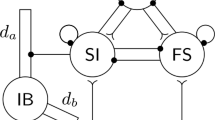
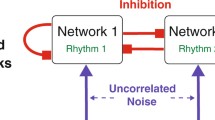
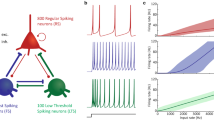
The Mapped Clock Oscillator (MCO) model is a representation of omnipresent transmembrane voltage oscillations in excitable cells. We present a generalized version of the MCO that can model neuronal electrical oscillations, both labile and omnipresent, entirely within the framework of a system of ordinary differential equations. The previously described MCO was a second-order system, whereas the model presented here, which we call the composite MCO (cMCO) is a fourth-order system. Furthermore, we show how this cMCO can also be adapted to describe a pair of cells that forms a functional unit, as illustrated here by a model of the CA3 pyramidal cell and its basket cell interneuron feedback loop. The model was able to reproduce the high frequencies (super gamma) and possibly chaotic dynamics observed in the biological system.
















Similar content being viewed by others
REFERENCES
Bardakjian, B. L., R. Aschenbrenner-Scheibe, J. L. Perez-Velazquez, and P. L. Carlen. Transmembrane voltage oscillations in CA3 neurons. In: Proceedings of the 17th International Conference on IEEE/EMBS, Montreal, Quebec, September 20–23, 1995, pp. 1503–1504.
Bardakjian, B. L., and N. E. Diamant. A Mapped Clock Oscillator model for transmembrane electrical activity in excitable cells. J. Theor. Biol. 166:225–235, 1994.
Bardakjian, B. L., T. Y. El-Sharkawy, and N. E. Diamant. On a population of labile synthesized relaxation oscillators. IEEE Trans. Biomed. Eng. BME-30:696–701, 1983.
Bardakjian, B. L., T. Y. El-Sharkawy, and N. E. Diamant. Interaction of coupled nonlinear oscillators having different intrinsic resting levels. J. Theor. Biol. 106:9–23, 1984.
Bardakjian, B. L., and S. K. Sarna. A computer model of human colonic electrical control activity (ECA). IEEE Trans. Biomed. Eng. 27:193–202, 1980.
Bardakjian, B. L., S. K. Sarna, and N. E. Diamant. Compositesynthesized relaxation oscillators: Application to modeling of colonic electrical control and response activity. J. Gastrointest. Motil. 2:109–116, 1990.
Basar, E. EEG-Brain Dynamics: Relation Between EEG and Brain Evoked Potentials. Amsterdam: Elsevier, 1980.
Berridge, M. J., and P. E. Rapp. A comparative survey of the function, mechanism and control of cellular oscillators. J. Exp. Biol. 81:217–279, 1979.
Chay, T. R. Bursting excitable cell models by a slow calcium current. J. Theor. Biol. 142:305–315, 1990.
Cole, K. S., and J. W. Moore. Potassium ion current in the squid giant axon: Dynamic characteristic. Biophys. J. 1:1–14, 1960.
Ermentrout, G. B., and N. Kopell. Frequency plateaus in a chain of weakly coupled oscillators: I. SIAM J. Math. Anal. 15:215–237, 1984.
Ermentrout, G. B., and N. Kopell. Multiple pulse interactions and averaging in systems of coupled neural oscillators. J. Math. Biol. 29:195–217, 1991.
Ermentrout, G. B., and N. Kopell. Fine structure of neural spiking and synchronization in the presence of conduction delays. Proc. Natl. Acad. Sci. U.S.A. 95:1259–1264, 1998.
Ermentrout, B., M. Pascal, and B. Gutkin. The effects of spike frequency adaptation and negative feedback on the synchronization of neural oscillators. Neural Comput. 13:1285–1310, 2001.
Fitzhugh, R. Impulses and physiological states in theoretical models of nerve membranes. Biophys. J. 1:445–466, 1961.
Glass, L., and M. C. Mackey. From Clocks to Chaos. Princeton: Princeton University Press, 1988.
Grasman, J. The mathematical modeling of entrained biological oscillators. Bull. Math. Biol. 46:407–422, 1984.
Gutfreund, Y., Y. Yarom, and I. Segev. Subthreshold oscillations and resonant frequency in guinea-pig cortical neurons: Physiology and modelling. J. Physiol. 483.3:621–640, 1995.
Jefferys, J. G. R., R. D. Traub, and M. A. Whittington. Neuronal networks for induced ‘40 Hz’ rhythms. Trends Neurosci. 19:202–208, 1996.
