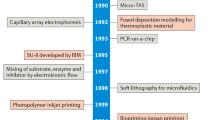
Abstract
Polydopamine (PDA) is a bioinspired material with tremendous potential for applications involving surface modifications. By simply immersing the substrate in the dopamine monomer solution, we are able to apply a hydrophilic and biofunctional PDA coating that adheres strongly to any surface, including (super)hydrophobic surface, with unprecedented ease. Using PDA, almost any materials can be immobilized on the surface in a single step by mixing them with the dopamine monomer solution. This review provides a comprehensive coverage of the applications of PDA in the device fabrication, surface modification, and biofunctionalization of biomedical microfluidic devices. While discussing the advantages and limitations of PDA, we pay special attention to its unique properties that specifically benefit biomedical microfluidic devices. We also discuss other potential applications of PDA beyond the current development. Through this review, we hope to promote PDA and encourage a broader adoption of PDA by the microfluidic community.

Adapted from Ref. (Shi et al. 2014) with the permission from American Chemical Society

Adopted from Ref. (You et al. 2012) with the permission from Wiley

Adopted from Ref. (Wood et al. 2013) with the permission from American Chemical Society

Adopted from Ref. (Feng et al. 2015) with the permission from Royal Society of Chemistry

Adopted from Ref. (Shi et al. 2014) with the permission from American Chemical Society

Adopted from Ref. (Sileika et al. 2011) with the permission from American Chemical Society
Similar content being viewed by others
References
Ball V (2014) Physicochemical perspective on “polydopamine” and “poly(catecholamine)” films for their applications in biomaterial coatings. Biointerphases 9:030801
Beckwith KM, Sikorski P (2013) Patterned cell arrays and patterned co-cultures on polydopamine-modified poly(vinyl alcohol) hydrogels. Biofabrication 5:045009
Bi Y, Zhou H, Jia H, Wei P (2017) Polydopamine-mediated preparation of an enzyme-immobilized microreactor for the rapid production of wax ester. RSC Adv 7:12283–12291
Bunka DHJ, Stockley PG (2006) Aptamers come of age: at last. Nat Rev Microbiol 4:588–596
Chen X, L-l Zhang, J-h Sun, Li H, D-f Cui (2014) A facile and simple high-performance polydimethylsiloxane casting based on self-polymerization dopamine. J Micromech Microeng 24:095006
Cheng G, Zheng SY (2014) Construction of a high-performance magnetic enzyme nanosystem for rapid tryptic digestion. Sci Rep 4:6947
Cheng G, Hao SJ, Yu X, Zheng SY (2015) Nanostructured microfluidic digestion system for rapid high-performance proteolysis. Lab Chip 15:650–654
Chien HW, Tsai WB (2012) Fabrication of tunable micropatterned substrates for cell patterning via microcontact printing of polydopamine with poly(ethylene imine)-grafted copolymers. Acta Biomater 8:3678–3686
Chien HW, Kuo WH, Wang MJ, Tsai SW, Tsai WB (2012) Tunable micropatterned substrates based on poly(dopamine) deposition via microcontact printing. Langmuir 28:5775–5782
Chiou C-H, Shin DJ, Zhang Y, Wang T-H (2013) Topography-assisted electromagnetic platform for blood-to-PCR in a droplet. Biosens Bioelectron 50:91–99
Choi K, Ng AHC, Fobel R, Wheeler AR (2012) Digital microfluidics. Annu Rev Anal Chem 5:413–440
