Abstract
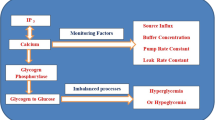
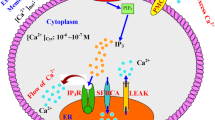
The calcium signals regulate the production and secretion of many signaling molecules like inositol trisphosphate (\(IP_{3}\)) and adenosine triphosphate (ATP) in various cells including pancreatic \(\beta\)-cells. The calcium signaling mechanisms regulating \(IP_{3}\), ATP and insulin responsible for various functions of \(\beta\)-cells are still not well understood. Any disturbance in these mechanisms can alter the functions of \(\beta\)-cells leading to diabetes and metabolic disorders. Therefore, a mathematical model is proposed by incorporating the reaction-diffusion equation for calcium dynamics and a system of first-order differential equations for \(IP_{3}\), ATP-production and insulin secretion with initial and boundary conditions. The model incorporates the temporal dependence of \(IP_{3}\)-production and degradation, ATP production and insulin secretion on calcium dynamics in a \(\beta\)-cell. The piecewise linear finite element method has been used for the spatial dimension and the Crank-Nicolson scheme for the temporal dimension to obtain numerical results. The effect of changes in source influxes and buffers on calcium dynamics and production of \(IP_{3}\), ATP and insulin levels in a \(\beta\)-cell has been analyzed. It is concluded that the dysfunction of source influx and buffers can cause significant variations in calcium levels and dysregulation of \(IP_{3}\), ATP and insulin production, which can lead to various metabolic disorders, diabetes, obesity, etc. The proposed model provides crucial information about the changes in mechanisms of calcium dynamics causing proportionate disturbances in \(IP_3\), ATP and insulin levels in pancreatic cells, which can be helpful for devising protocols for diagnosis and treatment of various metabolic diseases.












Similar content being viewed by others
References
https://www.slideshare.net/NurHanimAbdGhani/mwsgenaaespeppt.measuringerrorsppt
Ainscow EK, Rutter GA (2002) Glucose-stimulated oscillations in free cytosolic ATP concentration imaged in single islet \(\beta\)-cells: evidence for a Ca2+-dependent mechanism. Diabetes 51:S162–S170. https://doi.org/10.2337/diabetes.51.2007.S162
Aronoff SL, Berkowitz K, Shreiner B, Want L (2004) Glucose metabolism and regulation: beyond insulin and glucagon. Diabetes spectrum 17(3):183–190
Bertram R, Pedersen MG, Luciani DS, Sherman A (2006) A simplified model for mitochondrial \(ATP\) production. J Theor Biol 243(4):575–586
Bratanova-Tochkova TK, Cheng H, Daniel S, Gunawardana S, Liu YJ, Mulvaney-Musa J, Sharp GW (2002) Triggering and augmentation mechanisms granule pools and biphasic insulin secretion. Diabetes 51:S83–S90
Braun M, Ramracheya R, Bengtsson M, Zhang Q, Karanauskaite J, Partridge C, Rorsman P (2008) Voltage-gated ion channels in human pancreatic \(\beta\)-cells: electrophysiological characterization and role in insulin secretion. Diabetes 57(6):1618–1628
Chay TR (1986) On the effect of the intracellular calcium-sensitive \(K^{+}\) channel in the bursting pancreatic beta-cell. Biophys J 50(5):765–777. https://doi.org/10.1016/s0006-3495(86)83517-2
Chay TR (1997) Effects of extracellular calcium on electrical bursting and intracellular and luminal calcium oscillations in insulin secreting pancreatic beta-cells. Biophys J 73(3):1673–1688. https://doi.org/10.1016/s0006-3495(97)78199-2
Cotrina ML, Lin JHC, Alves-Rodrigues A, Liu S, Li J, Azmi-Ghadimi H, Nedergaard M (1998) Connexins regulate calcium signaling by controlling ATP release. Proc Natl Acad Sci USA 95(26):15735–15740
Crank J (1979) The mathematics of diffusion. Oxford University Press, Oxford
Dupont G, Falcke M, Kirk V, Sneyd J (2016) Models of calcium signalling, vol 43. Springer, New York
Ehrlich BE (2000) Regulation of the type III InsP3 receptor and its role in \(\beta\)-cell function. Cell Mol Life Sci CMLS 57(13):1938–1949. https://doi.org/10.1007/pl00000674
