Abstract
Purpose
We evaluated ocular symptoms and activity of retinal vasculitis of Behçet disease before and after infliximab therapy, using Behçet disease ocular attack score 24 (BOS24) and fluorescein angiography (FA) score. We also analyzed the efficacy of infliximab for ocular and extraocular symptoms.
Study design
Retrospective study.
Subject and methods
Using medical records, we analyzed FA and BOS24 to evaluate the association between the efficacy of infliximab therapy and FA as well as BOS24 scores. Further, we evaluated the association between FA scores and extraocular symptoms.
Results
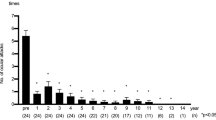
After 2 years of infliximab therapy, 6-month BOS24 was significantly reduced compared to that before treatment (PreBOS24-6M). After 4 years of infliximab therapy, 6-month BOS24 was also significantly reduced compared to preBOS24-6M. After 2 years of infliximab therapy, FA score median (interquartile range) decreased significantly compared to that before treatment [FA-2Y vs. preFA: 0 (0-0) vs.15.5 (12-24); P < 0.0001; n = 38]. The FA-4Y score in subjects followed for at least 4 years was also significantly lower than the preFA score [0 (0-0) vs.16.5 (12-24.5); P < 0.0001; n = 28]. Among 38 patients, extraocular symptoms were resolved following treatment in 29 cases (76.3%). No significant correlation was observed between the improvement in FA-2Y and FA-4Y scores and the occurrence and persistence of extraocular symptoms (P = 0.33; n = 38 or P = 0.4; n = 28).
Conclusion
Infliximab therapy is effective for the treatment of ocular and extraocular involvement of Behçet disease. BOS24 and FA scores are useful for evaluating the efficacy of infliximab therapy.

Similar content being viewed by others
Abbreviations
- DAS 28:
-
Disease activity score in rheumatoid arthritis
- BOS24:
-
Behçet disease ocular attack score 24
- FA:
-
Fluorescein angiography
References
Okada AA, Goto H, Ohno S, Mochizuki M. Multicenter study of infliximab for refractory uveoretinitis in Behçet disease. Arch Ophthalmol. 2012;130:592–8.
Takeuchi M, Kezuka T, Sugita S, Keino H, Namba K, Kaburaki T, et al. Evaluation of the long-term efficacy and safety of infliximab treatment for uveitis in Behçet’s disease: a multicenter study. Ophthalmology. 2014;121:1877–84.
Vallet H, Riviere S, Sanna A, Deroux A, Moulis G, Addimanda O, et al. Efficacy of anti-TNF alpha in severe and/or refractory Behçet’s disease: multicenter study of 124 patients. J Autoimmun. 2015;62:67–74.
Levy-Clarke G, Jabs DA, Read RW, Rosenbaum JT, Vitale A, Van Gelder RN. Expert panel recommendations for the use of anti-tumor necrosis factor biologic agents in patients with ocular inflammatory disorders. Ophthalmology. 2014;121(786–96):e3.
Bendtzen K, Geborek P, Svenson M, Larsson L, Kapetanovic MC, Saxne T. Individualized monitoring of drug bioavailability and immunogenicity in rheumatoid arthritis patients treated with the tumor necrosis factor alpha inhibitor infliximab. Arthritis Rheumatol. 2006;54:3782–9.
Sugita S, Yamada Y, Mochizuki M. Relationship between serum infliximab levels and acute uveitis attacks in patients with Behçet disease. Br J Ophthalmol. 2011;95:549–52.
Iwata D, Namba K, Mizuuchi K, Kitaichi N, Kase S, Takemoto Y, et al. Correlation between elevation of serum antinuclear antibody titer and decreased therapeutic efficacy in the treatment of Behçet’s disease with infliximab. Graefes Arch Clin Exp Ophthalmol. 2012;250:1081–7.
Prevoo ML, van’t Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheumatol. 1995;38:44–8.
Kaburaki T, Namba K, Sonoda K, Kezuka T, Keino H, Fukuhara T, et al. Behçet’s disease ocular attack score 24: evaluation of ocular disease activity before and after initiation of infliximab. Jpn J Ophthalmol. 2014;58:120–30.
Deuter CM, Kotter I, Wallace GR, Murray PI, Stubiger N, Zierhut M. Behçet’s disease: ocular effects and treatment. Prog Retin Eye Res. 2008;27:111–36.
Evereklioglu C. Current concepts in the etiology and treatment of Behçet disease. Surv Ophthalmol. 2005;50:297–350.
Ozdal PC, Ortac S, Taskintuna I, Firat E. Posterior segment involvement in ocular Behçet’s disease. Eur J Ophthalmol. 2002;12:424–31.
Takeuchi M, Hokama H, Tsukahara R, Kezuka T, Goto H, Sakai J, et al. Risk and prognostic factors of poor visual outcome in Behçet’s disease with ocular involvement Graefes. Arch Clin Exp Ophthalmol. 2005;243:1147–52.
No author listed. Criteria for diagnosis of Behcet’s disease. International Study Group for Behcet’s Disease. Lancet. 1990;335:1078–80.
Kim M, Kwon HJ, Choi EY, Kim SS, Koh HJ, Lee SC. Correlation between fluorescein angiographic findings and visual acuity in Behçet retinal vasculitis. Yonsei Med J. 2015;56:1087–96.
Kang HM, Lee SC. Long-term progression of retinal vasculitis in Behçet patients using a fluorescein angiography scoring system. Graefes Arch Clin Exp Ophthalmol. 2014;252:1001–8.
