Abstract
River regulation has altered the seasonal timing of flows in many rivers worldwide, impacting the survival and growth of riparian plants. In south-eastern Australia, demand for irrigation water in summer often results in high river flows during a season that would naturally experience low flows. Although unseasonal high summer flows are thought to significantly impact waterways, their effects on vegetation are poorly quantified. We investigated the responses of five grass species commonly occurring in riparian zones to different durations of submergence in summer. We experimentally tested the response of three exotic and two native grasses to four submergence treatments (4 weeks, 8 weeks, 2-week pulses and no submergence), and two levels of shading (no shading and 80% light reduction), over 8 weeks in summer and early autumn. All submergence treatments, including the 2-week pulse, resulted in the death of all plants of three species (Bromus catharticus, Dactylis glomerata and Rytidosperma caespitosum). Lolium perenne exhibited moderate survival rates in the shorter-duration unshaded submergence treatments, while Poa labillardierei largely survived all treatments. Similar responses across species were observed for plant height and biomass, although height generally increased while biomass growth was reduced by shading. These results show that even 2-week periods of summer submergence can reduce growth and cause the death of some riparian grasses. Although some species may survive longer submergence durations, impacts on other aspects of fitness, and ongoing effects of repeated unseasonal submergence, remain uncertain. Our study highlights that the impacts of unseasonal flows require further investigation and careful management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Natural flow regimes sustain the structure, composition and function of riparian plant communities (Bunn and Arthington 2002). However, river regulation has resulted in the alteration of these natural regimes across many of the world’s rivers, including flow seasonality (Nilsson and Svedmark 2002; Poff and Zimmerman 2010; Grill et al. 2019). For riparian plants, life history stages including growth, flowering, seed dispersal, germination and seedling establishment are often timed with patterns of seasonal flow (Poff et al. 1997; Stromberg et al. 2007; Greet et al. 2011). Consequently, when flow seasonality is altered, declines in populations of riparian plant species and ultimately changes in riparian plant community composition are likely (Greet et al. 2013a, b).
In flow-regulated rivers where the seasonal timing of high flows has been altered by river regulation, riparian vegetation can become partially inundated or even fully submerged at a time of year that would have naturally (i.e. pre-regulation) experienced low flows (Cottingham et al. 2010; Ye et al. 2018a). This occurs in many regulated rivers in the temperate region of south-eastern Australia, where the demand for irrigation water during the driest and warmest time of year has led to an increase of high flows during summer and early autumn (i.e. December to April) when rivers are used to deliver water to irrigators (Maheshwari et al. 1995; Humphries and Lake 1996; Greet et al. 2013a; Fig. 1).
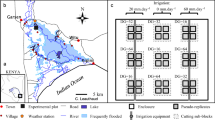
a Conceptual diagram adapted from Greet et al. (2011) illustrating typical changes in flow regimes in many reaches of regulated rivers in south-eastern Australia. Diagram shows pre-regulation peak flows in winter-spring and low flows in summer-autumn (solid line) and post-regulation higher summer-autumn flows due to irrigation water demands and lower winter-spring flows due to capture and storage of water in dams and weirs (dotted line). Also indicated are idealised life history stages of a typical perennial tufted grass, including growth and reproduction, seed release and germination, in relation to river flows. Inset (b–e) represent the four submergence treatments imposed on the study species in our study, including b no submergence; c 2-week pulses interspersed by 2-week dry periods; d 4-week continuous submergence; and e 8-week continuous submergence (see Table 1 for detailed description of treatments)
Submergence causes stress to plants primarily because gas exchange is impeded by the slow diffusion rates of gases in water inhibiting respiration (Jackson and Colmer 2005; Colmer and Voesenek 2009). Plants submerged by unseasonal summer flows are likely to face additional challenges. For example, summer flows may submerge plants during the peak growing season when their metabolic demands are greatest (Crawford 2004; Greet et al. 2011; Fig. 1). Water temperatures during summer and early autumn flows are also likely to be higher than during natural peak flows in late winter and spring. Warmer water temperatures can exacerbate submergence stress for plants, particularly in the growing season, resulting in increased loss of biomass and faster mortality rates compared to submergence in colder water (van Eck et al. 2005, 2006; Ye et al. 2018a). Summer and autumn flows may also be associated with increased water turbidity and reduced light, which can also exacerbate the impact of submergence on plants (Das et al. 2009).
In recent years, the demand for water during summer and autumn has increased across south-eastern Australia, with many of the region’s larger rivers, such as Victoria’s Campaspe and Goulburn Rivers, increasingly used to deliver flows across valleys to more-distant irrigation districts (inter-valley transfers; IVTs) (Cottingham et al. 2010). Given this growing demand for unseasonal flows, it is important to understand their impacts on riparian plants as well as terrestrial plants invading into riparian zones and whether such flows can be delivered in a manner to reduce or manipulate their impacts.
In this study, we investigated the response of five grass species commonly occurring in riparian zones of south-eastern Australia to different durations and frequencies of submergence in summer. Previous experimental studies have shown that some grass species can exhibit a degree of tolerance to submergence durations ranging from several days to more than 2 weeks (Imaz et al. 2012; Striker and Ploschuk 2018; Kitanovic 2019). However, less is known about the response of grasses to different submergence durations during unseasonal times of the year. We tested four combinations of submergence durations and frequencies that represent realistic potential scenarios for delivering summer flows (Fig. 1): single continuous submergence periods of either 4 or 8 weeks; 2-week submergence pulses interspersed with 2-week dry periods; and no submergence (simulating a typical pre-regulation summer low flow). These three submergence treatments reflect both our preconceptions of tolerance thresholds and the approximate previously delivered scenarios in regulated rivers in Victoria. There has been increasing awareness that the delivery of these summer flows as a single stable long-duration event can negatively impact vegetation that becomes submerged, as well as cause notching and erosion of riverbanks, leading to concerted efforts to vary levels by delivering flows as multiple pulsed events (Cottingham et al. 2018; Webb et al. 2019). However, the impact on vegetation of pulsed flows of shorter durations compared to single longer events is not well understood. We aimed to test which of these scenarios of submergence duration would result in the lowest plant mortality and reduction in plant growth. Each submergence scenario was also tested under unshaded and shaded conditions. Shade was included as a factor to investigate the effects of lower light availability that often occurs in turbid waters (e.g. Colmer and Voesenek 2009).
