensively managed landscapes to support native species suggests that maintaining and creating habitats in human-dominated landscapes can help conserve a large proportion of biodiversity. Furthermore, conservation in the future will need to rely increasingly on these smallholder-farm circa situm approaches, often dubbed as biodiversity “coldspots”. However, HGs in Kerala have received little or no formal attention from the perspectives of biodiversity conservation or climate change mitigation, although the Sri Lankan HGs figure prominently in that country's climate change mitigation programs.
From the multifunctional land-use planning perspective, the conservation feature of HGs should be given proper recognition. However, biodiversity conservation in managed ecosystems such as HGs should be sensitive to the economic needs of the farmers as well. The farmers often must sacrifice part of their incomes for maintaining high species diversity on the farmlands, which should be compensated. Unless this is done, more and more of the traditional land use systems are likely to be transformed into intensive agricultural production systems with a fewer number of species. In fact, many multi-species Kerala HGs have been converted into monospecific production systems of rubber and coconut primarily for greater economic gains in the past. Therefore, incentive schemes should be designed to repay the cost of maintaining these biodiverse agroecosystems involving a diverse range of native trees, i.e., payment for the ecosystem services provided by HGs, besides soft credits for developing ecological farming systems to make conservation appealing to the farmers.
Data availability
Original data are available with the author, which can be shared upon request. Analyzed and summary data are presented in the manuscript along with an Appendix describing the names of 753 plant species.
Code availability
Not applicable.
References
Bargali K, Bargali SS (2020) Effect of size and altitude on soil organic carbon stock in homegarden agroforestry system in Central Himalaya. India Acta Ecologica Sinica 40(6):483–491. https://doi.org/10.1016/j.chnaes.2020.10.002
Bhagwat S, Willis KJ, Birks HJB, Whittaker RJ (2008) Agroforestry: a refuge for tropical biodiversity? Trends Ecol Evol 23:261–267. https://doi.org/10.1016/j.tree.2008.01.005
Birhane E, Ahmed S, Hailemariam M, Negash M, Rannestad MM, Lindsey N (2020) Carbon stock and woody species diversity in homegarden agroforestry along an elevation gradient in southern Ethiopia. Agrofor Syst 94:1099–1110. https://doi.org/10.1007/s10457-019-00475-4
Brown S (1997) Estimating biomass and biomass change of tropical forests: a primer. A Forest Resources Assessment Publication, FAO Forestry Paper 134. Food and Agriculture Organization of the United Nations, Rome
Dawson IK, Guariguata MR, Loo J, Weber JC, Lengkeek A, Bush D, Cornelius J, Guarino L, Kindt R, Orwa C, Russell J, Jamnadass R (2013) What is the relevance of smallholders’ agroforestry systems for conserving tropical tree species and genetic diversity in circa situm, in situ and ex-situ settings? A review. Biodivers Conserv 22:301–324. https://doi.org/10.1007/s10531-012-0429-5
de la Cruz-Amo L, Bañares-de-Dios G, Cala V, Granzow-de la Cerda Í, Espinosa CI, Ledo A, Salinas N, Macía MJ, Cayuela L (2020) Trade-offs among aboveground, belowground, and soil organic carbon stocks along altitudinal gradients in andean tropical montane forests. Front Plant Sci 11:106. https://doi.org/10.3389/fpls.2020.00106
Ensslin A, Rutten G, Pommer U, Zimmermann R, Hemp A, Fischer M (2015) Effects of elevation and land use on the biomass of trees, shrubs and herbs at Mount Kilimanjaro. Ecosphere 6:45. https://doi.org/10.1890/ES14-00492.1
FAO (2004) Assessing carbon stocks and modelling win-win scenarios of carbon sequestration through land-use changes. Food and Agriculture Organization of the United Nations, Rome, p 156
