Abstract
The effective longitudinal diffusion of nanovectors along non-permeable and permeable capillaries has been studied considering the contribution of molecular and convective diffusion based on the Taylor's theory of shear dispersion. The problem is of importance in the transport of nanovectors used for the intravascular delivery of drugs and contrast agents.
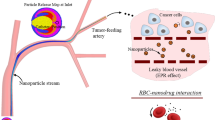
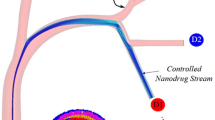
It has been shown that for a given capillary size and hemodynamic conditions a critical radius \(a_{\rm cr}\) exists for which the effective longitudinal diffusion along the capillary has a minimum: Nanovectors with \(a<a_{\rm cr}\) diffuse mainly by Brownian diffusion whereas nanovectors with \(a>a_{\rm cr}\) diffuse mainly by convection and the effective diffusion coefficient grows with \(a\). In permeable conduits, the effective diffusion reduces significantly compared to normal non-leaky vessels and it has been derived that \(a_{\rm cr}\) grows almost linearly with the hydraulic permeability \(L_{\rm p}\) of blood vessels.
It has been shown that the blood conduits with the largest effective longitudinal diffusivity could be preferentially targeted by the circulating vectors. Based on these findings, the following strategies are proposed to increase the number of nanovectors targeting the tumor vessels: (i) The use of nanovectors with a critical radius for normal vessels, (ii) the injecting of bolus of nanovectors with different radii, and (iii) the normalization of the tumor vasculature. Finally, it has been emphasized that the size of the vector should be selected depending on the body district where the tumoral mass is developing and on the type, malignancy, and state of the tumor.









Similar content being viewed by others
REFERENCES
Alivisatos, P. Semiconductor clusters, nanocrystals, and quantum dots. Science 271:933–937, 1996.
Aris, R. On the dispersion of a solute in a fluid flowing through a tube. Proc. R. Soc. Lond. A 235(1200):67–77, 1956.
Bloch, S. H., M. Wan, P. A. Dayton, and K. W. Ferrara. Optical observation of lipid- and polymer-shelled ultrasound microbubble contrast agents. Appl. Phys. Lett. 84(4):631–633, 2004.
Brizel, D. M., B. Klitzman, J. M. Cook, J. Edwards, G. Rosner, and M. W. Dewhirst. A comparison of tumor and normal tissue microvascular hematocrits and red cell fluxes in a rat window chamber model. Int. J. Radiat. Oncol. Biol. Phys. 15;25(2):269–276, 1993.
Cohen, M. H., K. Melnik, A. Boiasrki, M. Ferrari, F. J. Martin. Microfabrication of silicon-based nanoporous particulates for medical applications. Biomed. Microdevices 5:253–259, 2003.
Dash, R. K., G. Jayaraman, and K. N. Mehta. Shear augmented dispersion of a solute in a casson fluid flowing in a conduit. Ann. Biomed. Eng. 28:373–385, 2000.
Duncan, R. The dawning era of polymer therapeutics. Nat. Rev./Drug Discov. 2:347–360, 2003.
Ferrari, M. Cancer nanotechnology: Opportunities and challenges. Nat. Rev. Cancer 5:161, 2005.
Fung, Y. C., Biomechanics. Mechanical Properties of Living Tissues, 2nd ed. New York: Springer, 1993.
Gill, W. N., and R. Sankarasubramanian. Exact analysis of unsteady convective diffusion. Proc. R. Soc. Lond. A 316(1526):341–350, 1970.
Ganong, W. F. Review of Medical Physiology, 21st ed. New York: Lange Medical Books/McGraw-Hill, Medical Publishing Division, 2003.
Goldsmith, H. L., and S. Mason. The microrheology of dispersions. In: Rheology: Theory and Applications, Vol. 4, edited by F. R. Eirich. New York: Academic Press, 1968, pp. 85–250.
Goldsmith, H. L., and V. T. Turitto. Rheological aspects of thrombosis and haemostasis: Basic principles and applications. Thromb. Haemost. 55:415–435, 1986.
Hashizume, H., P. Baluk, S. Morikawa, J. W. McLean, G. Thurston, S. Roberge, R. K. Jain, and M. D. McDonald. Openings between defective endothelial cells explain tumor vessel leakiness. Am. J. Pathol. 156(4), 2000.
Hirsch, L. R., N. J. Halas, and J. L. West. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc. Natl. Acad. Sci. USA 100:13549–13554, 2003.
Jain, R. K. Transport of molecules, particles, and cells in solid tumors. Annu. Rev. Biomed. Eng. 1:241–263, 1999.
Netti, P. A., S. Roberge, Y. Boucher, L. T. Baxter, and R. K. Jain. Effect of transvascular fluid exchange on pressure–flow relationship in tumors: A proposed mechanism for tumor blood flow heterogeneity. Microvasc. Res. 52(1):27–46, 1996.
Oyewumi, M. O., and R. J. Mumper. Comparison of cell uptake, biodistribution and tumor retention of folate-coated and PEG-coated gadolinium nanoparticles in tumor-bearing mice. J. Control. Release 24:613–626, 2004.
Sevick, E. M., and R. K. Jain. Measurement of capillary filtration coefficient in a solid tumor. Cancer Res. 51:1352–1355, 1991.
Taylor, G. Dispersion of soluble matter in solvent flowing slowly through a tube. Proc. R. Soc. Lond. A 219(1137):186–203, 1953.
Winter, P. M., S. A. Wickline, and G. M. Lanza. Molecular imaging of angiogenesis in nascent Vx-2 rabbit tumors using a novel alpha(nu)beta3-targeted nanoparticle and 1.5 tesla magnetic resonance imaging. Cancer Res. 63:5838–5843, 2003.
Tong, R. T., Y. Boucher, S. V. Kozin, F. Winkler, D. J. Hicklin, R. K. Jain. Vascular normalization by vascular endothelial growth factor receptor 2 blockade induces a pressure gradient across the vasculature and improves drug penetration in tumors. Cancer Res. 64(11):3731–3736, 2004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Decuzzi, P., Causa, F., Ferrari, M. et al. The Effective Dispersion of Nanovectors Within the Tumor Microvasculature. Ann Biomed Eng 34, 633–641 (2006). https://doi.org/10.1007/s10439-005-9072-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-9072-6