We hypothesised that: (1) plant survival and growth will be greatest in response to no submergence compared to submerged conditions; (2) for submerged plants, their survival and growth will be greater in response to the 2-week submergence pulses compared to continual submergence; and (3) survival and growth of submerged plants will be reduced in shaded compared to unshaded conditions.
Methods
Study species
We experimentally tested our hypotheses on five grass species commonly found in riparian zones of south-eastern Australia, particularly northern Victoria. This region encompasses the southernmost river systems of the Murray-Darling Basin. Major waterways include the Goulburn, Campaspe and Loddon Rivers, which all flow into the Murray River. Rainfall is variable across the region, broadly ranging from 200 to 600 mm per annum (Bureau of Meteorology 2020). Grasses are a dominant structural component of many riparian communities of the waterways in this region, particularly on mid to upper banks (Roberts and Ludwig 2006; Cottingham et al. 2010).
Five grass species were selected for this study: Bromus catharticus (prairie grass), Dactylis glomerata (cocksfoot), Lolium perenne (perennial rye grass), Rytidosperma caespitosum (common wallaby grass) and Poa labillardierei (common tussock grass). These species are frequently recorded in riparian surveys along major rivers in the region, particularly downstream of dams (Greet et al. 2012, 2013a; Jones and Mole 2018). The first three species are undesirable exotic tufted pasture grasses widespread throughout south-eastern Australia, particularly in disturbed areas, while the latter two are desirable native species. Bromus catharticus is an annual or short-lived perennial and the remaining four species are perennial. Although all species occur both within and outside of riparian areas, P. labillardierei is particularly common along streams and alluvial flats, as well as being a dominant species of lowland temperate grasslands and grassy woodlands, and subalpine grasslands (Birch et al. 2015; Clarke 2015; VicFlora 2019).
The five species were chosen as a representative set of common grasses with tufted growth forms that includes both exotic and native species and which are also likely to show a gradation of tolerance to submergence duration. Three of the species, B. catharticus, D. glomerata and R. caespitosum, are considered to be terrestrial dry species, indicating that they occur more widely in the landscape but can invade or persist in riparian zones (Casanova 2011). However, experimental studies have shown that these species can exhibit a degree of tolerance to complete submergence. For example, Striker and Ploschuk (2018) found B. catharticus and D. glomerata survived five days of complete submergence, but growth declined during both the submergence and subsequent recovery periods. Similarly, Kitanovic (2019) found D. glomerata and R. caespitosum survived submergence for 35 days during late winter and early spring with 100% survival, while B. catharticus had a 50% survival rate, although plant growth declined with increasing submergence duration. Lolium perenne has been previously classified as a terrestrial damp species in northern Victorian wetlands (Reid and Quinn 2004) and can grow and survive under waterlogged conditions (McFarlane et al. 2003). In addition, Banach et al. (2009) found it exhibited a 100% survival rate and increased leaf biomass after 3 weeks of submergence, with survival rates dropping by 20–30% after 6 weeks of submergence. Poa labillardierei has been previously classified as a terrestrial damp species (Gehrig and Nicol 2010), indicating it can germinate and establish on saturated or damp ground but cannot tolerate flooding in a vegetative state (Casanova 2011). Recent experiments have shown that it can survive 35 days of submergence in experimental conditions (Kitanovic 2019).
Plant material
Seeds for the experiment were either sourced from a commercial seed supplier (R. caespitosum, L. perenne and D. glomerata) or collected in the field (P. labillardierei and B. catharticus) next to the Campaspe River in northern Victoria, near the township of Rochester (− 36.380974, 144.708964). Soil texture analysis conducted within two separate studies at riparian sites within the southern Murray-Darling Basin showed that soils were dominated by sand and sandy loam to a depth of 0.9 m (Hao et al. 2017; Hu et al. 2017). In April 2018, seeds were germinated in trays containing seed raising mix in a heated glasshouse with a temperature range of 18–25 °C and were kept moist via mist irrigation. After 1 month, 80 seedlings of each species were planted into individual 1.9 L pots containing a 7:1 sand to topsoil mix with a 3–5 cm base layer of pine bark. The pots were placed in an unheated polytunnel for 7 months to allow the plants to establish. Plants were watered for four minutes three times a day and fertilised with 0.5 g/L of fertiliser (N/P/K = 20:8.7:16.6) once a month for the first 3 months and then once every 2 months for the remaining period.
Experimental design
The experiment was conducted in a covered outdoor area in Burnley, Melbourne, Victoria (− 37.828299, 145.020861). The experimental design comprised 16 water tanks measuring 102 cm tall and 97.5 cm diameter. Each tank was randomly allocated to one of eight experimental treatments, resulting in two tanks per treatment. These treatments consisted of four levels of submergence: (1) no submergence (0 weeks), simulating a low summer flow more typical of pre-regulation conditions; (2) 2-week submergence pulses interspersed with 2-week dry periods; (3) 4 weeks of continual submergence; and (4) 8 weeks of continual submergence (Fig. 1). Each treatment also had two levels of shading (unshaded/shaded) to simulate lower light availability that may be experienced in turbid water. Full descriptions of each treatment are provided in Table 1. Tanks in the shaded treatments were covered with a shade cloth that reduced light penetration by 80%.