Mesquida M, Molins B, Llorenc V, Hernandez MV, Espinosa G, Dick AD, et al. Current and future treatments for Behçet’s uveitis: road to remission. Int Ophthalmol. 2014;34:365–81.
Atmaca LS, Sonmez PA. Fluorescein and indocyanine green angiography findings in Behçet’s disease. Br J Ophthalmol. 2003;87:1466–8.
Gedik S, Akova Y, Yilmaz G, Bozbeyoglu S. Indocyanine green and fundus fluorescein angiographic findings in patients with active ocular Behçet’s disease. Ocul Immunol Inflamm. 2005;13:51–8.
Keino H, Okada AA, Watanabe T, Taki W. Long-term efficacy of infliximab on background vascular leakage in patients with Behçet’s disease. Eye (Lond). 2014;28:1100–6.
Baneros-Rojas P, Berrozpe-Villabona C, Peraza-Nieves JE, Diaz-Valle D. Early treatment with infliximab in bilateral occlusive vasculitis as a presenting manifestation of Behçet’ disease. Arch Soc Esp Oftalmol. 2015;90:285–8.
Spitznas M. Understanding Fluorescein Angiography. Berlin: Springer; 2006. p. 73–99.
Nussenblatt R. Uveitis: Fundamentals and Clinical Practice. 4th ed. Philadelphia: Elsevier; 2010. p. 42–71.
Baneros-Rojas P, Berrozpe-Villabona C, Peraza-Nieves JE, Diaz-Valle D. Early treatment with infliximab in bilateral occlusive vasculitis as a presenting manifestation of Behçet’ disease. Arch Soc Esp Oftalmol. 2015;90:285–8.
De Bandt M, Sibilia J, Le Loet X, Prouzeau S, Fautrel B, Marcelli C, et al. Systemic lupus erythematosus induced by anti-tumour necrosis factor alpha therapy: a French national survey. Arthritis Res Ther. 2005;7:R545–51.
Teixeira MZ. Biological therapies (immunomodulatory drugs), worsening of psoriasis and rebound effect: new evidence of similitude. Homeopathy. 2016;105:344–55.
van der Maas A, Kievit W, van den Bemt BJ, van den Hoogen FH, van Riel PL, den Broeder AA. Down-titration and discontinuation of infliximab in rheumatoid arthritis patients with stable low disease activity and stable treatment: an observational cohort study. Ann Rheum Dis. 2012;71:1849–54.
Klarenbeek NB, van der Kooij SM, Guler-Yuksel M, van Groenendael JH, Han KH, Kerstens PJ, et al. Discontinuing treatment in patients with rheumatoid arthritis in sustained clinical remission: exploratory analyses from the BeSt study. Ann Rheum Dis. 2011;70:315–9.
Molander P, Farkkila M, Salminen K, Kemppainen H, Blomster T, Koskela R, et al. Outcome after discontinuation of TNFalpha-blocking therapy in patients with inflammatory bowel disease in deep remission. Inflamm Bowel Dis. 2014;20:1021–8.
Tanaka Y, Hirata S. Is it possible to withdraw biologics from therapy in rheumatoid arthritis. Clin Ther. 2013;38:2028–35.
Kim WB, Marinas JE, Qiang J, Shahbaz A, Greaves S, Yeung J. Adverse events resulting in withdrawal of biologic therapy for psoriasis in real-world clinical practice: a Canadian multicenter retrospective study. J Am Acad Dermatol. 2015;73:237–41.
Nakashita T, Ando K, Kaneko N, Takahashi K, Motojima S. Potential risk of TNF inhibitors on the progression of interstitial lung disease in patients with rheumatoid arthritis. BMJ Open. 2014;4:e005615.
Hansen RA, Gartlehner G, Powell GE, Sandler RS. Serious adverse events with infliximab: analysis of spontaneously reported adverse events. Clin Gastroenterol Hepatol. 2007;5:729–35.
Kawaguchi T, Kawazoe Y, Kamoi K, Miyanaga M, Takase H, Sugita S, et al. Clinical course of patients with Behçet’s uveitis following discontinuation of infliximab therapy. Jpn J Ophthalmol. 2014;58:75–80.
Al Rashidi S, Al Fawaz A, Kangave D, Abu El-Asrar AM. Long-term clinical outcomes in patients with refractory uveitis associated with Behçet disease treated with infliximab. Ocul Immunol Inflamm. 2013;21:468–74.
Adan A, Hernandez V, Ortiz S, Molina JJ, Pelegrin L, Espinosa G, et al. Effects of infliximab in the treatment of refractory posterior uveitis of Behçet’s disease after withdrawal of infusions. Int Ophthalmol. 2010;30:577–81.
Acknowledgements
We thank Ms. Teresa Nakatani for critical revision of the manuscript.
This research is supported by the Ministry of Health and Labour Sciences Research Grants of Japan for Research on Rare and Intractable Diseases.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
A. Umazume, None; T. Kezuka, Grant (Mitsubishi Tanabe Pharma), Lecture fee (Mitsubishi Tanabe Pharma); Y. Usui, None; J. Suzuki, None; H. Goto, Grant (Mitsubishi Tanabe Pharma), Lecture fee (Mitsubishi Tanabe Pharma).
About this article
Cite this article
Umazume, A., Kezuka, T., Usui, Y. et al. Evaluation of efficacy of infliximab for retinal vasculitis and extraocular symptoms in Behçet disease. Jpn J Ophthalmol 62, 390–397 (2018). https://doi.org/10.1007/s10384-018-0589-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10384-018-0589-2