Chuah YJ, Koh YT, Lim K, Menon NV, Wu Y, Kang Y (2015a) Simple surface engineering of polydimethylsiloxane with polydopamine for stabilized mesenchymal stem cell adhesion and multipotency. Sci Rep 5:18162
Chuah YJ, Kuddannaya S, Lee MH, Zhang Y, Kang Y (2015b) The effects of poly(dimethylsiloxane) surface silanization on the mesenchymal stem cell fate. Biomater Sci 3:383–390
Della Vecchia NF, Avolio R, Alfè M, Errico ME, Napolitano A, d’Ischia M (2013) Building-block diversity in polydopamine underpins a multifunctional eumelanin-type platform tunable through a quinone control point. Adv Funct Mater 23:1331–1340
Dertinger SKW, Chiu DT, Jeon NL, Whitesides GM (2001) Generation of gradients having complex shapes using microfluidic networks. Anal Chem 73:1240–1246
d’Ischia M, Napolitano A, Pezzella A, Meredith P, Sarna T (2009) Chemical and structural diversity in eumelanins: unexplored bio-optoelectronic materials. Angew Chem 48:3914–3921
d’Ischia M, Napolitano A, Ball V, Chen CT, Buehler MJ (2014) Polydopamine and eumelanin: from structure-property relationships to a unified tailoring strategy. Acc Chem Res 47:3541–3550
Dong Z, Zhang F, Wang D, Liu X, Jin J (2015) Polydopamine-mediated surface-functionalization of graphene oxide for heavy metal ions removal. J Solid State Chem 224:88–93
Dreyer DR, Miller DJ, Freeman BD, Paul DR, Bielawski CW (2013) Perspectives on poly(dopamine). Chem Sci 4:3796–3802
Duffy DC, McDonald JC, Schueller OJ, Whitesides GM (1998) Rapid prototyping of microfluidic systems in poly(dimethylsiloxane). Anal Chem 70:4974–4984
Eteshola E, Leckband D (2001) Development and characterization of an ELISA assay in PDMS microfluidic channels. Sens Actuators B Chem 72:129–133
Fan D, Wu C, Wang K, Gu X, Liu Y, Wang E (2016) A polydopamine nanosphere based highly sensitive and selective aptamer cytosensor with enzyme amplification. Chem Commun 52:406–409
Feng H, Zhang Q, Ma H, Zheng B (2015) An ultralow background substrate for protein microarray technology. Analyst 140:5627–5633
Fuard D, Tzvetkova-Chevolleau T, Decossas S, Tracqui P, Schiavone P (2008) Optimization of poly-di-methyl-siloxane (PDMS) substrates for studying cellular adhesion and motility. Microelectron Eng 85:1289–1293
Gao H, Sun Y, Zhou J, Xu R, Duan H (2013) Mussel-inspired synthesis of polydopamine-functionalized graphene hydrogel as reusable adsorbents for water purification. ACS Appl Mater Interfaces 5:425–432
Gomez FA (2008) Biological applications of microfluidics. Wiley, Hoboken
Hillborg H, Ankner JF, Gedde UW, Smith GD, Yasuda HK, Wikström K (2000) Crosslinked polydimethylsiloxane exposed to oxygen plasma studied by neutron reflectometry and other surface specific techniques. Polymer 41:6851–6863
Ho CC, Ding SJ (2013) The pH-controlled nanoparticles size of polydopamine for anti-cancer drug delivery. J Mater Sci 24:2381–2390
Ho C-C, Ding S-J (2014) Structure, properties and applications of mussel-inspired polydopamine. J Biomed Nanotechnol 10:3063–3084
Hong S, Kim KY, Wook HJ, Park SY, Lee KD, Lee DY, Lee H (2011) Attenuation of the in vivo toxicity of biomaterials by polydopamine surface modification. Nanomedicine 6:793–801
Hong D, Bae K, Hong SP, Park JH, Choi IS, Cho WK (2014) Mussel-inspired, perfluorinated polydopamine for self-cleaning coating on various substrates. Chem Commun 50:11649–11652
Jebrail MJ, Bartsch MS, Patel KD (2012) Digital microfluidics: a versatile tool for applications in chemistry, biology and medicine. Lab Chip 12:2452–2463