Kopell, N., and G. B. Ermentrout. Subcellular oscillations and bursting. Math. Biosci. 78:265–291, 1986.
Lee, Y. S., T. R. Chay, and T. Ree. On the mechanism of spiking and bursting in excitable cells. Biophys. Chem. 18:25–34, 1983.
Leung, L. S., and C. Y. Yim. Intrinsic membrane potential oscillations in hippocampal neurons et al. 553:261–274, 1991.
Linkens, D. A., and S. Datardina. Frequency entrainment of coupled Hodgkin–Huxley type oscillators for modeling gastrointestinal electrical activity. IEEE Trans. Biomed. Eng. BME-24:362–365, 1977.
Pavlidis, T. Biological Oscillators: Their Mathematical Analysis. New York: Academic Press, 1973.
Plant, R. E., and M. Kim. Mathematical description of a bursting pacemaker neuron by a modification of the Hodgkin–Huxley equations. Biophys. J. 16:227–244, 1976.
Rinzel, J. A formal classification of bursting mechanisms in excitable cells. In: Mathematical Topics in Population Biology, Morphogenesis, and Neurosciences, Lecture Notes in Biomathematics, vol. 71, edited by E. Teramoto and M. Yamaguti. Berlin: Springer-Verlag, 1987, pp. 267–281.
Sherman, A., and J. Rinzel. Model for synchronization of pancreatic \({\beta}\)-cells by gap junction coupling. Biophys. J. 59:547–559, 1991.
Stein, R. B. Some models of neuronal variability. Biophys. J. 7:37–68, 1967.
Traub, R. D., J. G. R. Jefferys, and M. A. Whittington. Simulation of gamma rhythms in networks of interneurons and pyramidal cells. J. Comput. Neurosci. 4:141–150, 1997.
Traub, R. D., J. G. R. Jefferys, and M. A. Whittington. Fast Oscillations in Cortical Circuits. Cambridge, MA: MIT Press, 1999.
Van der Pol, B., and J. van der Mark. The heart considered as a relaxation oscillator and an electrical model of the heart. Philos. Mag. 6:763–775, 1928.
White, J. A., C. C. Chow, J. Ritt, C. Soto-Trevino, and N. Kopell. Synchronization and oscillatory dynamics in heterogeneous, mutually inhibited neurons. J. Comput. Neurosci. 5:5–16, 1998.
Winfree, A. T. Electrical instability in cardiac muscle: Phase singularities and rotors. J. Theor. Biol. 138:353–405, 1989.
Winfree, A. T. The Geometry of Biological Time, 2nd ed. New York: Springer Verlag, 2001.
Wolf, A., J. B. Swift, H. L. Swinney, and J. A. Vastano. Determining Lyapunov exponents from a time series. Physica D 16:285–317, 1985.
Zariffa, J., M. Ebden, and B. L. Bardakjian. A synaptic input portal for a mapped clock oscillator model of neuronal electrical rhythmic activity. J. Neural. Eng. 1:158–164, 2004.
ACKNOWLEDGMENTS
This work has been supported by the Natural Sciences and Engineering Council of Canada (NSERC) and the “Fonds Québecois de Recherche sur la Nature et les Technologies”.
Author information
Authors and Affiliations
Corresponding author
APPENDIX
APPENDIX
This appendix describes the second-order MCO. The variables described here have the same meaning in the fourth-order cMCOs presented in this paper. Refer to Zariffa et al. 36 for more detailed explanations.
The MCO model was developed with the understanding that excitable cells can undergo rhythmic transmembrane depolarizations. These nonlinear oscillators have an “intrinsic frequency” but are influenced by their environment, including other nearby oscillators. Each oscillator is driven by a “clock,” consisting of two differential equations:
where \({\alpha}\) is the amplitude, \({\phi}\) is the phase and \({\omega}\) is the intrinsic frequency of the clock. Adding the effective stimuli (\({S_{\alpha}}\), \({S_{\phi}}\), \({S_{\gamma1}}\), and \({S_{\gamma2}}\)) applied to the clock's three input portals (called \({P_{\alpha}}\), \({P_{\phi}}\), and \({P_{\gamma}}\)) and including refractoriness (\({R(.)}\)), we obtain:
where \({\dot\phi}\) and \({\alpha}\) are forced to be nonnegative (i.e. negative values are set to zero). This clock is then mapped by a static nonlinearity (e.g. representing transmembrane ionic transport mechanisms) onto an observable output, \(y\), which is also affected by the effective stimulus (\({S_{\rho}}\)) applied to the mapper through its synaptic portal (\({P_{\rho}}\)):
where \(y\) represents the transmembrane voltage, \({a_{0}}\) is the intrinsic average level, \({a_{k}}\), \({b_{k}}\) are the Fourier coefficients of the waveform of the intrinsic oscillation, \({T_{k}}\) and \({U_{k}}\) are the \(k\)th Tchebychev polynomials of the first and second type, respectively, \(k\) is the harmonic index, \({S_{\alpha}}\), \({S_{\phi}}\), \({S_{\gamma1}}\), \({S_{\gamma2}}\), and \({S_{\rho}}\) represent the inputs to the four different portals of the oscillator (if the oscillator is uncoupled, then these are all zero), \(R({\phi})\) is the refractoriness function, which ensures that the neuron stops being sensitive to frequency inputs while it is bursting. It is implemented as a highpass Butterworth function shown in Eq. (A.6).