Ferraro PJ, Kiss A (2002) Direct payments to conserve biodiversity. Science 298:1718–1719. https://doi.org/10.1126/science.1078104
Fox TA, Rhemtulla JM, Ramankutty N, Leska C, Coyle T, Kunhamu TK (2017) Agricultural land-use change in Kerala, India: perspectives from above and below the canopy. Agric Ecosyst Environ 245:1–10. https://doi.org/10.1016/j.agee.2017.05.002
Girardin CAJ, Farfan-Rios W, Garcia K, Feeley KJ, Jørgensen PM, Murakami AA, Pérez LC, Seidel R, Paniagua N, Fuentes Claros AF, Maldonado C, Silman M, Salinas N, Reynel C, Neill DA, Serrano M, Caballero CJ, La Torre Cuadros MA, Macía MJ, Killeen TJ, Malhi Y (2013) Spatial patterns of above-ground structure, biomass and composition in a network of six Andean elevation transects. Plant Ecol Divers 7(1–2):161–171. https://doi.org/10.1080/17550874.2013.820806
Girardin CAJ, Malhi Y, Aragao LEOC, Mamani M, Huasco WH, Durand L, Feeley RJ, Rapp J, Silva-Espejo JE, Silman M, Salinas N, Whittaker RJ (2010) Net primary productivity allocation and cycling of carbon along a tropical forest elevational transect in the Peruvian Andes. Glob Change Biol 16:3176–3192. https://doi.org/10.1111/j.1365-2486.2010.02235.x
Grubb PJ, Lloyd JR, Pennington TD, Whitmore TC (1963) A comparison of montane and lowland rain forest in Ecuador I. The forest structure, physiognomy, and floristics. J Ecol 51(3):567–601. https://doi.org/10.2307/2257748
IUCN (2022) The IUCN Red List of Threatened Species. Version 2021-3. https://www.iucnredlist.org
Kabir MDE, Webb EL (2008a) Floristics and structure of southwestern Bangladesh homegardens. Int J Biodivers Sci Ecosyst Serv Manag 3:1–11. https://doi.org/10.1080/17451590809618183
Kabir MDE, Webb EL (2008b) Can homegardens conserve biodiversity in Bangladesh? Biotropica 40(1):95–103. https://doi.org/10.1111/j.1744-7429.2007.00346.x
Kareiva P, Marvier M (2003) Conserving biodiversity coldspots. Am Sci 91:344–351
Kerala State Land Use Board (KSLUB) (1995) Land Resources of Kerala State, Thiruvananthapuram, Kerala, p 209
Kitayama K, Aiba S (2002) Ecosystem structure and productivity of tropical rain forests along altitudinal gradients with contrasting soil phosphorus pools on Mount Kinabalu, Borneo. J Ecol 90:37–51. https://doi.org/10.1046/j.0022-0477.2001.00634.x
Kraenzel M, Castillo A, Moore T, Potvin C (2003) Carbon storage of harvest-age teak (Tectona grandis) plantations, Panama. For Ecol Manag 173:213–225. https://doi.org/10.1016/S0378-1127(02)00002-6
Kumar BM (2011) Species richness and aboveground carbon stocks in the homegardens of central Kerala, India. Agric Ecosyst Environ 140:430–440. https://doi.org/10.1016/j.agee.2011.01.006
Kumar BM, George SJ, Jamaludheen V, Suresh TK (1998) Comparison of biomass production, tree allometry and nutrient use efficiency of multipurpose trees grown in woodlot system and silvopastoral experiments in Kerala. India for Ecol Manag 112(1–2):145–163. https://doi.org/10.1016/S0378-1127(98)00325-9
Kumar BM, Kunhamu TK (2021) Carbon sequestration potential of agroforestry systems in India: a synthesis. In: Udwatta R, Jose S (eds) Agroforestry for ecosystem services. Springer, pp 389–430
Kumar BM, Nair PKR (eds) (2006) Tropical homegardens: a time-tested example of sustainable agroforestry. Volume 3 in the book series “Advances in agroforestry”. Springer Science, The Netherlands, p 377
Kumar BM, Nair PKR (2004) The enigma of tropical homegardens. Agrofor Syst 61(1):135–152. https://doi.org/10.1023/B:AGFO.0000028995.13227.ca
Kumar BM, Takeuchi K (2009) Agroforestry in the Western Ghats of peninsular India and the Satoyama landscapes of Japan: a comparison of two sustainable land use systems. Sustain Sci 4:215–232. https://doi.org/10.1007/s11625-009-0086-0
Leuschner C, Moser G, Bertsch C, Roderstein M, Hertel D (2007) Large altitudinal increase in tree root/shoot ratio in tropical mountain forests of Ecuador. Basic Appl Ecol 8:219–230. https://doi.org/10.1016/j.baae.2006.02.004