Jiang J, Zhu L, Zhu L, Zhu B, Xu Y (2011) Surface characteristics of a self-polymerized dopamine coating deposited on hydrophobic polymer films. Langmuir 27:14180–14187
Jolly P et al (2016) DNA aptamer-based sandwich microfluidic assays for dual quantification and multi-glycan profiling of cancer biomarkers. Biosens Bioelectron 79:313–319
Jun D-R, Choi YR, Kang RH, Choi S-W (2016) Polydimethylsiloxane fluidic device with polydopamine-coated inner channel for production of uniform droplets. Macromol Mater Eng 301:1044–1048
Kang SM et al (2010) One-step modification of superhydrophobic surfaces by a mussel-inspired polymer coating. Angew Chem 49:9401–9404
Kang SM et al (2012) One-step multipurpose surface functionalization by adhesive catecholamine. Adv Funct Mater 22:2949–2955
Kim BH et al (2011) Mussel-inspired block copolymer lithography for low surface energy materials of teflon, graphene, and gold. Adv Mater 23:5618–5622
Kim HW, McCloskey BD, Choi TH, Lee C, Kim MJ, Freeman BD, Park HB (2013a) Oxygen concentration control of dopamine-induced high uniformity surface coating chemistry. ACS Appl Mater Interfaces 5:233–238
Kim M, Song KH, Doh J (2013b) PDMS bonding to a bio-friendly photoresist via self-polymerized poly(dopamine) adhesive for complex protein micropatterning inside microfluidic channels. Colloids Surf B 112:134–138
Ku SH, Park CB (2010) Human endothelial cell growth on mussel-inspired nanofiber scaffold for vascular tissue engineering. Biomaterials 31:9431–9437
Ku SH, Lee JS, Park CB (2010a) Spatial control of cell adhesion and patterning through mussel-inspired surface modification by polydopamine. Langmuir 26:15104–15108
Ku SH, Ryu J, Hong SK, Lee H, Park CB (2010b) General functionalization route for cell adhesion on non-wetting surfaces. Biomaterials 31:2535–2541
Lara HH, Garza-Trevino EN, Ixtepan-Turrent L, Singh DK (2011) Silver nanoparticles are broad-spectrum bactericidal and virucidal compounds. J Nanobiotechnol 9:30
Lee H, Dellatore SM, Miller WM, Messersmith PB (2007) Mussel-inspired surface chemistry for multifunctional coatings. Science 318:426–430
Lee H, Rho J, Messersmith PB (2009) Facile conjugation of biomolecules onto surfaces via mussel adhesive protein inspired coatings. Adv Mater 21:431–434
Lee M, Ku SH, Ryu J, Park CB (2010) Mussel-inspired functionalization of carbon nanotubes for hydroxyapatite mineralization. J Mater Chem 20:8848
Lee BP, Messersmith PB, Israelachvili JN, Waite JH (2011) Mussel-inspired adhesives and coatings. Annu Rev Mater Res 41:99–132
Leung JM et al (2015) Surface modification of poly(dimethylsiloxane) with a covalent antithrombin–heparin complex for the prevention of thrombosis: use of polydopamine as bonding agent. J Mater Chem 3:6032–6036
Liang RP, Meng XY, Liu CM, Qiu JD (2011) PDMS microchip coated with polydopamine/gold nanoparticles hybrid for efficient electrophoresis separation of amino acids. Electrophoresis 32:3331–3340
Liang Y et al (2017) Metal-polydopamine frameworks and their transformation to hollow metal/N-doped carbon particles. Nanoscale 9:5323–5328
Liebscher J et al (2013) Structure of polydopamine: a never-ending story? Langmuir 29:10539–10548
Ling Y, Li W, Wang B, Gan W, Zhu C, Brady MA, Wang C (2016) Epoxy resin reinforced with nanothin polydopamine-coated carbon nanotubes: a study of the interfacial polymer layer thickness. RSC Adv 6:31037–31045