The Tchebychev polynomials are used in the static nonlinearity of the mapper because in the absence of any stimuli (\({S_{\alpha}} = {S_{\phi}} = {S_{\gamma1}} = {S_{\gamma2}} = {S_{\rho}} = 0\)) the static nonlinearity reduces to a standard Fourier series.2 The parameters of the static nonlinearity are the Fourier coefficients. Hence, the measured transmembrane voltage waveform of an uncoupled biological oscillator can be analyzed in a Fourier series to determine the model parameters of the mapper.
In Eqs. (A.3)–(A.5), four stimulus portals take their effect in different places. Refer to the main text and to Zariffa et al. 36 for a description of their meaning. In general, for the \(n\)th oscillator, having the set \({I_{nm}}\) of neighboring oscillators, the four portals are shown in Eqs. (A.7)–(A.11).
-
Portal \({P_{\phi}}\):
$$S_{\phi} = \frac{\sum_{m \epsilon I_{nm}}^{}(c_{\phi n m} y_{m}) - \beta_{\phi} y_{n} + a_{0} S_{\rho}}{\sigma_{n}}$$(A.7) -
Portal \({P_{\alpha}}\):
$$S_{\alpha} = \frac{\sum_{m \epsilon I_{nm}}^{}(c_{\alpha n m} y_{m}) - \beta_{\alpha} y_{n}}{\sigma_{n}}$$(A.8) -
Portal \({P_{\gamma}}\):
$$S_{\gamma 1} = \frac{\sum_{m \epsilon I_{nm}}^{}(c_{\gamma n m} \alpha_{m} \sin(\phi_{m}))}{\delta}$$(A.9)$$S_{\gamma 2} = \frac{\sum_{m \epsilon I_{nm}}^{}(c_{\gamma n m} \alpha_{m} \cos(\phi_{m}))}{\delta}$$(A.10) -
Portal \({P_{\rho}}\):
$$S_{\rho} = g\left(\frac{\sum_{m \epsilon I_{nm}}^{}c_{\rho n m} \dot\phi_{m} - \beta_{\rho} \dot\phi_{n}}{{\rm sgn}(a_{0})\omega_{n}}\right)$$(A.11)
The function \({g()}\) has the general shape shown in Fig. 17.
The variables have the following meanings:
-
\({\sigma_{n}}\) = \({(\sum_{k=1}^{N}(a_{k}^{2}+b_{k}^{2}))^{1/2}}\) is a normalization factor for the mapper, representing the amplitude of the intrinsic waveform.
-
\({\delta}\) is a static clock normalization factor. Unless otherwise specified it is set to 0.1.
-
\({\omega_{n}}\) is the \(n\)th oscillator's intrinsic frequency.
-
\({\beta_{\phi}}\), \({\beta_{\alpha}}\), and \({\beta_{\rho}}\) are feedback factors.
-
\({c_{\alpha}}\), \({c_{\phi}}\), \({c_{\gamma}}\), and \({c_{\rho}}\) represent the coupling factors between the oscillators. They are real numbers between 0 and 1, where 1 signifies that the corresponding portal is fully open, and 0 signifies that it is closed. The sign of \({c_{\rho}}\) is used to distinguish between excitatory and inhibitory synapses.
-
\({y_{m}}\) is the transmembrane voltage of the driving oscillator.
-
\({\eta}\) is a scaling factor for how much the resting level is allowed to vary.
Rights and permissions
About this article
Cite this article
Zariffa, J., Bardakjian, B.L. Neuronal Electrical Rhythms Described by Composite Mapped Clock Oscillators. Ann Biomed Eng 34, 128–141 (2006). https://doi.org/10.1007/s10439-005-9010-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-005-9010-7