Escher A, Long A (2013 Type 1 diabetes. BoD-Books on Demand. https://doi.org/10.5772/52153
Felix-Martinez GJ, Godinez-Fernandez JR (2017) Modeling the spatiotemporal distribution of \(Ca^{2+}\) during action potential firing in human pancreatic \(\beta\)-cells. Biomed Phys Eng Express 3(2):025020
Ferrannini E (2010) The stunned \(\beta\)-cell: a brief history. Cell Metab 11(5):349–352. https://doi.org/10.1016/j.cmet.2010.04.009
Fridlyand LE, Tamarina N, Philipson LH (2003) Modeling of \(Ca^{2+}\) flux in pancreatic \(\beta\)-cells: role of the plasma membrane and intracellular stores. Am J Physiol-Endocrinol Metab 285(1):E138–E154. https://doi.org/10.1152/ajpendo.00194.2002
Fridlyand LE, Harbeck MC, Roe MW, Philipson LH (2007) Regulation of \(cAMP\) dynamics by \(Ca^{2+}\) and G protein-coupled receptors in the pancreatic \(\beta\)-cell: a computational approach. Am J Physiol Cell Physiol 293(6):C1924–C1933. https://doi.org/10.1152/ajpcell.00555.2006
Fridlyand LE, Jacobson DA, Kuznetsov A, Philipson LH (2009) A model of action potentials and fast \(Ca^{2+}\) dynamics in pancreatic \(\beta\)-cells. Biophys J 96(8):3126–3139. https://doi.org/10.1016/j.bpj.2009.01.029
Fridlyand LE, Jacobson DA, Philipson LH (2013) Ion channels and regulation of insulin secretion in human \(\beta\)-cells: a computational systems analysis. Islets 5(1):1–15. https://doi.org/10.4161/isl.24166
Gromada J, Frokjar-Jensen J, Dissing S (1996) Glucose stimulates voltage-and calcium-dependent inositol trisphosphate production and intracellular calcium mobilization in insulin-secreting \(\beta\)TC3 cells. Biochem J 314(1):339–345
Hirose K, Kadowaki S, Tanabe M, Takeshima H, Iino M (1999) Spatiotemporal dynamics of inositol 1, 4, 5-trisphosphate that underlies complex \(Ca^{2+}\) mobilization patterns. Science 284(5419):1527–1530. https://doi.org/10.1126/science.284.5419.1527
Idevall-Hagren O, Tengholm A (2020) Metabolic regulation of calcium signaling in beta cells. Semin Cell Dev Biol 103:20–30. https://doi.org/10.1016/j.semcdb.2020.01.008
Jaberi-Douraki M, Schnell S, Pietropaolo M, Khadra A (2015) Unraveling the contribution of pancreatic beta-cell suicide in autoimmune type 1 diabetes. J Theor Biol 375:77–87. https://doi.org/10.1016/j.jtbi.2014.05.003
Jafri MS, Keizer J (1995) On the roles of \(Ca^{2+}\) diffusion, \(Ca^{2+}\) buffers, and the endoplasmic reticulum in \(IP_3\)-induced \(Ca^{2+}\) waves. Biophys J 69(5):2139–2153
Jagtap Y, Adlakha N (2018) Finite volume simulation of two dimensional calcium dynamics in a hepatocyte cell involving buffers and fluxes. Commun Math Biol Neurosci, 2018, Article-ID
Jha BK, Adlakha N, Mehta MN (2012) Finite volume model to study the effect of ER flux on cytosolic calcium distribution in astrocytes. arXiv preprint arXiv:1201.1107
Jha BK, Adlakha N, Mehta MN (2013) Two-dimensional finite element model to study calcium distribution in astrocytes in presence of VGCC and excess buffer. Int J Model Simul Sci Comput 4(02):1250030
Jha BK, Jha A, Adlakha N (2020) Three-dimensional finite element model to study calcium distribution in astrocytes in presence of VGCC and excess buffer. Differ Eq Dynam Syst 28(3):603–616. https://doi.org/10.1007/s12591-019-00502-x
Kaw AK, Kalu EK, Nguyen D (2011) Numerical methods with applications. Textbooks Collection. 11
Keizer J, Magnus G (1989) \(ATP\)-sensitive potassium channel and bursting in the pancreatic \(\beta\)-cell: a theoretical study. Biophys J 56(2):229–242
Kotwani M, Adlakha N, Mehta MN (2012) Numerical model to study calcium diffusion in fibroblasts cell for one dimensional unsteady state case. Appl Math Sci 6(102):5063–5072
Kumar H, Naik PA, Pardasani KR (2018) Finite element model to study calcium distribution in T lymphocyte involving buffers and ryanodine receptors. Proc Natl Acad Sci USA 88:585–590. https://doi.org/10.1007/s40010-017-0380-7
Manhas N, Pardasani KR (2014) Modelling mechanism of calcium oscillations in pancreatic acinar cells. J Bioenerg Biomembr 46(5):403–420. https://doi.org/10.1007/s10863-014-9561-0