Liu Q, Yu B, Ye W, Zhou F (2011) Highly selective uptake and release of charged molecules by pH-responsive polydopamine microcapsules. Macromol Biosci 11:1227–1234
Liu C-M, Liang R-P, Wang X-N, Wang J-W, Qiu J-D (2013a) A versatile polydopamine platform for facile preparation of protein stationary phase for chip-based open tubular capillary electrochromatography enantioseparation. J Chromatogr Sci 1294:145–151
Liu R, Guo Y, Odusote G, Qu F, Priestley RD (2013b) Core-shell Fe3O4 polydopamine nanoparticles serve multipurpose as drug carrier, catalyst support and carbon adsorbent. ACS Appl Mater Interfaces 5:9167–9171
Liu Y, Ai K, Liu J, Deng M, He Y, Lu L (2013c) Dopamine-melanin colloidal nanospheres: an efficient near-infrared photothermal therapeutic agent for in vivo cancer therapy. Adv Mater 25:1353–1359
Liu Y, Ai K, Lu L (2014) Polydopamine and its derivative materials: synthesis and promising applications in energy, environmental, and biomedical fields. Chem Rev 114:5057–5115
Loget G, Wood JB, Cho K, Halpern AR, Corn RM (2013) Electrodeposition of polydopamine thin films for DNA patterning and microarrays. Anal Chem 85:9991–9995
Lu YW, Lin PT, Pai CS (2007) Polydimethylsiloxane (PDMS) bonding strength characterization by a line force model in blister tests. Paper presented at the transducers 2007–2007 international solid-state sensors, actuators and microsystems conference, 10–14 June 2007
Lynge ME, van der Westen R, Postma A, Stadler B (2011) Polydopamine-a nature-inspired polymer coating for biomedical science. Nanoscale 3:4916–4928
Lynge ME, Schattling P, Stadler B (2015) Recent developments in poly(dopamine)-based coatings for biomedical applications. Nanomedicine 10:2725–2742
Madhurakkat Perikamana SK, Lee J, Lee YB, Shin YM, Lee EJ, Mikos AG, Shin H (2015) Materials from mussel-inspired chemistry for cell and tissue engineering applications. Biomacromol 16:2541–2555
Mata A, Fleischman AJ, Roy S (2005) Characterization of polydimethylsiloxane (PDMS) properties for biomedical micro/nanosystems. Biomed Microdevices 7:281–293
McCloskey BD et al (2010) Influence of polydopamine deposition conditions on pure water flux and foulant adhesion resistance of reverse osmosis, ultrafiltration, and microfiltration membranes. Polymer 51:3472–3485
Miller EM, Wheeler AR (2008) A digital microfluidic approach to homogeneous enzyme assays. Anal Chem 80:1614–1619
Ng AHC, Choi K, Luoma RP, Robinson JM, Wheeler AR (2012) Digital microfluidic magnetic separation for particle-based immunoassays. Anal Chem 84:8805–8812
Ng AHC, Chamberlain MD, Situ H, Lee V, Wheeler AR (2015) Digital microfluidic immunocytochemistry in single cells. Nat Commun 6:7513
Pipper J, Inoue M, Ng LFP, Neuzil P, Zhang Y, Novak L (2007) Catching bird flu in a droplet. Nat Med 13:1259–1263
Pipper J, Zhang Y, Neuzil P, Hsieh TM (2008) Clockwork PCR including sample preparation. Angew Chem 47:3900–3904
Postma A, Yan Y, Wang Y, Zelikin AN, Tjipto E, Caruso F (2009) Self-polymerization of dopamine as a versatile and robust technique to prepare polymer capsules. Chem Mater 21:3042–3044
Qiang W, Hu H, Sun L, Li H, Xu D (2015) Aptamer/polydopamine nanospheres nanocomplex for in situ molecular sensing in living cells. Anal Chem 87:12190–12196
Ren K, Dai W, Zhou J, Su J, Wu H (2011) Whole-teflon microfluidic chips. Proc Natl Acad Sci 108:8162–8166
