Abstract
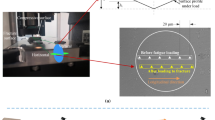
The purpose of this work was to investigate differences which may exist in the crack resistance of the microstructural bone tissues, i.e., osteonal, interstitial and trabecular bone. Indentations, using varying loads were used to initiate cracks of the same size scale as those which exist habitually in bone. The crack lengths and corresponding toughness values are presented for each of the tissues. Specimens were prepared using standard nanoindentation preparation techniques. Young’s modulus and hardness were measured using a Berkovich tip, while cracks were produced using a cube-corner tip. Crack lengths were subsequently measured using scanning electron microscopy. Cracks produced at the same loads were significantly longer in trabecular bone than in interstitial and osteonal cortical bone. Similarly, within individual subjects, cracks produced in interstitial bone were longer than those produced in osteonal bone. These results provide significant experimental evidence that bone microstructural tissues exhibit differing resistance to crack growth and may help explain the incidence of more microcracks in interstitial than osteonal bone. The ability of the technique to distinguish differences between individual bone tissues is promising in an area where the focus has switched to the microscale, and in particular, to measures bone quality.







Similar content being viewed by others
References
Ammann, P., and R. Rizzoli. Bone strength and its determinants. Osteoporos. Int. 14(3):S13–S18, 2003.
Antis, G. R., P. Chantikul, B. R. Lawn, and D. B. Marshall. A critical evaluation of indentation techniques for fracture toughness: I. Direct crack measurements. J. Am. Ceram. Soc. 64(9):533–538, 1981.
Bevill, G., S. K. Eswaran, A. Gupta, P. Papadopoulos, and T. M. Keaveny. Influence of bone volume fraction and architecture on computed large-deformation failure mechanisms in human trabecular bone. Bone 39(6):1218–1225, 2006.
Boivin, G., and P. J. Meunier. The degree of mineralization of bone tissue measured by computerized quantitative contact microradiography. Calcif. Tissue Int. 70(6):503–511, 2002.
Boyce, H. M., D. P. Fyhrie, M. C. Glotkowski, E. L. Radin, and M. B. Schaffler. Damage type and strain mode associations in human compact bone bending fatigue. J. Orthop. Res. 16:322–329, 1998.
Burr, D. B., and M. Hooser. Alterations to the en bloc basic fuchsin staining protocol for the demonstration of microdamage produced in vivo. Bone 17(4):431–433, 1995.
Burr, D. B., and T. Stafford. Validity of the bulk-staining technique to separate artifactual from in vivo bone damage. Clin. Orthop. Relat. Res. 260:305–308, 1990.
Currey, J. D., K. Brear, and P. Zioupos. The effects of ageing and changes in mineral content in degrading the toughness of human femora. J. Biomech. 29(2):257–260, 1996.
Fan, Z., P. Smith, F. Rauch, and G. F. Harris. Nanoindentation as a means of distinguishing clinical type of osteogenesis imperfecta. Compos. Part B: Eng. 38:411–415, 2007.
Fan, Z., J. G. Swadener, J. Y. Rho, M. E. Roy, and G. M. Pharr. Anisotropic properties of human tibial cortical bone as measured by nanoindentation. J. Orthop. Res. 20(4):806–810, 2002.
Fazzalari, N. L., and I. H. Parkinson. Fractal properties of cancellous bone of the iliac crest in vertebral crush fracture. Bone 23(1):53–57, 1998.
Gong, J. K., J. S. Arnold, and S. H. Cohn. Composition of trabecular and cortical bone. Anat. Rec. 149:325–331, 1964.
Hansma, P., P. Turner, B. Drake, E. Yurtsev, A. Proctor, P. Mathews, J. Lelujian, C. Randall, J. Adams, and R. Jungmann. The bone diagnostic instrument II. Indentation distance increase. Rev. Sci. Instrum. 79(6):064303, 2008.
Hassan, R., A. A. Caputo, and R. F. Bunshah. Fracture toughness of human enamel. J. Dent. Res. 60(4):820–827, 1981.
Heinrich, C., A. M. Wass, and A. S. Wineman. Determination of material properties using nanoindentation and multiple indenter tips. Int. J. Solids Struct. 46:365–376, 2009.
Hodgskinson, R., J. D. Currey, and G. P. Evans. Hardness, an indicator of the mechanical competence of cancellous bone. J. Orthop. Res. 7(5):754–758, 1989.
Imbeni, V., J. J. Kruzic, G. W. Marshall, S. J. Marshall, and R. O. Ritchie. The dentin–enamel junction and the fracture of human teeth. Nat. Mater. 4(3):229–232, 2005.
Koester, K. J., J. W. Ager, and R. O. Ritchie. The true toughness of human cortical bone measured with realistically short cracks. Nat. Mater. 7:672–677, 2008.
Kuhn, J. L., S. A. Goldstein, K. Choi, M. London, L. A. Feldkamp, and L. S. Matthews. Comparison of the trabecular and cortical tissue moduli from human iliac crests. J. Orthop. Res. 7(6):876–884, 1989.
Martin, R., O. Yeh, and D. Fyhrie. On sampling bones for microcracks. Bone 40(4):1159–1165, 2007.
McCalden, R., J. McGeough, M. Barker, and C. Court-Brown. Age-related changes in the tensile properties of cortical bone. The relative importance of changes in porosity, mineralization, and microstructure. J. Bone Joint Surg. (Am.) 75(8):1193–1205, 1993.
