Abstract
Background
The aim of this study was to evaluate the differences in upper gastrointestinal symptoms between generations and genders in relatively healthy Japanese subjects.
Methods
Altogether, 4086 healthy Japanese male and female (M/F) adults (M/F: 2244/1842) were analyzed. Among them, 3505 subjects (M/F: 1922/1583) were underwent a routine medical checkup at one of five hospitals in Saga, Japan from January 2013 to December 2013. The others were 581 (M/F: 322/259) healthy young volunteers at the Saga Medical School from April 2007 to March 2013. The participants were asked to complete the frequency scale for the symptoms of gastroesophageal reflex disease (FSSG) questionnaire, undergo upper gastrointestinal endoscopy, and submit to a rapid urease test to diagnose Helicobacter pylori infection. Among the 4086 subjects, the 2414 who had no H. pylori infection and no positive endoscopic findings were enrolled in the study.
Results
Subjects’ average age was 46.9 ± 12.2 years, with males’ and females’ ages being almost equivalent. The total FSSG score were high in females compared to males (P < 0.01) and decreased significantly with aging (P < 0.05). Among the generations, FSSG scores were the highest for those 20–29 years old, and they were significantly decreased with ageing in both males and females (P < 0.05).
Conclusion
The FSSG score was significantly higher in healthy Japanese females than in males, and the scores decreased with aging.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Annual public health surveys conducted by the Ministry of Health, Labor, and Welfare of Japan in 2013 indicated that 27.7% of men and 31.2% of females had some type of clinical symptoms [1]. The report suggested that, among those > 20 years of age, Japanese females tended to complain more frequently than males [1]. Whereas the prevalence of abdominal symptoms in a USA population-based study showed no significant difference between genders [2] and a previous study in Japan suggested that the prevalence of non-ulcer-related dyspepsia was not different between the genders [3], most studies did not clearly demonstrate gender differences regarding upper gastrointestinal symptoms [3,4,5]. Our previous studies suggested that upper gastrointestinal symptoms in relatively healthy Japanese subjects were more frequent in females than in males [6, 7].
A Japanese study of the clinical signs and symptoms of gastroesophageal reflux disease (GERD) developed a frequency scale for the symptoms of GERD (FSSG). It is a questionnaire for use with Japanese subjects and covers most upper gastrointestinal symptoms [8]. The questions are related to the 12 symptoms about which the Japanese subjects complained: e.g., “heartburn” and “acidic taste” with GERD; “heavy stomach” and “feeling full quickly” in dyspeptic patients [8,9,10,11].
The present study aimed to examine; (1) whether the upper gastrointestinal symptoms evaluated by FSSG were different between relatively healthy male and female Japanese subjects, and (2) whether the tendency evaluated in these two groups was affected by aging.
Methods
In all, 4086 male and female (M/F) healthy Japanese adults (M/F: 2244/1842) were analyzed. Among them, 3505 (M/F: 1922/1583) had undergone upper gastrointestinal endoscopy for health screening during routine medical checkups at five hospitals in Saga, Japan from January 2013 to December 2013 and were enrolled from our previous study [7]. The other 581 subjects (M/F: 322/259) were healthy volunteers (fifth-year medical students at Saga Medical School) who had undergone upper gastrointestinal endoscopy and were enrolled from a previous study conducted from April 2007 to March 2013 [6]. Those with a surgical history involving the upper gastrointestinal tract were excluded from the study. The subjects who were prescribed with the medicine which influenced the upper gastrointestinal symptoms including the gastric acid suppression medicine and/or who had the gastrointestinal disease were excluded from the analysis. We obtained informed consent from all participants. All the procedures performed in the present study were approved by the Ethical Committee of the Saga University Hospital.
All subjects (ages 24–83 years) completed an FSSG questionnaire [8] before endoscopy. Helicobacter pylori (H. pylori) infection was then diagnosed by the rapid urease test [12], the serum H. pylori immunoglobulin G antibody titer [13], and/or the urinary antibody-coated bacteria test via immunochromatography (RAPIRUN®; Otsuka Pharmaceutical Co., Ltd., Tokyo, Japan) [14]. The history of H. pylori eradication was confirmed by the patient’s medical records and history.