For each species, 54 plants of a similar size were selected for the experiment, with six randomly chosen for pre-treatment biomass harvesting (see below) and the remaining 48 allocated to one of the eight experimental treatments. Each treatment therefore comprised six replicate plants per species, split across two tanks, with each tank containing fifteen plants (three of each species). The exception was L. perenne, where only 50 plants were available; for this species, four plants (rather than six) were selected for pre-treatment biomass harvesting while two treatments (the unshaded 0-week and shaded 2-week treatments) received one fewer plant each (i.e. five rather than six replicate plants). Plants were placed on a platform inside each tank, which was positioned approximately halfway up the tank.
The experiment commenced on February 15, 2019, when tanks were filled to the appropriate levels with tap water and ran for 8 weeks. Water levels were approximately 45 cm above the level of the substrate within the pots, resulting in plants being submerged by an average of approximately 18 cm, depending on initial height. At approximately 2-week intervals, each plant was measured for height, defined as the longest green section of leaf. Once green leaf material was no longer evident, plants were recorded as dead. At the end of the experiment, plants recorded as dead were placed back in the polytunnel, watered regularly and monitored for recovery (none recovered). The remaining live plants were harvested for biomass assessment. For each plant, leaf and root material was separated, gently washed of soil, dried for at least 72 h at 60 °C and weighed.
The mean daily maximum air temperature during the 8 weeks of the experiment was 24.4 °C (range 15.6–38.1 °C) while the mean daily minimum was 14.6 °C (range 7.4–25.1 °C), measured at the Melbourne (Olympic Park) weather station (ID086338) located 3.5 km to the west (Bureau of Meteorology 2020). Water temperature, pH, electrical conductivity and dissolved oxygen were measured in each tank on two occasions during the fourth and fifth weeks of the experiment. Dissolved oxygen ranged between 2.98 and 8.04 mg/L, electrical conductivity ranged between 0.065 and 0.010 mS/cm and pH ranged between 5.91 and 6.95. Dissolved oxygen was lower in the shaded compared to unshaded 4-week and 8-week submergence treatments. Mean water temperatures across all tanks measured on the two occasions were 17.1 °C and 23.1 °C. As a comparison, in rivers of south-eastern Australia water temperatures generally show a marked difference between winter (generally less than 10 °C) and summer (22—26 °C) (Online Resource 1).
Analyses
Survival rates were assessed as the percentage of plants showing evidence of green leaf material. The log-rank test was used to test the null hypothesis that survival curves differed between treatments. Log-rank tests were constructed using the Kaplan–Meier method in R version 3.6.0 (R Core Team 2019), with the survival package version 2.38 (Therneau 2015). This approach takes into account uncensored data, where an experiment ends prior to the death of all individuals, so as to avoid underestimating the lifespans of these individuals (Pyke and Thompson 1986). However, due to the small number of replicate plants for each species in each treatment combination, comparisons were made separately between levels of shade and levels of submergence.
Growth was assessed using two variables: (1) height, defined as the longest green section of leaf (i.e. maximum green leaf height), measured at approximately 2-week intervals (days 0, 14, 29, 43 and 56 of the experiment), and (2) change in total biomass (roots and leaves combined), assessed at the end of the experiment on living plants. Trends in growth responses were also examined using relative height growth and rates of growth (cm per week) with both metrics showing very similar responses as height. Change in total biomass for each plant was calculated as the final biomass less the species mean biomass of the initially harvested plants (as described above). The height and biomass data were zero-inflated due to the high numbers of plants that died during the experiment, resulting in insufficient numbers of replicates for most species and treatments. As such, the analyses of growth (height and biomass) were performed on living plants only. Firstly, maximum green leaf height was compared, for living plants, between treatments at day 14. Secondly, maximum green leaf height was compared between treatments at day 29 for the three species that survived the first 4 weeks of inundation: L. perenne, R. caespitosum and P. labillardierei. Thirdly, maximum green leaf height and the change in total biomass at 8 weeks was analysed for P. labillardierei only, as most individuals of the other species did not survive in the submergence treatments.
Linear mixed effects models were used to evaluate the relationship of plant height and biomass with treatment. The data were explored visually to check for assumptions of normality with subsequent transformations applied where necessary (outlined below). All model covariates were binary or factor variables and were therefore not scaled prior to analyses. Treatments (shaded and submerged) and their interactions were included as fixed effects. We constructed linear mixed-effects models in R using the lmer function in package lme4 version 1.1–21 (Bates et al. 2015). The plant height models were constructed at different survey periods (2, 4 and 8 weeks) to compare heights at different stages during the experiment for those species with the majority of plants assessed as still alive. For comparisons at two weeks, the submergence treatment was treated as a two-level factor (i.e. 0 weeks vs. 2 weeks of submergence), with the 2-week, 4-week and 8-week treatments pooled because they were identical after 2 weeks. Similarly, for comparisons at 4 weeks, the submergence treatment was treated as a three-level factor (i.e. 0 weeks vs. 2 weeks vs. 4 weeks of submergence), with the 4-week and 8-week treatments pooled because they were identical after 4 weeks. Treatment tank identity was included as a random effect to account for variation between tanks. Fixed effect coefficients for the models indicated effect sizes for each of the treatment levels compared to controls, i.e. 0-week submergence and unshaded. Interactions were included for submergence and shade treatments. For the biomass model, the response variable data were log transformed to allow for model assumptions of normality. For this log-linear model, the exponentiated fixed effect coefficients provided the percentage increase in the response for that treatment compared to the control (percentage calculated as 10coeff − 1*100). Confidence intervals for each model were calculated using the confint function within the stats package. Post hoc contrasts to assess effects and significance between treatment factors were conducted on models using the emmeans function in the emmeans package version 1.4 (Lenth 2019), with significance level of 0.05.
