Abstract
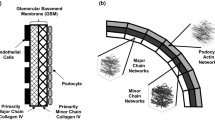
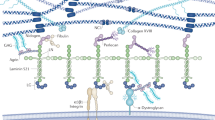
Collagen IV networks in the glomerular basement membrane (GBM) are essential for the maintenance and regulation of blood filtration in the kidneys. The GBM contains two different types of collagen IV networks: [α1(IV)]2α2(IV) and α3(IV)α4(IV)α5(IV), the latter of which has a higher number of supercoils (two or more collagens coiling around each other). To investigate the effects of supercoiling on the mechanical and permeability properties of collagen IV networks, we generated model collagen IV networks in the GBM and reconnected them to create different levels of supercoiling. We found that supercoiling greatly increases the stiffness of collagen IV networks but only minimally decreases the permeability. Also, doubling the amount of supercoils in a network had a bigger effect than doubling the stiffness of the supercoils. Our results suggest that the formation of supercoils is a specialized mechanism by the GBM that provides with a network stiff and strong enough to withstand the high hydrostatic pressures of filtration, yet porous enough that filtration is not hindered. Clinically, understanding the effects of supercoiling gives us insight into the mechanisms of GBM failure in some disease states where the normal collagen IV structure is disrupted.








Similar content being viewed by others
Abbreviations
- GBM:
-
Glomerular basement membrane
- RVE:
-
Representative volume element
- n i :
-
Node number
- α s :
-
Smallest angle
- M :
-
Midline
- p m :
-
Percent of length of midline/supercoil ratio
- r :
-
Radius
- F :
-
Force (N)
- E :
-
Elastic modulus (GPa)
- E sc :
-
Supercoil elastic modulus (GPa)
- A :
-
Collagen IV cross-sectional area (nm2)
- A sc :
-
Supercoil cross-sectional area (nm2)
- B :
-
Non-linearity parameter (Unitless)
- e G :
-
Green strain (Unitless)
- λ :
-
Stretch ratio (Unitless)
- σ ij :
-
Cauchy stress tensor (MPa)
- V :
-
Volume of RVE (nm3)
- x i :
-
Boundary nodal position (nm)
- Φ :
-
Collagen IV volume fraction (Unitless)
- l :
-
Length of collagen IV segment (nm)
- d ij :
-
Drag coefficient tensor of single segment (nm−1)
- R ij :
-
Direction cosines matrix (rad)
- C ij :
-
Coefficients matrix of drag (nm)
- V n :
-
Volume of collagen IV network (nm3)
- D ij :
-
Drag coefficient tensor of whole network (nm−2)
- k ij :
-
Darcy permeability (nm2)
References
Abrahamson, D. Development of kidney glomerular endothelial cells and their role in basement membrane assembly. Organogenesis 5:275–287, 2009.
Barocas, V. H., K. D. Dorfman, and Y. Segal. A model of strain-dependent glomerular basement membrane maintenance and its potential ramifications in health and disease. J. Biomech. Eng. 134(081006):1–8, 2012.
Bolton, G. R., W. M. Deen, and B. S. Daniels. Assessment of the charge selectivity of glomerular basement membrane using Ficoll sulfate. Am. J. Physiol. 274:F889–F896, 1998.
Brenner, B. M., C. Baylis, and W. M. Deen. Transport of molecules across renal glomerular capillaries. Physiol. Rev. 56:502–534, 1976.
Chandran, P. L., and V. H. Barocas. Affine versus non-affine fibril kinematics in collagen networks: theoretical studies of network behavior. J. Biomech. Eng. 128:259–270, 2006.
Chandran, P. L., and V. H. Barocas. Deterministic material-based averaging theory model of collagen gel micromechanics. J. Biomech. Eng. 129:137–147, 2007.
Chen, X., F. Wudl, A. Mal, H. Shen, and S. Nutt. New thermally remendable highly cross-linked polymeric materials. Macromolecules 36:1802–1807, 2003.
Daniels, B., E. Hauser, W. Deen, and T. Hostetter. Glomerular basement membrane: in vitro studies of water and protein permeability. Am. J. Physiol. 262:F919–F926, 1992.
Deen, W. M., M. J. Lazzara, and B. D. Myers. Structural determinants of glomerular permeability. Am. J. Physiol. Renal Physiol. 281:F579–F596, 2001.
Drummond, J. E., and M. I. Tahir. Laminar viscous flow through regular arrays of parallel solid cylinders. Int. J. Multiph. Flow 10:515–540, 1984.
Edwards, A., B. S. Daniels, and W. M. Deen. Hindered transport of macromolecules in isolated glomeruli. II. Convection and pressure effects in basement membrane. Biophys. J. 72:214–222, 1997.