Manhas N, Sneyd J, Pardasani KR (2014) Modelling the transition from simple to complex Ca2+ oscillations in pancreatic acinar cells. J Biosci 39(3):463–484. https://doi.org/10.1007/s12038-014-9430-3
Manhas N, Pardasani KR (2014) Mathematical model to study \(IP_3\) dynamics dependent calcium oscillations in pancreatic acinar cells. J Med Imaging Health Inform 4(6):874–880. https://doi.org/10.1166/jmihi.2014.1333
Misler S, Barnett DW, Gillis KD, Pressel DM (1992) Electrophysiology of stimulus-secretion coupling in human \(\beta\)-cells. Diabetes 41(10):1221–1228
Naik P, Pardasani KR (2013) Finite element model to study effect of buffers in presence of voltage gated \(Ca^{2+}\) channels on calcium distribution in oocytes for one dimensional unsteady state case. Int J Mod Biol Med 4(3):190–203
Naik PA, Pardasani KR (2015) One dimensional finite element model to study calcium distribution in oocytes in presence of \(VGCC\), \(RyR\) and buffers. J Med Imaging Health Inform 5(3):471–476. https://doi.org/10.1166/jmihi.2015.1431
Naik PA, Pardasani KR (2017) Three-dimensional finite element model to study calcium distribution in oocytes. Netw Model Anal Health Inform Bioinform 6:1–11. https://doi.org/10.1007/s13721-017-0158-5
Naik PA (2020) Modeling the mechanics of calcium regulation in T lymphocyte: a finite element method approach. Int J Biomath 13(05):2050038
Özis T, Aksan EN, Özdes A (2003) A finite element approach for solution of Burgers’ equation. Appl Math Comput 139(2–3):417–428. https://doi.org/10.1016/S0096-3003(02)00204-7
Pathak KB, Adlakha N (2015) Finite element model to study calcium signalling in cardiac myocytes involving pump, leak and excess buffer. J Med Imaging Health Inform 5(4):683–688. https://doi.org/10.1166/jmihi.2015.1443
Pathak KB, Adlakha N (2015) Finite element model to study one dimensional calcium dyanmics in cardiac myocytes. J Multiscale Modell 6(02):1550003. https://doi.org/10.1142/S1756973715500031
Pedersen MG, Bertram R, Sherman A (2005) Intra-and inter-islet synchronization of metabolically driven insulin secretion. Biophys J 89(1):107–119. https://doi.org/10.1529/biophysj.104.055681
Pedersen MG (2009) Contributions of mathematical modeling of beta cells to the understanding of beta-cell oscillations and insulin secretion. J Diabetes Sci Technol 3(1):12–20. https://doi.org/10.1177/193229680900300103
Politi A, Gaspers LD, Thomas AP, Höfer T (2006) Models of \(IP_3\) and \(Ca^{2+}\) oscillations: frequency encoding and identification of underlying feedbacks. Biophys J 90(9):3120–3133. https://doi.org/10.1529/biophysj.105.072249
Portuesi R, Cherubini C, Gizzi A, Buzzetti R, Pozzilli P, Filippi S (2013) A stochastic mathematical model to study the autoimmune progression towards type 1 diabetes. Diabetes Metab Res Rev 29(3):194–203
Rorsman P (2005) Insulin secretion: function and therapy of pancreatic \(\beta\)-cells in diabetes. Br J Diabetes Vasc Dis 5(4):187–191. https://doi.org/10.1177/14746514050050040201
Rorsman P, Braun M, Zhang Q (2012) Regulation of calcium in pancreatic \(\alpha\)-and \(\beta\)-cells in health and disease. Cell Calcium 51(3–4):300–308. https://doi.org/10.1016/j.ceca.2011.11.006
Sabatini PV, Speckmann T, Lynn FC (2019) Friend and foe: \(\beta\)-cell \(Ca^{2+}\) signaling and the development of diabetes. Mol Metab 21:1–12. https://doi.org/10.1016/j.molmet.2018.12.007
Seino S, Shibasaki T, Minami K (2011) Dynamics of insulin secretion and the clinical implications for obesity and diabetes. J Clin Investig 121(6):2118–2125. https://doi.org/10.1172/JCI45680
Smith GD, Wagner J, Keizer J (1996) Validity of the rapid buffering approximation near a point source of calcium ions. Biophys J 70(6):2527–2539. https://doi.org/10.1016/S0006-3495(96)79824-7
Smith GD (1996) Analytical steady-state solution to the rapid buffering approximation near an open \(Ca^{2+}\) channel. Biophys J 71(6):3064–3072. https://doi.org/10.1016/S0006-3495(96)79500-0
