Abstract
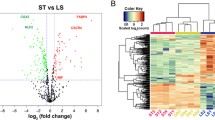
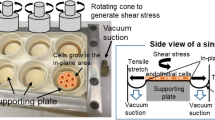
This study demonstrated the effects of the directionality of oscillatory wall shear stress (WSS) on proliferation and proatherogenic gene expression (I-CAM, E-Selectin, and IL-6) in the presence of inflammatory mediators leukotriene B4 (LTB4) and bacterial lipopolysaccharide (LPS) from endothelial cells grown in an orbiting culture dish. Computational fluid dynamics (CFD) was applied to quantify the flow in the dish, while an analytical solution representing an extension of Stokes second problem was used for validation. Results indicated that WSS magnitude was relatively constant near the center of the dish and oscillated significantly (0–0.9 Pa) near the side walls. Experiments showed that LTB4 dominated the shear effects on cell proliferation and area. Addition of LPS didn’t change proliferation, but significantly affected cell area. The expression of I-CAM1, E-Selectin and IL-6 were altered by directional oscillatory shear index (DOSI, a measure of the biaxiality of oscillatory shear), but not shear magnitude. The significance of DOSI was further reinforced by the strength of its interactions with other atherogenic factors. Hence, directionality of shear appears to be an important factor in regulating gene expression and provides a potential explanation of the propensity for increased vascular lesions in regions in the arteries with oscillating biaxial flow.








Similar content being viewed by others
Change history
22 June 2016
An erratum to this article has been published.
References
Aiello, R. J., D. Brees, P. A. Bourassa, L. Royer, S. Lindsey, T. Coskran, M. Haghpassand, and O. L. Francone. Increased atherosclerosis in hyperlipidemic mice with inactivation of ABCA1 in macrophages. Arterioscler. Thromb. Vasc. Biol. 22:630–637, 2002.
Berson R. E., M. R. Purcell and M. K. Sharp. Computationally determined shear on cells grown in orbiting culture dishes. Adv. Exp. Med. Biol. 614:189–198, 2008.
Bourdillon, M. C., R. N. Poston, C. Covacho, E. Chignier, G. Bricca, and J. L. McGregor. ICAM-1 deficiency reduces atherosclerotic lesions in double-knockout mice (ApoE(-/-)/ICAM-1(-/-)) fed a fat or a chow diet. Arterioscler. Thromb. Vasc. Biol. 20:2630–2635, 2000.
Chakraborty, A., S. Chakraborty, V. R. Jala, B. Haribabu, M. K. Sharp, and R. E. Berson. Effects of biaxial oscillatory shear stress on endothelial cell proliferation and morphology. Biotechnol. Bioeng. 109:695–707, 2012.
Chappell, D. C., S. E. Varner, R. M. Nerem, R. M. Medford, and R. W. Alexander. Oscillatory shear stress stimulates adhesion molecule expression in cultured human endothelium. Circ. Res. 82:532–539, 1998.
Cunningham, K. S., and A. I. Gotlieb. The role of shear stress in the pathogenesis of atherosclerosis. Lab. Invest. 85:9–23, 2005.
Dahl, A., M. Sultan, A. Jung, R. Schwartz, M. Lange, M. Steinwand, K. J. Livak, H. Lehrach, and L. Nyarsik. Quantitative PCR based expression analysis on a nanoliter scale using polymer nano-well chips. Biomed. Microdevices 9:307–314, 2007.
Frank, P. G., and M. P. Lisanti. ICAM-1: role in inflammation and in the regulation of vascular permeability. Am. J. Physiol. Heart Circ. Physiol. 295:H926–H927, 2008.
Fujihara, M., M. Muroi, K. Tanamoto, T. Suzuki, H. Azuma, and H. Ikeda. Molecular mechanisms of macrophage activation and deactivation by lipopolysaccharide: roles of the receptor complex. Pharmacol. Ther. 100:171–194, 2003.
Galkina, E., and K. Ley. Vascular adhesion molecules in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 27:2292–2301, 2007.
Inoguchi, H., T. Tanaka, Y. Maehara, and T. Matsuda. The effect of gradually graded shear stress on the morphological integrity of a huvec-seeded compliant small-diameter vascular graft. Biomaterials 28:486–495, 2007.
Ku, D. N., D. P. Giddens, C. K. Zarins, and S. Glagov. Pulsatile flow and atherosclerosis in the human carotid bifurcation. Positive correlation between plaque location and low oscillating shear stress. Arteriosclerosis 5:293–302, 1985.
Levesque, M. J., and R. M. Nerem. The elongation and orientation of cultured endothelial cells in response to shear stress. J. Biomech. Eng. 107:341–347, 1985.
Libby, P. Inflammation in atherosclerosis. Nature 420:868–874, 2002.