Resnick PR (1989) The preparation and properties of a new family of amorphous fluoropolymers: teflon® AF. J Fluor Chem 45:100
Rivera JG, Messersmith PB (2012) Polydopamine-assisted immobilization of trypsin onto monolithic structures for protein digestion. J Sep Sci 35:1514–1520
Sackmann EK, Fulton AL, Beebe DJ (2014) The present and future role of microfluidics in biomedical research. Nature 507:181–189
Salazar P, Martín M, Gonzalez-Mora JL (2016) Polydopamine-modified surfaces in biosensor applications. In: Méndez-Vilas A, Solano A (eds) Polymer science: research advances, practical applications and educational aspects. Formatex Research Center, pp 385–396
Shah GJ, Ohta AT, Chiou EPY, Wu MC (2009) EWOD-driven droplet microfluidic device integrated with optoelectronic tweezers as an automated platform for cellular isolation and analysis. Lab Chip 9:1732–1739
Shamsi MH, Choi K, Ng AHC, Wheeler AR (2014) A digital microfluidic electrochemical immunoassay. Lab Chip 14:547–554
Shen B, Xiong B, Wu H (2015) Convenient surface functionalization of whole-teflon chips with polydopamine coating. Biomicrofluidics 9:044111
Shi X, Ostrovidov S, Shu Y, Liang X, Nakajima K, Wu H, Khademhosseini A (2014) Microfluidic generation of polydopamine gradients on hydrophobic surfaces. Langmuir 30:832–838
Shin SR et al (2016) Aptamer-based microfluidic electrochemical biosensor for monitoring cell-secreted trace cardiac biomarkers. Anal Chem 88:10019–10027
Sileika TS, Kim HD, Maniak P, Messersmith PB (2011) Antibacterial performance of polydopamine-modified polymer surfaces containing passive and active components. ACS Appl Mater Interfaces 3:4602–4610
Sun K et al (2012) Mussel-inspired anchoring for patterning cells using polydopamine. Langmuir 28:2131–2136
Tsai W-B, Chen W-T, Chien H-W, Kuo W-H, Wang M-J (2011) Poly(dopamine) coating of scaffolds for articular cartilage tissue engineering. Acta Biomater 7:4187–4194
Tuerk C, Gold L (1990) Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 249:505–510
Wang Y et al (2015a) An ultra hydrophilic dendrimer-modified magnetic graphene with a polydopamine coating for the selective enrichment of glycopeptides. J Mater Chem B 3:8711–8716
Wang Z et al (2015b) Bioadhesive microporous architectures by self-assembling polydopamine microcapsules for biomedical applications. Chem Mater 27:848–856
Wei X, Yang X, Han ZP, Qu FF, Shao L, Shi YF (2013) Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin 34:747–754
Weinhold M, Soubatch S, Temirov R, Rohlfing M, Jastorff B, Tautz FS, Doose C (2006) Structure and bonding of the multifunctional amino acid l-DOPA on Au(110). J Phys Chem Ref Data 110:23756–23769
Whitesides GM (2006) The origins and the future of microfluidics. Nature 442:368–373
Wöllenstein J, Plaza JA, Cané C, Min Y, Böttner H, Tuller HL (2003) A novel single chip thin film metal oxide array. Sens Actuators B Chem 93:350–355
Wong T-S, Kang SH, Tang SKY, Smythe EJ, Hatton BD, Grinthal A, Aizenberg J (2011) Bioinspired self-repairing slippery surfaces with pressure-stable omniphobicity. Nature 477:443–447
Wood JB, Szyndler MW, Halpern AR, Cho K, Corn RM (2013) Fabrication of DNA microarrays on polydopamine-modified gold thin films for SPR imaging measurements. Langmuir 29:10868–10873