Endlich, N., and K. Endlich. The challenge and response of podocytes to glomerular hypertension. Semin. Nephrol. 32:327–341, 2012.
Girton, T., T. Oegema, and R. Tranquillo. Exploiting glycation to stiffen and strengthen tissue equivalents for tissue engineering. J. Biomed. Mater. Res. 46:87–92, 1999.
Gunwar, S., F. Ballester, M. E. Noelken, Y. Sado, Y. Ninomiya, and B. G. Hudson. Glomerular basement membrane. Identification of a novel disulfide-cross-linked network of alpha3, alpha4, and alpha5 chains of type IV collagen and its implications for the pathogenesis of Alport syndrome. J. Biol. Chem. 273:8767–8775, 1998.
Gyoneva, L., Y. Segal, K. D. Dorfman, and V. H. Barocas. Mechanical response of wild-type and Alport murine lens capsules during osmotic swelling. Exp. Eye Res. 113:87–91, 2013.
Hadi, M. F., and V. H. Barocas. Microscale fiber network alignment affects macroscale failure behavior in simulated collagen tissue analogs. J. Biomech. Eng. 135(021026):1–8, 2013.
Haraldsson, B., J. Nystrom, and W. Deen. Properties of the glomerular barrier and mechanisms of proteinuria. Physiol. Rev. 88:451–487, 2008.
Hironaka, K., H. Makino, Y. Yamasaki, and Z. Ota. Renal basement membranes by ultrahigh resolution scanning electron microscopy. Kidney Int. 43:334–345, 1993.
Hofmann, H., T. Voss, K. Kühn, and J. Engel. Localization of flexible sites in thread-like molecules from electron micrographs: Comparison of interstitial, basement membrane and intima collagens. J. Mol. Biol. 172:325–343, 1984.
Hudson, B. G., R. Kalluri, S. Gunwar, M. Weber, F. Ballester, J. K. Hudson, M. E. Noelken, M. Sarras, W. R. Richardson, J. Saus, et al. The pathogenesis of Alport syndrome involves type IV collagen molecules containing the alpha 3 (IV) chain: evidence from anti-GBM nephritis after renal transplantation. Kidney Int 42:179–187, 1992.
Hudson, B. G., S. T. Reeders, and K. Tryggvason. Type IV collagen: structure, gene organization, and role in human diseases. J. Biol. Chem. 268:26033–26036, 1993.
Hulmes, D. J. S. Building collagen molecules, fibrils, and suprafibrillar structures. J. Struct. Biol. 137:2–10, 2002.
Kalluri, R., C. F. Shield, P. Todd, B. G. Hudson, and E. G. Neilson. Isoform switching of type IV collagen is developmentally arrested in X-linked Alport syndrome leading to increased susceptibility of renal basement membranes to endoproteolysis. J. Clin. Invest. 99:2470–2478, 1997.
Kashtan, C. E. Alport syndrome: an inherited disorder of renal, ocular, and cochlear basement membranes. Medicine (Baltimore) 78:338, 1999.
Kefalides, N. A., and B. Denduchisi. Structural components of epithelial and endothelial basement membranes. Biochemistry 8:4613–4621, 1969.
Khoshnoodi, J., V. Pedchenko, and B. G. Hudson. Mammalian collagen IV. Microsc. Res. Tech. 71:357–370, 2008.
Kim, K., Y. Kim, M. Gubler, M. Steffes, P. Lane, C. Kashtan, J. Crosson, and S. Mauer. Structural–functional relationships in Alport syndrome. J. Am. Soc. Nephrol. 5:1659–1668, 1995.
Knupp, C., and J. M. Squire. A new twist in the collagen story—the type VI segmented supercoil. EMBO J. 20:372–376, 2001.
Krag, S., and T. T. Andreassen. Mechanical properties of the human lens capsule. Prog. Retin. Eye Res. 22:749–767, 2003.
Kriz, W., M. Elger, P. Mundel, and K. V. Lemley. Structure-stabilizing forces in the glomerular tuft. J. Am. Soc. Nephrol. 5:1731–1739, 1995.
Kriz, W., W. Kretzler, A. P. Provoost, and I. Shirato. Stability and leakiness: opposing challenges to the glomerulus. Kidney Int. 49:1570–1574, 1996.
Kühn, K. Basement membrane (type IV) collagen. Matrix Biol. 14:439–445, 1995.
Lake, S. P., M. F. Hadi, V. K. Lai, and V. H. Barocas. Mechanics of a fiber network within a non-fibrillar matrix: model and comparison with collagen-agarose co-gels. Ann. Biomed. Eng. 40:2111–2121, 2012.
