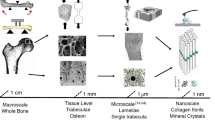
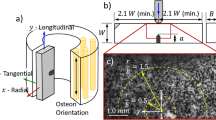
Abstract
The role of bone fatigue damage at the nanostructural level, and its effect on fatigue properties is an understudied and important subject. In this study, nanoindentation was used to probe the micro-mechanical properties of bovine tibiae subjected to fatigue loading in four-point bending. Indentation tests were conducted in the same 30 μm × 120 μm region before fatigue loading and after loading to fracture. Using an optical microscope and scanning electron microscope, the morphology of the initial residual indentation before fatigue loading appeared the same as that after loading to fracture. The mechanical properties calculated using nanoindentation were reduced modulus and hardness, the time constant based on creep, long-term creep viscosity and the dissipated work. Differences of each parameter before loading and post fracture were examined using paired t-tests. The results show that the reduced modulus decreased significantly (p = 9.47 × 10–3) by 7.62–15.16% after fracture whereas the time constant of creep increased slightly (p = 0.049) by 2.81–5.41%. There was no clear change in hardness or dissipated work with fatigue loading. Fatigue loading has the largest effect on bone’s reduced modulus compared to all other mechanical properties.









Similar content being viewed by others
Data availability
The raw/processed data required to reproduce these findings cannot be shared at this time as the data also forms part of an ongoing study.
References
Bentolila V, Boyce TM, Fyhrie DP, Drumb R, Skerry TM, Schaffler MB (1998) Intracortical remodeling in adult rat long bones after fatigue loading. Bone 23(3):275–281. https://doi.org/10.1016/S8756-3282(98)00104-5
Boyce TM, Fyhrie DP, Glotkowski MC, Radin EL, Schaffler MB (1998) Damage type and strain mode associations in human compact bone bending fatigue. J Orthop Res 16(3):322–329. https://doi.org/10.1002/jor.1100160308
Burket J, Gourion-Arsiquaud S, Havill LM, Baker SP, Boskey AL, van der Meulen MCH (2011) Microstructure and nanomechanical properties in osteons relate to tissue and animal age. J Biomech 44(2):277–284. https://doi.org/10.1016/j.jbiomech.2010.10.018
Burr DB (2019) Stress concentrations and bone microdamage: John Currey’s contributions to understanding the initiation and arrest of cracks in bone. Bone 127:517–525. https://doi.org/10.1016/j.bone.2019.07.015
Burr DB, Hooser M (1995) Alterations to the en bloc basic fuchsin staining protocol for the demonstration of microdamage produced in vivo. Bone 17(4):431–433. https://doi.org/10.1016/S8756-3282(95)00241-3
Burr DB, Martin RB, Schaffler MB, Radin EL (1985) Bone remodeling in response to in vivo fatigue microdamage. J Biomech 18(3):189–200. https://doi.org/10.1016/0021-9290(85)90204-0
Burr DB, Milgrom C (2000) Musculoskeletal fatigue and stress fractures. CRC Press, Boca Raton
Burr DB, Turner CH, Naick P, Forwood MR, Ambrosius W, Sayeed Hasan M, Pidaparti R (1998) Does microdamage accumulation affect the mechanical properties of bone? J Biomech 31(4):337–345. https://doi.org/10.1016/S0021-9290(98)00016-5
Chaudhry B, Ashton H, Muhamed A, Yost M, Bull S, Frankel D (2009) Nanoscale viscoelastic properties of an aligned collagen scaffold. J Mater Sci Mater Med 20(1):257. https://doi.org/10.1007/s10856-008-3574-3
Currey J (2005) Structural heterogeneity in bone: good or bad? J Musculoskelet Neuron Interact 5(4):317
Currey JD (1964) Some effects of ageing in human Haversian systems. J Anat 98(Pt 1):69–75
Donnelly E, Baker SP, Boskey AL, van der Meulen MCH (2006) Effects of surface roughness and maximum load on the mechanical properties of cancellous bone measured by nanoindentation. J Biomed Mater Res A 77A(2):426–435. https://doi.org/10.1002/jbm.a.30633
Draper ERC, Goodship AE (2003) A novel technique for four-point bending of small bone samples with semi-automatic analysis. J Biomech 36(10):1497–1502. https://doi.org/10.1016/S0021-9290(03)00129-5