All graphs were produced in R version 3.6.0 (R Core Team 2019), using either the base package or the ggplot2 package version 3.2.1 (Wickham 2016).
Results
Survival
There was no significant difference in the survival of P. labillardierei between shade or submergence levels, as survival rates after 8 weeks were 100% across all treatments, except the shaded 4-week treatment where one plant died (Fig. 2; Online Resource 2). For the remaining four species, survival was significantly different between treatments, with 100% survival rates after 8 weeks in the 0-week treatment (both shaded and unshaded), compared to the survival rates after 8 weeks when submerged, which were either very low (L. perenne) or zero (B. catharticus, D. glomerata and R. caespitosum) irrespective of submergence duration (Fig. 2). Lolium perenne was the only species to significantly differ in survival between levels of shade (Online Resource 2). Survival rates for this species were 67% and 50% after 8 weeks in the unshaded 2-week pulse and unshaded 4-week treatments, respectively, whereas survival was zero in the equivalent shaded treatments. However, in the 8-week submergence treatments, survival rates of L. perenne were zero irrespective of shading (Fig. 2).
Survival rates of plants by species during the experiment. Grey shading in each panel indicates periods of submergence, while white (no shading) indicates periods of emergence. The initial number of plants per species for each treatment combination was six, with the exception of Lolium perenne in the unshaded 0-week and shaded 2-week pulse treatments (five plants)
In the unshaded and shaded 2-week pulse treatments, survival rates for all species were 100% by the end of the first 2 weeks of submergence. However, survival rates declined for all species, except P. labillardierei, during the following 2 weeks of non-submergence/emergence (Fig. 2). By the end of this period, all B. catharticus plants in the 2-week pulse treatment had died, while for D. glomerata 100% of unshaded plants and 66% of shaded plants had died in the 2-week pulse treatment.
Growth
There was an overall negative effect of submergence on maximum green leaf height of B. catharticus, D. glomerata, R. caespitosum and L. perenne by the end of the first 2 weeks (Fig. 3). This was particularly evident for B. catharticus and D. glomerata, while there was a relatively smaller negative effect for R. caespitosum and L. perenne (Fig. 4a). In the 0-week treatment, B. catharticus, D. glomerata and L. perenne increased in height during the first 2 weeks when shaded compared to unshaded (Fig. 3), with a strong positive effect of shading for these three species (Fig. 4a). In contrast, the maximum green leaf height of P. labillardierei remained similar across all treatments within the first 2 weeks (Figs. 3, 4a).
Effect sizes from linear mixed-effects models of treatment covariates (and interactions) on plant height growth after: a 2 weeks of treatment for all species, with submergence treated as a two-level binary factor (i.e. 0 weeks of submergence vs. 2 weeks of submergence with the 2-week, 4-week and 8-week treatments pooled); b 4 weeks of treatment for Rytidosperma caespitosum, Lolium perenne and Poa labillardierei, with submergence included as a three-level factor (i.e. 0 weeks vs. 2 weeks vs. 4 weeks of submergence with the 4-week and 8-week treatment pooled); and (c) 8 weeks of treatment for P. labillardierei with all submergence treatments and shading included. Interactions included are denoted by ‘*’. Levels of treatment on y-axis are in comparison to 0-week (no) submergence and unshaded treatments. Error bars represent 95% CI. The vertical dashed line occurs at effect size of zero (no effect). Sub = submergence treatment
After 4 weeks, the majority of B. catharticus and D. glomerata plants had died. Submergence continued to have a strong negative effect on the maximum green leaf height of R. caespitosum and L. perenne plants recorded as alive (Fig. 3). However, there were no significant differences in the effect sizes between the 2-week pulse and the 4-week submergence treatments (Fig. 4b). Both L. perenne and P. labillardierei increased in height in the 0-week shaded treatment (Figs. 3, 4b).
By the end of the experiment, P. labillardierei was the only species to exhibit a relatively stable maximum green leaf height across all submergence treatments (Fig. 3). While submergence, regardless of duration, was estimated to reduce growth, these differences were uncertain (Fig. 4c). In the 2-week pulse and 4-week treatments, heights were higher when shaded, although these differences were not significant. Poa labillardierei was also taller in the 0-week treatment when shaded compared to unshaded (Fig. 3).
After 8 weeks, there was a decrease in the relative biomass of P. labillardierei when shaded (compared to unshaded) and when submerged (compared to 0-week submergence) (Fig. 5a). There were no significant differences in the decline in biomass between the 2-week, 4-week and 8-week submergence treatments. Interactions between the shading and submergence treatments indicated that the increase in P. labillardierei biomass when unshaded compared to shaded was only apparent in the 0-week treatment (Fig. 5b).
The modelled effect of treatments on percentage difference in total biomass of Poa labillardierei plants after 8 weeks: a excluding interactions and b including interactions. Levels of treatments on y-axis are in comparison to 0-week (no) submergence and unshaded treatments, with interactions included (denoted by *). Error bars indicate 95% CI. The vertical dashed line occurs at difference of zero (no effect). Sub = submergence treatment
Discussion
Our study shows that complete submergence in summer and early autumn under experimental conditions ultimately resulted in the death of almost all of the grass species tested after 8 weeks, irrespective of whether the submergence occurred as 2-week pulses or as longer continuous periods. The exception was P. labillardierei, which largely survived all submergence durations. As such, for four out of the five species studied, these results support our first hypothesis that plant survival and growth would be greatest in response to no submergence compared to submerged conditions. However, there was little support for our second hypothesis, which predicted that for plants exposed to submergence, survival and growth would be greater in response to the 2-week submergence pulses compared to continual submergence. Instead, plant survival and height was similarly low for four of the five species across all submergence treatments. We found only minimal support for our third hypothesis that survival and growth of submerged plants would be reduced in shaded compared to unshaded conditions, with L. perenne surviving better in the 2-week pulse and 4-week continual submergence treatments when unshaded compared to shaded.