Malhi Y, Girardin CAJ, Goldsmith GR, Doughty CE, Salinas N, Metcalfe DB, Huasco WH, Silva-Espejo JE, del Aguilla-Pasquell J, Amézquita FF, Aragão LEOC, Guerrieri R, Ishida FY, Bahar NHA, Farfan-Rios W, Phillips OL, Meir P, Silman M (2017) The variation of productivity and its allocation along a tropical elevation gradient: a whole carbon budget perspective. New Phytol 214:1019–1032. https://doi.org/10.1111/nph.14189
Marshall AR, Willcock S, Platts PJ, Lovett JC, Balmford A, Burgess ND, Latham JE, Munishi PKT, Salter R, Shirima DD, Lewis SL (2012) Measuring and modelling aboveground carbon and tree allometry along a tropical elevation gradient. Biol Conserv 154:20–33. https://doi.org/10.1016/j.biocon.2012.03.017
Mattsson E, Ostwald M, Nissanka SP (2018) What is good about Sri Lankan homegardens with regards to food security? A synthesis of the current scientific knowledge of a multifunctional land-use system. Agroforest Syst 92: 1469–1484. https://doi.org/10.1007/s10457-017-0093-6
Moser G, Leuschner C, Hertel D, Graefe S, Soethe N, Iost S (2011) Elevation effects on the carbon budget of tropical mountain forests (S Ecuador): the role of the belowground compartment. Global Change Biol 17:2211–2226. https://doi.org/10.1111/j.1365-2486.2010.02367.x
Nair PKR, Kumar BM, Nair VD (2009) Agroforestry as a strategy for carbon sequestration. J Plant Nutr Soil Sci 172:10–23. https://doi.org/10.1002/jpln.200800030
Nair PKR, Kumar BM, Nair VD (2021) An introduction to agroforestry: four decades of scientific developments, 2nd edn. Springer Science, The Netherlands
Nair PKR, Nair VD, Kumar BM, Showalter JM (2010) Carbon sequestration in agroforestry systems. Adv Agron 108:237–307. https://doi.org/10.1016/S0065-2113(10)08005-3
Nath CD, Schroth G, Burslem DFRP (2016) Why do farmers plant more exotic than native trees? A case study from the Western Ghats, India. Agric Ecosyst Environ 230:315–328. https://doi.org/10.1016/j.agee.2016.05.013
Nayar TS, Sibi M, Rasiya Beegam A, Mohanan N, Rajkumar G (2008) Flowering plants of Kerala: status and statistics. Rheedea 18:95–106
Pascual U, Phelps J, Garmendia E, Brown K, Corbera E, Martin A, Gomez-Baggethun E, Muradian R (2014) Social equity matters in payments for ecosystem services. Bioscience 64(11):1027–1036. https://doi.org/10.1093/biosci/biu146
Phillips J, Ramirez S, Wayson C, Duque A (2019) Differences in carbon stocks along an elevational gradient in tropical mountain forests of Colombia. Biotropica 51(4):490–499. https://doi.org/10.1111/btp.12675
Raich JW, Russell AE, Kitayama K, Parton WJ, Vitousek PM (2006) Temperature influences carbon accumulation in moist tropical forests. Ecology 87:76–87. https://doi.org/10.1890/05-0023
Richards MB, Mendez VE (2014) Interactions between carbon sequestration and shade tree diversity in a smallholder coffee cooperative in El Salvador. Conserv Biol 28:489–497. https://doi.org/10.1111/cobi.12181
Saha SK, Nair PKR, Nair VD, Kumar BM (2009) Soil carbon stock in relation to plant diversity of homegardens in Kerala, India. Agrofor Syst 76(1):53–65. https://doi.org/10.1007/s10457-009-9228-8
Sasidharan N (2012) Flowering plants of Kerala—version 2.0. DVD No. 14. Kerala Forest Research Institute, Peechi, Kerala, India
Selmants PC, Litton CM, Giardina CP, Asner GP (2014) Ecosystem carbon storage does not vary with mean annual temperature in Hawaiian tropical montane wet forests. Glob Change Biol 20:2927–2937. https://doi.org/10.1111/gcb.12636
Shanmughavel P, Francis K (1996) Aboveground biomass production and nutrient distribution in growing bamboo (Bambusa bambos (L.) Voss). Biomass Bioenergy 10(6):383–391. https://doi.org/10.1016/0961-9534(95)00124-7
Shannon CE, Weaver W (1949) The mathematical theory of communication. University of Illinois Press, Urbana, p 125
Sharma R, Mina U, Kumar BM (2022) Homegardens agroforestry systems in achievement of sustainable development goals: a review. Agron Sustain Dev 42:44. https://doi.org/10.1007/s13593-022-00781-9