Yan Y, Zheng Z, Deng C, Li Y, Zhang X, Yang P (2013) Hydrophilic polydopamine-coated graphene for metal ion immobilization as a novel immobilized metal ion affinity chromatography platform for phosphoproteome analysis. Anal Chem 85:8483–8487
Yang HC, Wu QY, Wan LS, Xu ZK (2013) Polydopamine gradients by oxygen diffusion controlled autoxidation. Chem Commun 49:10522–10524
Ye Q, Zhou F, Liu W (2011) Bioinspired catecholic chemistry for surface modification. Chem Soc Rev 40:4244–4258
You I et al (2012) Polydopamine microfluidic system toward a two-dimensional, gravity-driven mixing device. Angew Chem 51:6126–6130
You I, Lee TG, Nam YS, Lee H (2014) Fabrication of a micro-omnifluidic device by omniphilic/omniphobic patterning on nanostructured surfaces. ACS Nano 8:9016–9024
Yu B, Liu J, Liu S, Zhou F (2010) Pdop layer exhibiting zwitterionicity: a simple electrochemical interface for governing ion permeability. Chem Commun 46:5900–5902
Zhang Y, Nguyen N-T (2017) Magnetic digital microfluidics: a review. Lab Chip 17:994–1008
Zhang Y, Wang TH (2013) Full-range magnetic manipulation of droplets via surface energy traps enables complex bioassays. Adv Mater 25:2903–2908
Zhang Y, Park S, Liu K, Tsuan J, Yang S, Wang T-H (2011) A surface topography assisted droplet manipulation platform for biomarker detection and pathogen identification. Lab Chip 11:398–406
Zhang W, Yang FK, Han Y, Gaikwad R, Leonenko Z, Zhao B (2013a) Surface and tribological behaviors of the bioinspired polydopamine thin films under dry and wet conditions. Biomacromol 14:394–405
Zhang Y, Shin DJ, Wang T-H (2013b) Serial dilution via surface energy trap-assisted magnetic droplet manipulation. Lab Chip 13:4827–4831
Zhang Y, Geng X, Ai J, Gao Q, Qi H, Zhang C (2015) Signal amplification detection of DNA using a sensor fabricated by one-step covalent immobilization of amino-terminated probe DNA onto the polydopamine-modified screen-printed carbon electrode. Sens Actuators B Chem 221:1535–1541
Zhang P, He M, Zeng Y (2016) Ultrasensitive microfluidic analysis of circulating exosomes using a nanostructured graphene oxide/polydopamine coating. Lab Chip 16:3033–3042
Zhao Y, Liu X, Li J, Qiang W, Sun L, Li H, Xu D (2016) Microfluidic chip-based silver nanoparticles aptasensor for colorimetric detection of thrombin. Talanta 150:81–87
Zhou P, Deng Y, Lyu B, Zhang R, Zhang H, Ma H, Lyu Y, Wei S (2014) Rapidly-deposited polydopamine coating via high temperature and vigorous stirring: formation, characterization and biofunctional evaluation. PLoS ONE 9(11):e113087
Zou Z, Jang A, MacKnight E, Wu P-M, Do J, Bishop PL, Ahn CH (2008) Environmentally friendly disposable sensors with microfabricated on-chip planar bismuth electrode for in situ heavy metal ions measurement. Sens Actuators B Chem 134:18–24
Acknowledgements
The authors would like to thank the start-up grant from the School of Mechanical and Aerospace Engineering at Nanyang Technological University. This work is also supported by the Ageing Research Institute for Society and Education (ARISE), Nanyang Technological University, Singapore (Grant Reference Number ARISE/2017/22), and Singapore Ministry of Education AcRF Tier 1 Grant RG49/17.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kanitthamniyom, P., Zhang, Y. Application of polydopamine in biomedical microfluidic devices. Microfluid Nanofluid 22, 24 (2018). https://doi.org/10.1007/s10404-018-2044-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-018-2044-6