All subjects underwent upper gastrointestinal endoscopy, with findings of reflux esophagitis diagnosed as grade A, B, C, or D using the Los Angeles classification [15]. FSSG comprises 12 questions (7 on reflux symptoms, 5 on acid-related dyspepsia) [8]. Each symptom was assigned a score [never = 0; occasionally (30%) = 1; sometimes (50%) = 2; often (70%) = 3; always (100%) = 4]. The 12 questions were “Do you get heartburn?” “Does your stomach get bloated?” “Does your stomach ever feel heavy after meals?” “Do you sometimes subconsciously rub your chest with your hand?” “Do you ever feel sick after meals?” “Do you get heartburn after meals?” “Do you have an unusual (e.g., burning) sensation in your throat?” “Do you feel full while eating meals?” “Do some things get stuck when you swallow?” “Do you feel a bitter liquid (acid) coming up into your throat?” “Do you burp a lot?” and “Do you get heartburn if you bend over?”
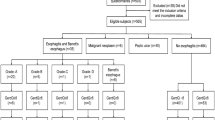
A flowchart outlining the selection process for the analyzed subjects is shown in Fig. 1. Among the 4086 healthy adults who correctly completed the questionnaires, 291 were excluded from analysis because of the inquiry loss and 1032 because of the presence of an H. pylori infection. Subjects who had undergone successful eradication of H. pylori (n = 505: M/F: 399/107)—504 from the medical checkup group, 1 from the medical students group—were included in the study. Among 2763 subjects, 345 with endoscopic reflux esophagitis (M/F: grade A, 236/60; grade B, 36/11; grade C, 2/0; grade D, 0/0) were excluded. Four patients > 80 years old were also excluded because of the small number of the subjects in that age group. Finally, 2414 subjects with no H. pylori infection and no abnormal upper gastrointestinal endoscopy findings were enrolled, as indicated in Fig. 1. The enrolled healthy subjects were then divided into six age groups (those in their 20, 30, 40, 50, 60 and 70 s).
Statistical analysis
Descriptive statistics for continuous and categorical variables were reported as a mean with standard deviation, and as frequency and percentage, respectively. The Mann–Whitney U test and Kruskal–Wallis test were used to test the difference of the FSSG frequency score by gender and age groups. Pearson correlation coefficients were calculated to measure the associations for the FSSG frequency score with age group by gender. Furthermore, a multiple linear regression model was used to estimate the difference in the FSSG frequency score between age group adjusting for gender. All statistical analyses were performed with SSPS version 22 software (IBM Japan Ltd., Tokyo, Japan). P < 0.05 was considered to indicate a statistically significant difference.
Results
Table 1 shows the characteristics of the healthy subjects (with no H. pylori infection and no endoscopic findings of reflux esophagitis). The average age was 46.9 ± 12.2 years. The ages of males and females were almost equivalent. Table 2 shows FSSG scores of the subjects in each age and gender group. Associations between age group and FSSG frequency score were significantly independent (P < 0.05). The total FSSG scores were significantly high in females compared to males (P < 0.01). The FSSG scores of those in their 20 s (20–29 years) were higher than those for any of the other generations. In addition, the FSSG score was higher in the young generations, with the score decreasing significantly along with aging. This decrease was statistically significant in the overall group of subjects (males + females) (β: − 1.58, P < 0.05), and was reflected in the significant decrease in the FSSG score with aging in females and males (P < 0.05). Both generation and gender were significant predictors of the FSSG score.
Tables 3 and 4 show the influence of the age and gender on the reflux symptoms and acid-related dyspepsia scores, respectively. Associations between age group and FSSG frequency score were significantly independent (P < 0.05). As indicated in Table 3, the decrease in the scores with aging was statistically significant in the acid-related dyspepsia score (β: − 2.11, P < 0.05) and the reflux symptoms score (β:− 0.77, P < 0.05). The age-related decrease was significant when the analysis was performed separately by gender (i.e., in males and females separately) except the dyspepsia in males (Table 4).
Discussion
Our previous studies indicated that, among relatively healthy subjects, females complained of upper gastrointestinal symptoms more commonly than males in the younger generations [6, 16] and middle-aged to older generations [7, 17]. The present study indicated that: (1) among relatively healthy subjects, FSSG-identified upper gastrointestinal symptoms were more common in females than males; (2) complaints of upper gastrointestinal symptoms diminished concomitantly with aging; and (3) age-related changes in symptoms were typical both in those with acid-related dyspepsia and reflux symptoms.