Sprietsma JE, Schuitemaker GE (1994) Diabetes can be prevented by reducing insulin production. Med Hypoth 42(1):15–23. https://doi.org/10.1016/0306-9877(94)90029-9
Stamatakis M, Mantzaris NV (2006) Modeling of \(ATP\)-mediated signal transduction and wave propagation in astrocytic cellular networks. J Theor Biol 241(3):649–668. https://doi.org/10.1016/j.jtbi.2006.01.002
Tamarina NA, Kuznetsov A, Rhodes CJ, Bindokas VP, Philipson LH (2005) Inositol (1, 4, 5)-trisphosphate dynamics and intracellular calcium oscillations in pancreatic \(\beta\)-cells. Diabetes 54(11):3073–3081. https://doi.org/10.2337/diabetes.54.11.3073
Tewari S, Pardasani KR (2009) Finite difference model to study the effects of \(Na^{+}\) influx on cytosolic \(Ca^{2+}\) diffusion. Int J Biol Med Sci 1(2008):4–205
Varadi A, Molnar E, Ashcroft SJ (1995) Characterisation of endoplasmic reticulum and plasma membrane \(Ca^{2+}\)-ATPases in pancreatic \(\beta\)-cells and in islets of Langerhans. Biochim et Biophys Acta Biomembranes 1236(1):119–127. https://doi.org/10.1016/0005-2736(95)00103-A
Wagner J, Keizer J (1994) Effects of rapid buffers on \(Ca^{2+}\) diffusion and \(Ca^{2+}\) oscillations. Biophys J 67(1):447–456. https://doi.org/10.1016/S0006-3495(94)80500-4
Wagner J, Fall CP, Hong F, Sims CE, Allbritton NL, Fontanilla RA, Nuclli R (2004) A wave of \(IP_3\) production accompanies the fertilization \(Ca^{2+}\) wave in the egg of the frog, Xenopus laevis: theoretical and experimental support. Cell Calcium 35(5):433–447. https://doi.org/10.1016/j.ceca.2003.10.009
Wang S, Sherman A (2008) Identifying the targets of the amplifying pathway for insulin secretion in pancreatic \(\beta\)-cells by kinetic modeling of granule exocytosis. Biophys J 95(5):2226–2241. https://doi.org/10.1529/biophysj.107.124990
Wang Z, Haydon PG, Yeung ES (2000) Direct observation of calcium-independent intercellular \(ATP\) signaling in astrocytes. Anal Chem 72(9):2001–2007
Zimny ML, Blackard WG (1975) The surface structure of isolated pancreatic islet cells. Cell Tissue Res 164(4):467–471. https://doi.org/10.1007/BF00219938
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A
Appendix A
The piecewise linear finite element scheme is employed to solve equations (25), (11), (12) and (13). The Euler-Lagrange equation has been used to find out the variational form of equation (25) which is given as follows:
Here, \('w'\) denotes the \([Ca^{2+}]\) and e=1, 2.... 40. The \(\mu ^{e}\) defined as:
The cell is discretized into 40 elements. Since the thickness of elements is relatively small, therefore, \(w^{(e)}\) can be interpolated by a linear function as given below:
The equation (A2) is expressed as:
where \(P^T=\begin{bmatrix} 1&\quad x \end{bmatrix}\), \(C^{(e)}=\begin{bmatrix} c_1\\ c_2 \end{bmatrix}\).
Substituting the nodal values for each element in equation (A2),
where \(\bar{w}^{(e)}=\begin{bmatrix} w_i\\ w_j \end{bmatrix}\), \(P^{(e)}=\begin{bmatrix} 1&{} \quad x_i\\ 1&{} \quad x_j \end{bmatrix}\),
where
From equation (A3) and (A7) we obtain,
and
Substituting equation (A8) and (A9) in equation (A1), integrations are evaluated to obtain \(A^{(e)}\) for each element,
Then the equation (A12) can be extremize as given below:
where
From equation (A13) we get a system of first order linear differential equations expressed as:
Here \(\bar{w}^{(e)}\)=\(\left[ w_{1},w_{2},w_{3},\ldots ,w_{41}\right]\). \(\bar{A}\) and \(\bar{B}\) represents the system matrices and \(\bar{C}\) represents a system vector. Temporal system given in equation (A14) is solved using Crank Nicolson scheme and simulated on MATLAB to obtain the numerical results.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vaishali, Adlakha, N. Model of Calcium Dynamics Regulating \(IP_{3}\), ATP and Insulin Production in a Pancreatic \(\beta\)-Cell. Acta Biotheor 72, 2 (2024). https://doi.org/10.1007/s10441-024-09477-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10441-024-09477-x