Four of the five study species had a high overall mortality rate, including in the 2-week pulse treatment. These four species are not considered riparian specialists; rather, they are terrestrial species that occur widely, but are also often recorded in riparian zones along waterways in south-eastern Australia. As such, their tolerance to submergence is likely to be lower than riparian and wetland specialists, many of which can tolerate submergence durations ranging from weeks to months, including at various times of the year (Mommer et al. 2006; Vivian et al. 2014; Nicol et al. 2018). However, based on the reported tolerances of the study species in other experiments, which range from days to multiple weeks (Banach et al. 2009; Striker and Ploschuk 2018; Kitanovic 2019), we expected a higher degree of survival of the study species in the 2-week pulse treatment. For example, a study investigating the response of riparian grasses to experimental submergence in late winter and early spring, including three of the species investigated here (B. catharticus, P. labillardierei and R. caespitosum) and using the same experimental tank set-up, found that all species had a 100% survival rate under the maximum submergence period of 35 days, with the exception of a low number of B. catharticus plants (Kitanovic 2019). As such, the high mortality rates of four of the five species in this study after 2 weeks of submergence appears to reflect a lower tolerance than reported in other experiments.
Our study differs from many other experimental studies on plant responses to submergence in that plants were submerged during summer. One possible interactive effect on plants with summer submergence is the potential for higher water temperatures. In the current study, mean temperatures measured on two occasions were 17.1 °C and 23.1 °C, in contrast to the lower mean water temperature of 12.9 °C reported by Kitanovic (2019). The primary cause of stress for submerged plants is the low diffusion of oxygen and carbon dioxide in water, which substantially reduces gas exchange to the plant; however, with increasing temperatures, plant survival and growth rates can decline further due to higher respiration rates, accelerated carbohydrate utilisation, and more rapid leaf decay (van Eck et al. 2005; Bailey-Serres and Voesenek 2008; Das et al. 2009; Ye et al. 2018a, b). For example, survival rates of ten grassland species from riparian zones of the River Rhine, including four perennial grasses, were lower in response to simulated summer submergence (water temperatures of 20 °C) compared to winter submergence (maximum water temperature of 6 °C), although the magnitude of the difference varied between species (van Eck et al. 2006). Experiments on submergence tolerance of Oryza sativa (rice) have also shown declines in plant survival with increasing water temperatures (Das et al. 2009). However, differences in survival and growth at different times of the year may also be related to other factors, such as seasonal growth patterns, with plants potentially more sensitive to submergence during the growing season (van Eck et al. 2006). Untangling the relative importance of water temperature on riparian plant survival and growth compared to other potentially influential factors, such as submergence during the growing season, photoperiod, and interactions with water quality and light availability, will assist in understanding and predicting plant responses.
Of the five species, P. labillardierei was the only one to survive submergence while also maintaining relatively constant leaf height and biomass. This suggests an ‘acquiescent’ submergence response strategy, where submergence is tolerated by limiting underwater growth and downregulating processes to conserve energy, in contrast to an ‘escape’ strategy, where species rapidly elongate stems or leaves to the water surface to re-establish contact with the air (Bailey-Serres and Voesenek 2008; Colmer and Voesenek 2009). Species exhibiting the acquiescent strategy tend to have fitness advantages in environments exposed to short duration or deep flooding, i.e. situations where rapid shoot elongation becomes overly costly (Voesenek et al. 2004; Colmer and Voesenek 2009). For P. labillardierei, such a strategy likely reflects its distribution on mid to upper riverbanks, as well as in terrestrial habitats, where any flooding is likely to be of short duration. The maximum duration submergence that P. labillardierei can survive is currently undocumented, including under more typical seasonal flooding. However, given the differential response of other species to submergence during different seasons (e.g. van Eck et al. 2006), it might be expected that its tolerance to winter/spring submergence is even greater, such as in Kitanovic (2019). Although we found survival and biomass maintenance of P. labillardierei was relatively high after 8 weeks of submergence, it is possible that other aspects of its fitness may be affected by prolonged unseasonal submergence. For example, changes to seasonal timing of flows can negatively impact other critical plant life cycle stages, such as flowering, seed production and germination (Poff et al. 1997; Greet et al. 2013b), which in turn could lead to reduced population size or vigour over time. Survival of unseasonal submergence may also depend on plant age (e.g. Denton and Ganf 1994), with the tolerance of smaller or younger plants (e.g. germinants) likely to be lower.
Light is generally limited for submerged plants because light is diffused in water and can be further reduced by turbidity, with studies showing that survival and growth underwater can be improved with increased light intensity (Bailey-Serres and Voesenek 2008; Das et al. 2009; Li et al. 2011). In this study, only L. perenne showed some degree of increasing survival rates under unshaded conditions when submerged, but only in the two shorter submergence periods (2-week pulse and 4-week continuous submergence). However, after 8 weeks, the maximum green leaf height of P. labillardierei was marginally taller in both the shaded 2-week and shaded 4-week treatments compared to the shaded 8-week treatments. This suggests that if submergence of this species occurs under low light conditions, shorter durations may allow more opportunity for leaf elongation, although there was no difference in biomass increase. It is possible that shading—or low light—impacts may be more important for less flood-tolerant species (e.g. B. catharticus, D. glomerata and R. caespitosum) and only during shorter (non-lethal) periods of submergence. Alternatively, reductions in plant survival and growth may be more affected by other characteristics of turbid flows rather than simply light reduction, such as damage from sediment deposition (e.g. Lowe et al. 2010; Catford and Jansson 2014), that we did not examine in this study.