A previous study of Dutch and Japanese working populations indicated that women complained of upper gastrointestinal symptoms more often than men [18]. Other studies showed that functional dyspepsia patients in young generations of Korean and Japanese populations complained of more serious upper gastrointestinal symptoms than did aged populations [19,20,21,22]. An Internet survey in Japan of 15,000 subjects indicated that it was young females who most complained of acid-related dyspepsia [23]. The results of these reports—similar to the results of the present study—indicated that upper gastrointestinal symptoms were most significant in the young females. These results suggest that young females might overstate the upper gastrointestinal symptoms even in the clinical situation.
Several studies indicated that functional dyspepsia was more common in older and/or aged women in both Japan [24] and Western countries [25], whereas in Taiwan functional dyspepsia was more common in the younger generations [26]. The upper gastrointestinal symptoms were sometimes accompanied by other clinical symptoms, including headache [27] and gastric dysfunction [28,29,30,31,32] in an age-related manner. Judging from these data and the present results, complaints of the upper gastrointestinal symptoms occurred more often in young females, although they might not portend as serious a situation as when they occur in older and/or aged generations.
The present study indicated that relatively healthy young females complained of upper gastrointestinal symptoms more often than males overall and females of other generations, as shown by the FSSG questionnaire.
References
Itoh M, Ibe T, Umeda K, et al. Health status and receiving medical treatment. J Health Welf Stat. 2014;61:87–94 (Japanese).
Talley NJ, Zinsmeister AR, Schleck CD, Melton LJ 3rd. Dyspepsia and dyspepsia subgroups: a population-based study. Gastroenterology. 1992;102:1259–68.
Oshima T, Miwa H. Epidemiology of functional gastrointestinal disorders in Japan and in the world. J Neurogastroenterol Motil. 2015;21:320–9.
Kiyota K. Epidemiology of non-ulcer dyspepsia (NUD) in Japan. Nihon Shokakibyo Gakkai Zasshi. 1992;89:1973–81 (Japanese).
Stanghellini V, Chan FKL, Hasler WL, et al. Gastroduodenal disorders. Gastroenterology. 2016;150:1380–92.
Kawakubo H, Tanaka Y, Tsuruoka N, et al. Upper gastrointestinal symptoms are more frequent in female than male young healthy Japanese volunteers as evaluated by questionnaire. J Neurogastroenterol Motil. 2016;22:248–53.
Takeshita E, Sakata Y, Hara M, et al. Higher frequency of reflux symptoms and acid-related dyspepsia in women than men regardless of endoscopic esophagitis: analysis of 3505 Japanese subjects undergoing medical health checkups. Digestion. 2016;93:266–71.
Kusano M, Shimoyama Y, Sugimoto S, et al. Development and evaluation of FSSG: frequency scale for symptoms of GERD. J Gastroenterol. 2004;39:888–91.
Danjo A, Yamaguchi K, Fujimoto K, et al. Comparison of endoscopic findings with symptom assessment systems (FSSG and QUEST) for gastroesophageal reflux disease in Japanese centres. J Gastroenterol Hepatol. 2009;24:633–8.
Komatsu-Tanaka M, Iwakiri R, Fujimoto K, et al. Clinical symptoms of FSSG in gastroesophageal reflux disease are critical for PPI treatment: Japanese multi-centers with 185 patients. Dig Endosc. 2012;24:407–11.
Akutagawa K, Iwakiri R, Hara M, et al. Risk factors for low response to proton-pump inhibitor treatment in reflux esophagitis and non-erosive reflux disease evaluated by the frequency scale for the symptoms of gastroesophageal reflux disease. Esophagus. 2015;12:225–32.
Vaira D, Perna F. How useful is the rapid urease test for evaluating the success of Helicobacter pylori eradication therapy? Nat Clin Pract Gastroenterol Hepatol. 2007;4:600–1.
Kawai T, Kawakami K, Kudo T, et al. A new antibody test kit (E plate) for evaluation of Helicobacter pylori eradication. Intern Med. 2002;41:780–3.
Murakami K, Kamada T, Ishikawa H, et al. An evaluation of the performance of a novel stick-type kit for rapid detection of Helicobacter pylori antibodies in urine. Clin Lab. 2011;57:481–7.