Shorrocks VM, Templeton JK, Iyer GC (1965) Mineral nutrition, growth and nutrient cycle of Hevea brasiliensis: 3. Relationship between girth and shoot dry weight. J Rubber Res Inst Malaya 19:5–92
Simpson EH (1949) Measurement of diversity. Nature 163:688–1688. https://doi.org/10.1038/163688a0
Sundqvist MK, Sanders NJ, Wardle DA (2013) Community and ecosystem responses to elevational gradients: processes, mechanisms, and insights for global change. Annu Rev Ecol Evol Syst 44:261–280. https://doi.org/10.1146/annurev-ecolsys-110512-135750
Thompson ID, Ferreira J, Gardner T, Guariguata M, Koh LP, Okab K, Pan Y, Schmitt CB, Tylianakis J, Barlow J, Kapos V, Kurz WA, Parrotta JA, Spalding MD, van Vliet (2012) Forest biodiversity, carbon and other ecosystem services: relationships and impacts of deforestation and forest degradation (Chapter 2). In: Parrotta JA, Wildburger C, Mansourian S (eds) Understanding relationships between biodiversity, carbon, forests and people: the key to achieving REDD+ objectives. A global assessment report prepared by the Global Forest Expert Panel on Biodiversity, Forest Management, and REDD+. IUFRO World Series Volume 31. Vienna, p 161
Tilman D, Lehman CL, Thomson T (1997) Plant diversity and ecosystem productivity: theoretical considerations. Proc Natl Acad Sci USA 94:1857–1861. https://doi.org/10.1073/pnas.94.5.1857
Venter M, Dwyer J, Dieleman W, Ramachandra A, Gillieson D, Laurance S, Cernusak LA, Beehler B, Jensen R, Bird MI (2017) Optimal climate for large trees at high elevations drives patterns of biomass in remote forests of Papua New Guinea. Glob Change Biol 23(11):4873–4883. https://doi.org/10.1111/gcb.13741
Acknowledgements
The study described in this paper was conducted during the tenure of the author as an ICAR Emeritus Scientist at Kerala Agricultural University, Thrissur. Mr. R. Abijith, Mr. P.S. Sreebin, and Mr. Sonik Anto assisted in the fieldwork and Dr. T.K. Kunhamu, Professor and Head, Department of Silviculture and Agroforestry, Kerala Agricultural University, Thrissur, provided the necessary office and laboratory space.
Funding
This research was funded under the Emeritus Scientist Scheme of the Indian Council of Agricultural Research, New Delhi, which was awarded to the author as per Letter F. No. 9 (24)/2018-ES-HRD dated 2 July 2018 of the Indian Council of Agricultural Research, New Delhi. The author also reports a non-financial support relationship with Kerala Agricultural University, Vellanikkara that includes office and administrative support.
Author information
Authors and Affiliations
Contributions
BMK conceived the study, collected and analyzed the data, and prepared the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. The author further declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Consent for publication
Not applicable.
Ethical approval
An ethics approval was not required as per the Kerala Agricultural University guidelines and national regulations. However, the expert committee of the Indian Council of Agricultural Research, New Delhi has reviewed and approved the study as a competitive research grant.
Consent to participate
Verbal informed consent was obtained from all research participants (180 farmer respondents) of the field study according to institutional and national requirements and no minors (non-adults) participated in this research. Verbal approval (prior) from the head of the family also was obtained for conducting the field survey.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix 1
Appendix 1
See Table 5.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, B.M. Do carbon stocks and floristic diversity of tropical homegardens vary along an elevational gradient and based on holding size in central Kerala, India?. Agroforest Syst 97, 751–783 (2023). https://doi.org/10.1007/s10457-023-00821-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-023-00821-7