An important finding of our study was that plant mortality in the pulse treatment only became apparent during the 2-week dry period while plants were emergent. Other experimental studies have also found that mortality of plants can become evident during a recovery period following submergence (Denton and Ganf 1994). This highlights the importance of considering the capacity for plants to recover after the removal of stress (van Eck et al. 2004; Striker and Ploschuk 2018) and that some indicators of plant health may not be indicative of longer-term survival. Thus, any investigation of the tolerance of minimum submergence periods should include monitoring of plant survival for a period following plant re-emergence. This is likely to be particularly important for plants recovering from summer submergence, as they may face additional impacts of tissue desiccation from exposure to hot and dry conditions (Jensen et al. 2008; Greet et al. 2013b).
Management implications
Our findings have important implications for the management of unseasonal summer flows. Our results suggest that the delivery of flows in pulses of 2 weeks with a 2-week break to allow plants to recover in between flow events, rather a continuous single 4-week block, will not necessarily reduce plant mortality. We chose a 2-week flow period for our minimum period of submergence, as the reported tolerances of a broad range of riparian and wetland species, and terrestrial grasses found in riparian zones, is generally in the order of weeks to months (e.g. Lowe et al. 2010; Nicol et al. 2018; Kitanovic 2019). However, for four of the five species examined, this duration still resulted in high plant mortality. As such, if a pulse strategy was to be adopted, even shorter durations are likely to be required if the goal is to reduce the mortality of plants with similar tolerances to those in our study. However, the minimum duration of submergence in summer that many plants in riparian zones can tolerate requires further investigation. Additional experimental studies would also be useful for determining the mechanisms by which summer flows can result in rapid mortality, particularly isolating the relative importance of high water temperatures in comparison to other effects of season and interactions with plant life cycles.
A reduction in the discharge of unseasonal summer flows would reduce the risk of complete submergence of vegetation at mid to higher elevations on the bank, such as the group of terrestrial grasses examined in this study. This may allow plants to be partially exposed to the air, likely resulting in significant increases in survival capacity (e.g. Grimoldi et al. 1999; Vivian et al. 2014). However, the tolerance of riparian species typical of lower bank elevations in the region, such as Alternanthera denticulata and Persicaria decipiens, to summer submergence also remains largely unknown and requires further investigation, including any long-term impacts of pulsed submergence. Future experimental studies are also needed to examine the response to unseasonal submergence of species that exhibit an escape response via stem elongation, as submergence at different times of year and/or in different water temperatures may induce a different response in such species. For example, warm (20 °C and 30 °C) water temperatures induced rapid stem elongation in the flood-tolerant species Alternanthera philoxeroides compared to cold (10 °C) temperatures (Ye et al. 2018b).
Three of the grasses included in this study are introduced to south-eastern Australia and are therefore generally undesired components of riparian vegetation communities. Summer flows could potentially be used to control undesired species such as these by exceeding their tolerance to submergence when they are most susceptible. However, there are risks and uncertainties in adopting this strategy. For example, summer flows are likely to also impact non-target (e.g. native) species such as the similarly intolerant native R. caespitosum, which despite being a terrestrial grass can still serve the function of providing cover and habitat during dry and hot months on higher elevation parts of a bank. Thus, using summer flows to control undesirable species could result in an overall decline in vegetation cover and increased bare ground, especially in areas dominated by introduced terrestrial plants and where tolerant species such as P. labillardierei are lacking. Furthermore, while short summer flows may not kill some riparian species (e.g. P. labillardierei), they may still suffer from reduced growth and/or vigour with implications for reproduction (e.g. reduced flowering) and population persistence. The impacts of repeated summer submergence on these other aspects of fitness, particularly over the long term, are uncertain and requires further research. Other risks of such a strategy include the potential for benefiting introduced riparian species commonly found in the region that have been found to be favoured by summer submergence, including the semi-aquatic rhizomatous grass Paspalum distichum and the herb Ludwigia palustris (Greet et al. 2013b). Summer flows may also promote the dispersal of exotic plant species, potentially shifting the composition of riparian plant communities towards increasing dominance by exotics (Greet et al. 2012, 2013b). Thus the use of summer flows for reducing the extent of undesirable plant species in the riparian zone is a risky strategy.
Conclusions
With ever-increasing demands on our river systems, river managers must continually balance the flow regime requirements of riverine environments with society’s water needs and demands. Given that demands for unseasonal flows are also increasing, particularly in south-eastern Australia, it is important to understand how these flows may impact different biota and be best managed. By demonstrating experimentally that even short, 2-week periods of unseasonal summer submergence can cause high mortality of some terrestrial grasses commonly found in riparian habitats, our study suggests that the impacts of unseasonal flows require careful consideration and management.
References
Bailey-Serres J, Voesenek LACJ (2008) Flooding stress: acclimations and genetic diversity. Annu Rev Plant Biol 59:313–339. https://doi.org/10.1146/annurev.arplant.59.032607.092752
Banach K, Banach AM, Lamers LPM et al (2009) Differences in flooding tolerance between species from two wetland habitats with contrasting hydrology: implications for vegetation development in future floodwater retention areas. Ann Bot 103:341–351. https://doi.org/10.1093/aob/mcn183
Bates D, Machler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67. https://doi.org/10.18637/jss.v067.i01
Birch JL, Berwick FB, Walsh N et al (2015) Distribution of morphological diversity within widespread Australian species of Poa (Poaceae, tribe Poeae) and implications for taxonomy of the genus. Aust Syst Bot 27:333–354. https://doi.org/10.1071/SB14028
Bunn SE, Arthington AH (2002) Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ Manag 30:492–507. https://doi.org/10.1007/s00267-002-2737-0
Bureau of Meteorology (2020) Climate data online. https://www.bom.gov.au/climate/data/. Accessed 28 Apr 2020
Casanova MT (2011) Using water plant functional groups to investigate environmental water requirements. Freshw Biol 56:2637–2652. https://doi.org/10.1111/j.1365-2427.2011.02680.x
Catford JA, Jansson R (2014) Drowned, buried and carried away: effects of plant traits on the distribution of native and alien species in riparian ecosystems. New Phytol 204:19–36. https://doi.org/10.1111/nph.12951
Clarke I (2015) Name those grasses. Royal Botanic Gardens Victoria, South Yarra
Colmer TD, Voesenek LACJ (2009) Flooding tolerance: Suites of plant traits in variable environments. Funct Plant Biol 36:665–681. https://doi.org/10.1071/FP09144
Cottingham P, Koster W, Roberts J, Vietz GJ (2018) Assessment of potential inter-valley transfers (IVT) of water from the Goulburn River. Report prepared for the Goulburn-Broken Catchment Management Authority.