Lundell LR, Dent J, Bennett JR, et al. Endoscopic assessment of esophagitis: clinical and functional correlates and further validation of the Los Angeles classification. Gut. 1999;45:172–80.
Tanaka Y, Sakata Y, Hara M, et al. Risk Factors for Helicobacter pylori infection and endoscopic reflux esophagitis in healthy young Japanese volunteers. Intern Med. 2017;56:2979–83.
Yoshioka T, Takeshita E, Sakata Y, et al. Helicobacter pylori infection status had no influence on upper gastrointestinal symptoms: a cross-sectional analysis of 3005 Japanese subjects without upper gastrointestinal lesions undergoing medical health checkups. Esophagus. 2017;14:249–53.
Schlemper RJ, van der Werf SD, Vandenbroucke JP, et al. Peptic ulcer, non-ulcer dyspepsia and irritable bowel syndrome in The Netherlands and Japan. Scand J Gastroenterol. 1993;200(suppl):33–41.
Lee SY, Masaoka T, Han HS, et al. A prospective study on symptom generation according to spicy food intake and TRPV1 genotypes in functional dyspepsia patients. Neurogastroenterol Motil. 2016;28:1401–8.
Lee SP, Lee S-Y, Kim JH, et al. Factors related to upper gastrointestinal symptom generation in 2275 Helicobacter pylori seroprevalent adults. Dig Dis Sci. 2017;62:1561–70.
Haruma K, Kinoshita Y, Sakamoto S, et al. Lifestyle factors and efficacy of lifestyle interventions in gastroesophageal reflux disease patients with functional dyspepsia: primary care perspectives from the LEGEND study. Intern Med. 2015;54:695–701.
Miwa H, Kusano M, Arisawa T, et al. Evidence-based clinical practice guidelines for functional dyspepsia. J Gastroenterol. 2015;50:125–39.
Miwa H. Life style in persons with functional gastrointestinal disorders-large-scale internet survey of lifestyle in Japan. Neurogastroenterol Motil. 2012;24:464–71.
Miwa H. Why dyspepsia can occur without organic disease: pathogenesis and management of functional dyspepsia. J Gastroenterol. 2012;47:862–71.
Baron JH, Sonnenberg A. Hospital admissions and primary care attendances for nonulcer dyspepsia, reflux esophagitis and peptic ulcer in Scotland 1981–2004. Eur J Gastroenterol Hepatol. 2008;20:180–6.
Chang FY, Chen PH, Wu TC, et al. Prevalence of functional gastrointestinal disorders in Taiwan: questionnaire-based survey for adults based on the Rome III criteria. Asia Pac J Clin Nutr. 2012;21:594–600.
Kunishi Y, Iwata Y, Ota M, et al. Abdominal migraine in a middle-aged woman. Intern Med. 2016;55:2793–8.
Asano H, Tomita T, Nakamura K, et al. Prevalence of gastric motility disorders in patients with functional dyspepsia. J Neurogastroenterol Motil. 2017 (In press).
Mori H, Suzuki H, Matsuzaki J, et al. Gender difference of gastric emptying in healthy volunteers and patients with functional dyspepsia. Digestion. 2017;95:72–8.
Göktaş Z, Köklü S, Dikmen D, et al. Nutritional habits in functional dyspepsia and its subgroups: a comparative study. Scand J Gastroenterol. 2016;51:903–7.
Yamaguchi D, Sakata Y, Tsuruoka N, et al. Characteristics of patients with non-variceal upper gastrointestinal bleeding taking antithrombotic agents. Dig Endosc. 2015;27:30–6.
Fujiwara Y, Arakawa T. Overlap in patients with dyspepsia/functional dyspepsia. J Neurogastroenterol Motil. 2014;20:447–57.
Acknowledgements
We thank Nancy Schatken, BS, MT (ASCP), from Edanz Group (www.edanzediting.com/ac) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human rights statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Informed consent, or substitute for it, was obtained from all patients included in the study.
Conflict of interest
The authors declare that there were no conflicts of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kawachi, K., Sakata, Y., Hara, M. et al. Higher frequency of upper gastrointestinal symptoms in healthy young Japanese females compared to males and older generations. Esophagus 15, 83–87 (2018). https://doi.org/10.1007/s10388-017-0598-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10388-017-0598-0