Erben RG (1997) Embedding of bone samples in methylmethacrylate: an improved method suitable for bone histomorphometry, histochemistry, and immunohistochemistry. J Histochem Cytochem 45(2):307–313. https://doi.org/10.1177/002215549704500215
Faingold A, Cohen SR, Wagner HD (2012) Nanoindentation of osteonal bone lamellae. J Mech Behav Biomed Mater 9:198–206. https://doi.org/10.1016/j.jmbbm.2012.01.014
Fan Z, Rho J-Y (2003) Effects of viscoelasticity and time-dependent plasticity on nanoindentation measurements of human cortical bone. J Biomed Mater Res A 67A(1):208–214. https://doi.org/10.1002/jbm.a.10027
Feng L, Chittenden M, Schirer J, Dickinson M, Jasiuk I (2012) Mechanical properties of porcine femoral cortical bone measured by nanoindentation. J Biomech 45(10):1775–1782. https://doi.org/10.1016/j.jbiomech.2012.05.001
Fischer-Cripps AC (2004) A simple phenomenological approach to nanoindentation creep. Mater Sci Eng A 385(1):74–82. https://doi.org/10.1016/j.msea.2004.04.070
Fratzl P (2007) Biomimetic materials research: what can we really learn from nature’s structural materials? J R Soc Interface 4(15):637–642. https://doi.org/10.1098/rsif.2007.0218
Fratzl P, Gupta HS (2007) Nanoscale mechanisms of bone deformation and fracture. In: Bäuerlein E, Behrens P, Epple M (eds) Handbook of biomineralization. Wiley, Hoboken, pp 397–414
Fratzl P, Weinkamer R (2007) Nature’s hierarchical materials. Prog Mater Sci 52(8):1263–1334. https://doi.org/10.1016/j.pmatsci.2007.06.001
Frost HL (1960) Presence of microscopic cracks in vivo in bone. Henry Ford Hosp Med Bull 8(1):25–35
Hengsberger S, Ammann P, Legros B, Rizzoli R, Zysset P (2005) Intrinsic bone tissue properties in adult rat vertebrae: modulation by dietary protein. Bone 36(1):134–141. https://doi.org/10.1016/j.bone.2004.09.013
Hengsberger S, Kulik A, Zysset P (2002) Nanoindentation discriminates the elastic properties of individual human bone lamellae under dry and physiological conditions. Bone 30(1):178–184. https://doi.org/10.1016/S8756-3282(01)00624-X
Isaksson H, Nagao S, MaŁkiewicz M, Julkunen P, Nowak R, Jurvelin JS (2010) Precision of nanoindentation protocols for measurement of viscoelasticity in cortical and trabecular bone. J Biomech 43(12):2410–2417. https://doi.org/10.1016/j.jbiomech.2010.04.017
Iyo T, Maki Y, Sasaki N, Nakata M (2004) Anisotropic viscoelastic properties of cortical bone. J Biomech 37(9):1433–1437. https://doi.org/10.1016/j.jbiomech.2003.12.023
Jowsey J (1966) Studies of Haversian systems in man and some animals. J Anat 100(Pt 4):857–864
Koester KJ, Ager JW, Ritchie RO (2008) The true toughness of human cortical bone measured with realistically short cracks. Nat Mater 7(8):672–677. https://doi.org/10.1038/nmat2221
Lakes RS, Katz JL, Sternstein SS (1979) Viscoelastic properties of wet cortical bone—I Torsional and Biaxial Studies. J Biomech 12(9):657–678. https://doi.org/10.1016/0021-9290(79)90016-2
Les CM, Spence CA, Vance JL, Christopherson GT, Patel B, Turner AS, Divine GW et al (2004) Determinants of ovine compact bone viscoelastic properties: effects of architecture, mineralization, and remodeling. Bone 35(3):729–738. https://doi.org/10.1016/j.bone.2004.04.006
Les CM, Vance JL, Christopherson GT, Turner AS, Divine GW, Fyhrie DP (2005) Long-term ovariectomy decreases ovine compact bone viscoelasticity. J Orthop Res 23(4):869–876. https://doi.org/10.1016/j.orthres.2004.12.001
Lewis G, Nyman JS (2008) The use of nanoindentation for characterizing the properties of mineralized hard tissues: State-of-the art review. J Biomed Mater Res B Appl Biomater 87B(1):286–301. https://doi.org/10.1002/jbm.b.31092
Li S, Abdel-Wahab A, Silberschmidt VV (2013) Analysis of fracture processes in cortical bone tissue. Eng Fract Mech 110:448–458. https://doi.org/10.1016/j.engfracmech.2012.11.020
Locke M (2004) Structure of long bones in mammals. J Morphol 262(2):546–565. https://doi.org/10.1002/jmor.10282
Loundagin LL, Edwards WB (2020) Stressed volume around vascular canals explains compressive fatigue life variation of secondary osteonal bone but not plexiform bone. J Mech Behav Biomed Mater 111:104002. https://doi.org/10.1016/j.jmbbm.2020.104002