Cottingham PD, Stewardson MJ, Roberts J et al (2010) Ecosystem response modelling in the Goulburn River: how much water is too much? In: Saintilan N, Overton IC (eds) Ecosystem response modelling in the Murray-Darling Basin. CSIRO Publishing, Canberra, pp 391–408
Crawford RM (2004) Seasonal differences in plant responses to flooding and anoxia. Can J Bot 81:1224–1246. https://doi.org/10.1139/b03-127
Das KK, Panda D, Sarkar RK et al (2009) Submergence tolerance in relation to variable floodwater conditions in rice. Environ Exp Bot 66:425–434. https://doi.org/10.1016/j.envexpbot.2009.02.015
Denton M, Ganf GG (1994) Response of juvenile Melaleuca halmaturorum to flooding: management implications for a seasonal wetland, Bool Lagoon, South Australia. Aust J Mar Freshw Res 45:1395–1408
Gehrig S, Nicol J (2010) Aquatic and littoral vegetation of the Murray River downstream of Lock 1, the Lower Lakes, Murray Estuary and Coorong. A literature review. South Australian Research and Development Institute (Aquatic Sciences), Adelaide. SARDI Publication Number F2
Greet J, Cousens RD, Webb JA (2013a) More exotic and fewer native plant species: riverine vegetation patterns associated with altered seasonal flow patterns. River Res Appl 29:686–706. https://doi.org/10.1002/rra.2571
Greet J, Cousens RD, Webb JA (2012) Flow regulation affects temporal patterns of riverine plant seed dispersal: potential implications for plant recruitment. Freshw Biol 57:2568–2579. https://doi.org/10.1111/fwb.12028
Greet J, Cousens RD, Webb JA (2013b) Seasonal timing of inundation affects riparian plant growth and flowering: implications for riparian vegetation composition. Plant Ecol 214:87–101. https://doi.org/10.1007/s11258-012-0148-8
Greet J, Webb JA, Cousens RD (2011) The importance of seasonal flow timing for riparian vegetation dynamics: a systematic review using causal criteria analysis. Freshw Biol 56:1231–1247. https://doi.org/10.1111/j.1365-2427.2011.02564.x
Grill G, Lehner B, Thieme M et al (2019) Mapping the world’s free-flowing rivers. Nature 569:215–221. https://doi.org/10.1038/s41586-019-1111-9
Grimoldi AA, Insausti P, Roitman GG, Soriano A (1999) Responses to flooding intensity in Leontodon taraxacoides. New Phytol 141:119–128. https://doi.org/10.1046/j.1469-8137.1999.00325.x
Hao F, Huang D, Lilburne P (2017) A preliminary investigation into the modelled impact of environmental flows on soil moisture content of stream banks in the Wimmera region, Victoria. University of Melbourne, Masters Thesis, Australia
Hu K, Lin A, Liu C (2017) Soil analysis and modelling of soil moisture response to environmental flow in the riparian zone of the Campaspe River. University of Melbourne, Masters Thesis, Australia
Humphries P, Lake PS (1996) Environmental flows in lowland rivers: experimental flow manipulation in the Campaspe River, northern Victoria. In: 23rd Hydrology and Water Resources Symposium, vol 1, pp 197–202
Imaz JA, Giménez DO, Grimoldi AA, Striker GG (2012) The effects of submergence on anatomical, morphological and biomass allocation responses of tropical grasses Chloris gayana and Panicum coloratum at seedling stage. Crop Pasture Sci 63:1145. https://doi.org/10.1071/CP12335
Jackson MB, Colmer TD (2005) Response and adaptation by plants to flooding stress. Ann Bot 96:501–505. https://doi.org/10.1093/aob/mci205
Jensen AE, Walker KF, Paton DC (2008) The role of seedbanks in restoration of floodplain woodlands. River Res Appl 24:632–649. https://doi.org/10.1002/rra.1161
Jones CS, Mole B (2018) Victorian Environmental Flows Monitoring and Assessment Program (VEFMAP) Stage 6: monitoring vegetation response to environmental flow delivery in Victoria 2017/18. Arthur Rylah Institue for Environmental Research, Melbourne
Kitanovic V (2019) Flooding tolerances of six riparian grass species subject to environmental flows. University of Melbourne, Masters Thesis, Australia
Lenth R (2019) emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.4.@@ https://CRAN.R-project.org/package=emmeans
Li F, Li Y, Qin H, Xie Y (2011) Plant distribution can be reflected by the different growth and morphological responses to water level and shade in two emergent macrophyte seedlings in the Sanjiang Plain. Aquat Ecol 45:89–97. https://doi.org/10.1007/s10452-010-9334-8
Lowe BJ, Watts RJ, Roberts J, Robertson A (2010) The effect of experimental inundation and sediment deposition on the survival and growth of two herbaceous riverbank plant species. Plant Ecol 209:57–69. https://doi.org/10.1007/s11258-010-9721-1
Maheshwari BL, Walkers KF, Mcmahon TA (1995) Effects of regulation on the flow regime of the River Murray, Australia. Regul Rivers Res Manag 10:15–38
McFarlane NM, Ciavarella TA, Smith KF (2003) The effects of waterlogging on growth, photosynthesis and biomass allocation in perennial ryegrass (Lolium perenne L.) genotypes with contrasting root development. J Agric Sci 141:241–248. https://doi.org/10.1017/S0021859603003502
Mommer L, Lenssen JPM, Huber H et al (2006) Ecophysiological determinants of plant performance under flooding: a comparative study of seven plant families. J Ecol 94:1117–1129. https://doi.org/10.1111/j.1365-2745.2006.01175.x