Mehrabian, M., H. Allayee, J. Wong, W. Shi, X. P. Wang, Z. Shaposhnik, C. D. Funk, and A. J. Lusis. Identification of 5-lipoxygenase as a major gene contributing to atherosclerosis susceptibility in mice. Circ. Res. 91:120–126, 2002.
Mehrabian, M., J. Wong, X. Wang, Z. Jiang, W. Shi, A. M. Fogelman, and A. J. Lusis. Genetic locus in mice that blocks development of atherosclerosis despite extreme hyperlipidemia. Circ. Res. 89:125–130, 2001.
Nakashima, Y., E. W. Raines, A. S. Plump, J. L. Breslow, and R. Ross. Upregulation of VCAM-1 and ICAM-1 at atherosclerosis-prone sites on the endothelium in the ApoE-deficient mouse. Arterioscler. Thromb. Vasc. Biol. 18:842–851, 1998.
Nerem, R. M., M. J. Levesque, and J. F. Cornhill. Vascular endothelial morphology as an indicator of the pattern of blood flow. J. Biomech. Eng. 103:172–176, 1981.
Owatverot, T. B., S. J. Oswald, Y. Chen, J. J. Wille, and F. C. Yin. Effect of combined cyclic stretch and fluid shear stress on endothelial cell morphological responses. J. Biomech. Eng. 127:374–382, 2005.
Pfenniger, A., M. J. Meens, R. M. Pedrigi, B. Foglia, E. Sutter, G. Pelli, V. Rochemont, T. V. Petrova, R. Krams, and B. R. Kwak. Shear stress-induced atherosclerotic plaque composition in ApoE(-/-) mice is modulated by connexin37. Atherosclerosis 243:1–10, 2015.
Ridger, V., R. Krams, A. Carpi, and P. C. Evans. Hemodynamic parameters regulating vascular inflammation and atherosclerosis: a brief update. Biomed. Pharmacother. 62:536–540, 2008.
Ross, R. Atherosclerosis is an inflammatory disease. Am. Heart J. 138:S419–S420, 1999.
Sawa, Y., T. Ueki, M. Hata, K. Iwasawa, E. Tsuruga, H. Kojima, H. Ishikawa, and S. Yoshida. LPS-induced IL-6, IL-8, VCAM-1, and ICAM-1 expression in human lymphatic endothelium. J. Histochem. Cytochem. 56:97–109, 2008.
Schieffer, B., T. Selle, A. Hilfiker, D. Hilfiker-Kleiner, K. Grote, U. J. Tietge, C. Trautwein, M. Luchtefeld, C. Schmittkamp, S. Heeneman, M. J. Daemen, and H. Drexler. Impact of interleukin-6 on plaque development and morphology in experimental atherosclerosis. Circulation 110:3493–3500, 2004.
Schuett, H., M. Luchtefeld, C. Grothusen, K. Grote, and B. Schieffer. How much is too much? Interleukin-6 and its signalling in atherosclerosis. Thromb. Haemost. 102:215–222, 2009.
Sukovich, D. A., K. Kauser, F. D. Shirley, V. DelVecchio, M. Halks-Miller, and G. M. Rubanyi. Expression of interleukin-6 in atherosclerotic lesions of male ApoE-knockout mice: inhibition by 17beta-estradiol. Arterioscler. Thromb. Vasc. Biol. 18:1498–1505, 1998.
Sumagin, R., E. Lomakina, and I. H. Sarelius. Leukocyte-endothelial cell interactions are linked to vascular permeability via ICAM-1-mediated signaling. Am. J. Physiol. Heart Circ. Physiol. 295:H969–H977, 2008.
Thomas, J. M., A. Chakraborty, M. K. Sharp, and R. E. Berson. Spatial and temporal resolution of shear in an orbiting petri dish. Biotechnol. Prog. 27:460–465, 2011.
VanderLaan, P. A., C. A. Reardon, and G. S. Getz. Site specificity of atherosclerosis: site-selective responses to atherosclerotic modulators. Arterioscler. Thromb. Vasc. Biol. 24:12–22, 2004.
Venables, W. N., and B. D. Ripley. Modern Applied Statistics with S. New York: Springer, 2002.
Yamaguchi, T., Y. Yamamoto, and H. Liu. Computational mechanical model studies on the spontaneous emergent morphogenesis of the cultured endothelial cells. J. Biomech. 33:115–126, 2000.
Zhou, J., Y. S. Li, and S. Chien. Shear stress-initiated signaling and its regulation of endothelial function. Arterioscler. Thromb. Vasc. Biol. 34:2191–2198, 2014.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Associate Editor Kerry Hourigan oversaw the review of this article.
An erratum to this article is available at https://doi.org/10.1007/s10439-016-1665-8.
Rights and permissions
About this article
Cite this article
Chakraborty, A., Chakraborty, S., Jala, V.R. et al. Impact of Bi-Axial Shear on Atherogenic Gene Expression by Endothelial Cells. Ann Biomed Eng 44, 3032–3045 (2016). https://doi.org/10.1007/s10439-016-1626-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-016-1626-2