Meunier, P. J., and G. Boivin. Bone mineral density reflects bone mass but also the degree of mineralization of bone: therapeutic implications. Bone 21(5):373–377, 1997.
Morgan, E. F., H. H. Bayraktar, and T. M. Keaveny. Trabecular bone modulus-density relationships depend on anatomic site. J. Biomech. 36(7):897–904, 2003.
Mullins, L. P., M. S. Bruzzi, and P. McHugh. Measurement of the microstructural fracture toughness of cortical bone using indentation fracture. J. Biomech. 40(14):3285–3288, 2007.
Mullins, L. P., M. S. Bruzzi, and P. E. McHugh. Calibration of a constitutive model for the post-yield behaviour of cortical bone. J. Mech. Behav. Biomed. Mater. 2(5):460–470, 2009.
Nalla, R. K., J. J. Kruzic, and R. O. Ritchie. On the origin of the toughness of mineralized tissue: microcracking or crack bridging? Bone 34(5):790–798, 2004.
O’Brien, F. J., D. A. Hardiman, J. G. Hazenberg, M. V. Mercy, S. Moshin, D. Taylor, and T. C. Lee. The behaviour of microcracks in compact bone. Eur. J. Morphol. 42(1/2):71–79, 2005.
O’Brien, F. J., D. Taylor, G. R. Dickson, and T. C. Lee. Visualisation of three-dimensional microcracks in compact bone. J. Anat. 197:413–420, 2000.
O’Brien, F. J., D. Taylor, and T. C. Lee. Microcrack accumulation at different intervals during fatigue testing of compact bone. J. Biomech. 36(7):973–980, 2003.
O’Brien, F. J., D. Taylor, and T. C. Lee. The effect of bone microstructure on the initiation and growth of microcracks. J. Orthop. Res. 23(2):475–480, 2005.
Oliver, W. C., and G. M. Pharr. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J. Mater. Res. 7(6):1564–1583, 1992.
Quinn, D. G. On the Vickers indentation fracture toughness test. J. Am. Ceram. Soc. 90(3):673–680, 2007.
Rho, J. Y., and G. M. Pharr. Effects of drying on the mechanical properties of bovine femur measured by nanoindentation. J. Mater. Sci.: Mater. Med. 10:485–488, 1999.
Rho, J. Y., P. Zioupos, J. D. Currey, and G. M. Pharr. Microstructural elasticity and regional heterogeneity in human femoral bone of various ages examined by nano-indentation. J. Biomech. 35(2):189–198, 2002.
Stein, M. S., C. D. L. Thomas, S. A. Feik, J. D. Wark, and J. G. Clement. Bone size and mechanics at the femoral diaphysis across age and sex. J. Biomech. 31(12):1101–1110, 1998.
Vashishth, D. Rising crack-growth-resistance behavior in cortical bone: implications for toughness measurements. J. Biomech. 37(6):943–946, 2004.
Viguet-Carrin, S., P. Garnero, and P. D. Delmas. The role of collagen in bone strength. Osteoporos. Int. 17(3):319–336, 2006.
Wang, X., Y. J. Yoon, and H. Ji. A novel scratching approach for measuring age-related changes in the in situ toughness of bone. J. Biomech. 40(6):1401–1404, 2007.
Wasserman, N., J. Yerramshetty, and O. Akkus. Microcracks colocalize within highly mineralized regions of cortical bone tissue. Eur. J. Morphol. 42(1/2):43–51, 2005.
Yan, J., K. B. Clifton, J. Mecholsky, J. John, and R. L. Reep. Fracture toughness of manatee rib and bovine femur using a chevron-notched beam test. J. Biomech. 39(6):1066–1074, 2006.
Yeh, O. C., and T. M. Keaveny. Relative roles of microdamage and microfracture in the mechanical behavior of trabecular bone. J. Orthop. Res. 19(6):1001–1007, 2001.
Yeni, Y. N., C. U. Brown, Z. Wang, and T. L. Norman. The influence of bone morphology on fracture toughness of the human femur and tibia. Bone 21(5):453–459, 1997.
Zioupos, P. Accumulation of in vivo fatigue microdamage and its relation to biomechanical properties in ageing human cortical bone. J. Microsc. 201(2):270–278, 2001.
Zioupos, P. In vivo fatigue microcracks in human bone: Material properties of the surrounding bone matrix. Eur. J. Morphol. 42(1/2):31–41, 2005.
Zioupos, P., and J. D. Currey. Changes in the stiffness, strength, and toughness of human cortical bone with age. Bone 22(1):57–66, 1998.
Zysset, P. K., X. Edward Guo, C. Edward Hoffler, K. E. Moore, and S. A. Goldstein. Elastic modulus and hardness of cortical and trabecular bone lamellae measured by nanoindentation in the human femur. J. Biomech. 32(10):1005–1012, 1999.
Acknowledgments
The authors wish to acknowledge funding from the Programme for Research in Third Level Institutions (PRTLI), administered by the Higher Education Authority (HEA), Ireland. The vertebrae used in this study were provided by Trinity College Dublin and Royal College of Surgeons under the “Bone for Life” project in partnership with National University of Ireland, Galway.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mullins, L.P., Sassi, V., McHugh, P.E. et al. Differences in the Crack Resistance of Interstitial, Osteonal and Trabecular Bone Tissue. Ann Biomed Eng 37, 2574–2582 (2009). https://doi.org/10.1007/s10439-009-9797-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-009-9797-8