McCalden RW, McGeough JA, Barker MB, Court-Brown CM (1993) Age-related changes in the tensile properties of cortical bone. The relative importance of changes in porosity, mineralization, and microstructure. J Bone Joint Surg Am 75(8):1193–205. https://doi.org/10.2106/00004623-199308000-00009
Nalla RK, Kinney JH, Ritchie RO (2003) Effect of orientation on the in vitro fracture toughness of dentin: the role of toughening mechanisms. Biomaterials 24(22):3955–3968. https://doi.org/10.1016/S0142-9612(03)00278-3
Nalla RK, Kinney JH, Ritchie RO (2003) Mechanistic fracture criteria for the failure of human cortical bone. Nat Mater 2(3):164–168. https://doi.org/10.1038/nmat832
Nalla RK, Kruzic JJ, Ritchie RO (2004) On the origin of the toughness of mineralized tissue: microcracking or crack bridging? Bone 34(5):790–798. https://doi.org/10.1016/j.bone.2004.02.001
Norman J, Shapter JG, Short K, Smith LJ, Fazzalari NL (2008) Micromechanical properties of human trabecular bone: a hierarchical investigation using nanoindentation. J Biomed Mater Res A 87A(1):196–202. https://doi.org/10.1002/jbm.a.31766
Oliver WC, Pharr GM (1992) An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J Mater Res 7(6):1564–1583. https://doi.org/10.1557/JMR.1992.1564
Oliver WC, Pharr GM (2004) Measurement of hardness and elastic modulus by instrumented indentation: advances in understanding and refinements to methodology. J Mater Res 19(1):3–20. https://doi.org/10.1557/jmr.2004.19.1.3
Oyen ML (2005) Spherical indentation creep following ramp loading. J Mater Res 20(8):2094–2100. https://doi.org/10.1557/JMR.2005.0259
Pattin CA, Caler WE, Carter DR (1996) Cyclic mechanical property degradation during fatigue loading of cortical bone. J Biomech 29(1):69–79. https://doi.org/10.1016/0021-9290(94)00156-1
Qin Q-H, Qu C, Ye J (2005) Thermoelectroelastic solutions for surface bone remodeling under axial and transverse loads. Biomaterials 26(33):6798–6810. https://doi.org/10.1016/j.biomaterials.2005.03.042
Qin Q-H, Ye J-Q (2004) Thermoelectroelastic solutions for internal bone remodeling under axial and transverse loads. Int J Solids Struct 41(9):2447–2460. https://doi.org/10.1016/j.ijsolstr.2003.12.026
Qin QH (2013) Mechanics of cellular bone remodeling: coupled thermal, electrical, and mechanical field effects. CRC Press, Taylor & Francis, Boca Raton
Qu C, Qin Q-H, Kang Y (2006) A hypothetical mechanism of bone remodeling and modeling under electromagnetic loads. Biomaterials 27(21):4050–4057. https://doi.org/10.1016/j.biomaterials.2006.03.015
Rho J-Y, Pharr GM (1999) Effects of drying on the mechanical properties of bovine femur measured by nanoindentation. J Mater Sci Mater Med 10(8):485–488. https://doi.org/10.1023/A:1008901109705
Rho J-Y, Tsui TY, Pharr GM (1997) Elastic properties of human cortical and trabecular lamellar bone measured by nanoindentation. Biomaterials 18(20):1325–1330. https://doi.org/10.1016/S0142-9612(97)00073-2
Ritchie R, Buehler M, Hansma P (2009) Plasticity and toughness in bone. Phys Today. https://doi.org/10.1063/1.3156332
Ritchie RO (2011) The conflicts between strength and toughness. Nat Mater 10(11):817–822. https://doi.org/10.1038/nmat3115
Sasso M, Haïat G, Yamato Y, Naili S, Matsukawa M (2008) Dependence of ultrasonic attenuation on bone mass and microstructure in bovine cortical bone. J Biomech 41(2):347–355. https://doi.org/10.1016/j.jbiomech.2007.09.001
Seto J, Gupta HS, Zaslansky P, Wagner HD, Fratzl P (2008) Tough lessons from bone: extreme mechanical anisotropy at the mesoscale. Adv Funct Mater 18(13):1905–1911. https://doi.org/10.1002/adfm.200800214
Subit D, Dios EdPd, Valazquez-Ameijide J, Arregui-Dalmases C, and Crandall J (2011) Tensile material properties of human rib cortical bone under quasi-static and dynamic failure loading and influence of the bone microstucture on failure characteristics. Physics. http://arxiv.org/abs/1108.0390v2
Tai K, Dao M, Suresh S, Palazoglu A, Ortiz C (2007) Nanoscale heterogeneity promotes energy dissipation in bone. Nat Mater 6(6):454–462. https://doi.org/10.1038/nmat1911