Nicol JM, Ganf GG, Walker KF, Gawne B (2018) Response of three arid zone floodplain plant species to inundation. Plant Ecol 219:57–67. https://doi.org/10.1007/s11258-017-0777-z
Nilsson C, Svedmark M (2002) Basic principles and ecological consequences of changing water regimes: riparian plant communities. Environ Manage 30:468–480. https://doi.org/10.1007/s00267-002-2735-2
Poff NL, Allan JD, Bain MB et al (1997) The natural flow regime: a paradigm for river conservation and restoration. Bioscience 47:769–784. https://doi.org/10.2307/1313099
Poff NL, Zimmerman JKH (2010) Ecological responses to altered flow regimes: a literature review to inform the science and management of environmental flows. Freshw Biol 55:194–205. https://doi.org/10.1111/j.1365-2427.2009.02272.x
Pyke DA, Thompson JN (1986) Statistical analysis of survival and removal rate experiments. Ecology 67:240–245
R Core Team (2019) R: a language and environment for statistical computing, version 3.6.1. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org
Reid MA, Quinn GP (2004) Hydrologic regime and macrophyte assemblages in temporary floodplain wetlands: implications for detecting responses to environmental water allocations. Wetlands 24:586–599
Roberts J, Ludwig JA (2006) Riparian vegetation along current-exposure gradients in floodplain wetlands of the River Murray. Aust J Ecol 79:117. https://doi.org/10.2307/2260787
Striker GG, Ploschuk RA (2018) Recovery from short-term complete submergence in temperate pasture grasses. Crop Pasture Sci 69:745. https://doi.org/10.1071/cp18055
Stromberg JC, Lite SJ, Marler R et al (2007) Altered stream-flow regimes and invasive plant species: the Tamarix case. Glob Ecol Biogeogr 16:381–393. https://doi.org/10.1111/j.1466-8238.2007.00297.x
Therneau T (2015) A package for survival analysis in S, version 2.38. https://CRAN.R-project.org/package=survival
van Eck WHJM, Lenssen JPM, Rengelink RHJ et al (2005) Water temperature instead of acclimation stage and oxygen concentration determines responses to winter floods. Aquat Bot 81:253–264. https://doi.org/10.1016/j.aquabot.2004.10.006
van Eck WHJM, Lenssen JPM, Van De Steeg HM et al (2006) Seasonal dependent effects of flooding on plant species survival and zonation: a comparative study of 10 terrestrial grassland species. Hydrobiologia 565:59–69. https://doi.org/10.1007/s10750-005-1905-7
van Eck WHJM, van de Steeg HM, Blom CWPM, de Kroon H (2004) Is tolerance to summer flooding correlated with distribution patterns in river floodplains? A comparative study of 20 terrestrial grassland species. Oikos 107:393–405
VicFlora (2020) Flora of Victoria, Royal Botanic Gardens Victoria. https://vicflora.rbg.vic.gov.au. Accessed 30 Apr 2020
Vivian LM, Marshall DJ, Godfree RC (2014) Response of an invasive native wetland plant to environmental flows: implications for managing regulated floodplain ecosystems. J Environ Manag 132:268–277. https://doi.org/10.1016/j.jenvman.2013.11.015
Voesenek LACJ, Rijnders JHGM, Peeters AJM et al (2004) Plant hormones regulate fast shoot elongation under water: from genes to communities. Ecology 85:16–27. https://doi.org/10.1890/02-740
Webb A, Guo D, King E et al (2019) Commonwealth Environmental Water Office long term intervention monitoring project Goulburn River selected area: summary report 2017–18. Report prepared for the Commonwealth Environmental Water Office
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Ye X, Meng J, Zeng B, Wu M (2018a) Improved flooding tolerance and carbohydrate status of flood-tolerant plant Arundinella anomala at lower water temperature. PLoS ONE 13:1–12. https://doi.org/10.1371/journal.pone.0192608
Ye X, Zeng B, Meng JL et al (2018b) Responses in shoot elongation, carbohydrate utilization and growth recovery of an invasive species to submergence at different water temperatures. Sci Rep 8:306. https://doi.org/10.1038/s41598-017-18735-7
Acknowledgements
We thank Scott McKendrick, Bryan Mole and Marjorie Pereira for assistance with data collection; and the assistance of Burnley nursery staff Nicholas Osborne, Sascha Andrusiak and Rowan Berry. We also thank Vanja Kitanovic for seed collection and plant potting. We gratefully acknowledge Andrew Boulton, Andrew Bennett, Jemma Cripps, Geoff Sutter, Sally Kenny and two anonymous reviewers for feedback on earlier versions of the manuscript, and Ben Fanson for advice on data analyses. This work was funded by the Victorian Government through the Victorian Environmental Flows Monitoring and Assessment Program. The production of this paper was supported by the Applied Aquatic Ecology writing retreat, which in turn was supported by the Capability Fund of the Arthur Rylah Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Télesphore Sime-Ngando.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vivian, L.M., Greet, J. & Jones, C.S. Responses of grasses to experimental submergence in summer: implications for the management of unseasonal flows in regulated rivers. Aquat Ecol 54, 985–999 (2020). https://doi.org/10.1007/s10452-020-09788-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-020-09788-4