Lee, K., and D. Mooney. Hydrogels for tissue engineering. Chem. Rev. 101:1869–1879, 2001.
Lyubchenko, Y., and L. Shlyakhtenko. Visualization of supercoiled DNA with atomic force microscopy in situ. Proc. Natl. Acad. Sci. USA 94:496–501, 1997.
Martinez-Hernandez, A., and P. Amenta. The basement membrane in pathology. Lab. Investig. 48:656, 1983.
Miner, J. H., and J. R. Sanes. Collagen IV alpha 3, alpha 4, and alpha 5 chains in rodent basal laminae: sequence, distribution, association with laminins, and developmental switches. J. Cell Biol. 127:879–891, 1994.
Mizuno, D., C. Tardin, C. F. Schmidt, and F. C. Mackintosh. Nonequilibrium mechanics of active cytoskeletal networks. Science 315:370–373, 2007.
Nachtrab, S., S. C. Kapfer, C. H. Arns, M. Madadi, K. Mecke, and G. E. Schröder-Turk. Morphology and linear-elastic moduli of random network solids. Adv. Mater. 23:2633–2637, 2011.
Orgel, J., T. Irving, A. Miller, and T. Wess. Microfibrillar structure of type I collagen in situ. Proc. Natl. Acad. Sci. USA 103:9001–9005, 2006.
Pan, N., and D. Brookstein. Physical properties of twisted structures. II. Industrial yarns, cords, and ropes. J. Appl. Polym. Sci. 83:610–630, 2002.
Pavenstädt, H., W. Kriz, and M. Kretzler. Cell biology of the glomerular podocyte. Physiol. Rev. 83:253–307, 2003.
Ruben, G. C., and P. D. Yurchenco. High resolution platinum-carbon replication of freeze-dried basement membrane. Microsc. Res. Technol. 28:13–28, 1994.
Sakai, F., and W. Kriz. The structural relationship between mesangial cells and basement membrane of the renal glomerulus. Anat. Embryol. (Berl) 176:373–386, 1987.
Sangani, A. S., and A. Acrivos. Slow flow past periodic arrays of cylinders with application to heat transfer. Int. J. Multiph. Flow 8:193–206, 1982.
Srinivasan, M., S. G. M. Uzel, A. Gautieri, S. Keten, and M. J. Buehler. Alport syndrome mutations in type IV tropocollagen alter molecular structure and nanomechanical properties. J. Struct. Biol. 168:503–510, 2009.
Stylianopoulos, T., A. Yeckel, J. J. Derby, X.-J. Luo, M. S. Shephard, E. A. Sander, and V. H. Barocas. Permeability calculations in three-dimensional isotropic and oriented fiber networks. Phys. Fluids 20(123601):2008, 1994.
Suleiman, H., L. Zhang, R. Roth, J. E. Heuser, J. H. Miner, A. S. Shaw, and A. Dani. Nanoscale protein architecture of the kidney glomerular basement membrane. Elife 2:01149, 2013.
Welling, L., M. Zupka, and D. Wellin. Mechanical properties of basement membrane. News Physiol. Sci. 10:30–35, 1995.
Wyss, H. M., J. M. Henderson, F. J. Byfield, L. A. Bruggeman, Y. Ding, C. Huang, J. H. Suh, T. Franke, E. Mele, M. R. Pollak, J. H. Miner, P. A. Janmey, D. A. Weitz, and R. T. Miller. Biophysical properties of normal and diseased renal glomeruli. Am. J. Physiol. Cell Physiol. 300:C397–C405, 2011.
Yurchenco, P. D., and G. C. Ruben. Basement membrane structure in situ: evidence for lateral associations in the type IV collagen network. J. Cell Biol. 105:2559–2568, 1987.
Yurchenco, P. D., and G. C. Ruben. Type IV collagen lateral associations in the EHS tumor matrix. Comparison with amniotic and in vitro networks. Am. J. Pathol. 132:278–291, 1988.
Yurchenco, P., and J. Schittny. Molecular architecture of basement membranes. FASEB J. 4:1577–1590, 1990.
Acknowledgments
This work was carried out in part using computing resources at the University of Minnesota Supercomputing Institute and was supported by the National Science Foundation Graduate Research Fellowship and Grant No. NSF CMMI 1300649. This work was also supported by resources and the use of facilities at the Minneapolis VA Health Care System.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Gerhard A. Holzapfel oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Gyoneva, L., Segal, Y., Dorfman, K.D. et al. Effect of Supercoiling on the Mechanical and Permeability Properties of Model Collagen IV Networks. Ann Biomed Eng 43, 1695–1705 (2015). https://doi.org/10.1007/s10439-014-1187-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-014-1187-1