Tertuliano OA, Greer JR (2016) The nanocomposite nature of bone drives its strength and damage resistance. Nat Mater 15(11):1195–1202. https://doi.org/10.1038/nmat4719
Trębacz H, Zdunek A, Cybulska J, Pieczywek P (2013) Effects of fatigue on microstructure and mechanical properties of bone organic matrix under compression. Australas Phys Eng Sci Med 36(1):43–54. https://doi.org/10.1007/s13246-013-0185-1
Vashishth D, Koontz J, Qiu SJ, Lundin-Cannon D, Yeni YN, Schaffler MB, Fyhrie DP (2000) In vivo diffuse damage in human vertebral trabecular bone. Bone 26(2):147–152. https://doi.org/10.1016/S8756-3282(99)00253-7
Wagermaier W, Klaushofer K, Fratzl P (2015) Fragility of bone material controlled by internal interfaces. Calcif Tissue Int 97(3):201–212. https://doi.org/10.1007/s00223-015-9978-4
Wu Z, Baker TA, Ovaert TC, Niebur GL (2011) The effect of holding time on nanoindentation measurements of creep in bone. J Biomech 44(6):1066–1072. https://doi.org/10.1016/j.jbiomech.2011.01.039
Wu Z, Ovaert TC, Niebur GL (2012) Viscoelastic properties of human cortical bone tissue depend on gender and elastic modulus. J Orthop Res 30(5):693–699. https://doi.org/10.1002/jor.22001
Yin L, Venkatesan S, Webb D, Kalyanasundaram S, Qin Q-H (2009) Effect of cryo-induced microcracks on microindentation of hydrated cortical bone tissue. Mater Charact 60(8):783–791. https://doi.org/10.1016/j.matchar.2009.01.004
Zhang J, Michalenko MM, Kuhl E, Ovaert TC (2010) Characterization of indentation response and stiffness reduction of bone using a continuum damage model. J Mech Behav Biomed Mater 3(2):189–202. https://doi.org/10.1016/j.jmbbm.2009.08.001
Zhang J, Niebur GL, Ovaert TC (2008) Mechanical property determination of bone through nano- and micro-indentation testing and finite element simulation. J Biomech 41(2):267–275. https://doi.org/10.1016/j.jbiomech.2007.09.019
Zimmermann EA, Gludovatz B, Schaible E, Busse B, Ritchie RO (2014) Fracture resistance of human cortical bone across multiple length-scales at physiological strain rates. Biomaterials 35(21):5472–5481. https://doi.org/10.1016/j.biomaterials.2014.03.066
Zimmermann EA, Launey ME, Barth HD, Ritchie RO (2009) Mixed-mode fracture of human cortical bone. Biomaterials 30(29):5877–5884. https://doi.org/10.1016/j.biomaterials.2009.06.017
Zimmermann EA, Launey ME, Ritchie RO (2010) The significance of crack-resistance curves to the mixed-mode fracture toughness of human cortical bone. Biomaterials 31(20):5297–5305. https://doi.org/10.1016/j.biomaterials.2010.03.056
Zimmermann EA, Ritchie RO (2015) Bone as a structural material. Adv Healthc Mater 4(9):1287–1304. https://doi.org/10.1002/adhm.201500070
Zysset PK, Edward Guo X, Edward Hoffler C, Moore KE, Goldstein SA (1999) Elastic modulus and hardness of cortical and trabecular bone lamellae measured by nanoindentation in the human femur. J Biomech 32(10):1005–1012. https://doi.org/10.1016/S0021-9290(99)00111-6
Acknowledgements
The authors would like to express their gratitude to EditSprings (http://www.editsprings.com/) for the expert linguistic services provided.
Funding
This work is financially supported by the National Natural Science Foundation of China (Grant Nos. 11972247 and 11772204).
Author information
Authors and Affiliations
Contributions
XJM: Data curation, Formal analysis, Investigation, Validation, Writing-original draft. QHQ: Conceptualization, Supervision, Writing-review & editing, Validation. CYQ: Methodology, Resources, Writing-original draft, Writing-review & editing, Supervision, Project administration, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare. The authors declare that they do not have any competing financial or associative interests that could influence the work submitted.
Ethical approval
Cattle bones used in this study were taken from local slaughterhouses, and no animals were euthanized specifically for this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meng, X., Qin, Q. & Qu, C. Changes in bone’s micromechanical properties caused by fatigue fracture. Int J Fract 231, 243–255 (2021). https://doi.org/10.1007/s10704-021-00584-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10704-021-00584-1