Summary
In recent years, new treatment options for both common and rare bone diseases have become available. The sclerostin antibody romosozumab is the most recently approved drug for the therapy of postmenopausal osteoporosis. Its anabolic capacity makes it a promising treatment option for severe osteoporosis. Other sclerostin antibodies for the treatment of rare bone diseases such as osteogenesis imperfecta are currently being investigated. For rare bone diseases such as X‑linked hypophosphatemia (XLH) and hypophosphatasia (HPP), specific therapies are now also available, showing promising data in children and adults with a severe disease course. However, long-term data are needed to assess a sustained benefit for patients.
Zusammenfassung
Neue therapeutische Möglichkeiten zur Therapie der Osteoporose, aber auch seltener Knochenerkrankungen stehen neuerdings zu Verfügung. Der Sclerostin-Antikörper Romosozumab wurde unlängst zur Behandlung der postmenopausalen Osteoporose zugelassen. Durch seine osteoanabole Wirkung scheint Romosozumab eine vielversprechende Therapieoption der schweren Osteoporose zu sein. Andere Sclerostin-Antikörper werden derzeit bei seltenen Knochenerkrankungen wie der Osteogenesis imperfecta untersucht. Für den Phosphatdiabetes („X-linked hypophosphatemia“, XLH) und die Hypophosphatasie (HPP) stehen nun spezifische Therapien zur Verfügung, die in den bisherigen Studien überzeugende Ergebnisse zeigten. Langzeitdaten sind jedoch erforderlich, um das Risiko-Nutzen-Profil abschätzen zu können.
Similar content being viewed by others
References
Kocijan R, Klaushofer K, Misof BM. Osteoporosis therapeutics 2020. Handbook of experimental pharmacology. 2020.
Padhi D, et al. Single-dose, placebo-controlled, randomized study of AMG 785, a sclerostin monoclonal antibody. J Bone Miner Res. 2011;26(1):19–26.
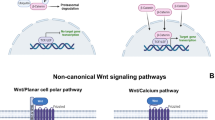
MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17(1):9–26.
Brunkow ME, et al. Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein. Am J Hum Genet. 2001;68(3):577–89.
McClung MR, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014;370(5):412–20.
Cosman F, et al. Romosozumab treatment in postmenopausal women with osteoporosis. N Engl J Med. 2016;375(16):1532–43.
Cosman F, et al. Romosozumab FRAME study: a post hoc analysis of the role of regional background fracture risk on nonvertebral fracture outcome. J Bone Miner Res. 2018;33(8):1407–16.
Saag KG, et al. Romosozumab or alendronate for fracture prevention in women with osteoporosis. N Engl J Med. 2017;377(15):1417–27.
Lewiecki EM, et al. A phase III randomized placebo-controlled trial to evaluate efficacy and safety of romosozumab in men with osteoporosis. J Clin Endocrinol Metab. 2018;103(9):3183–93.
Miyauchi A, et al. Increased bone mineral density for 1 year of romosozumab, vs placebo, followed by 2 years of denosumab in the Japanese subgroup of the pivotal FRAME trial and extension. Arch Osteoporos. 2019;14(1):59.
Lewiecki EM, et al. One year of romosozumab followed by two years of denosumab maintains fracture risk reductions: results of the FRAME extension study. J Bone Miner Res. 2019;34(3):419–28.
McClung MR. Romosozumab for the treatment of osteoporosis. Osteoporos Sarcopenia. 2018;4(1):11–5.
Ebina K, et al. Effects of prior osteoporosis treatment on early treatment response of romosozumab in patients with postmenopausal osteoporosis. Bone. 2020;140:115574.
Langdahl BL, et al. Romosozumab (sclerostin monoclonal antibody) versus teriparatide in postmenopausal women with osteoporosis transitioning from oral bisphosphonate therapy: a randomised, open-label, phase 3 trial. Lancet. 2017;390(10102):1585–94.
Bovijn J, et al. Evaluating the cardiovascular safety of sclerostin inhibition using evidence from meta-analysis of clinical trials and human genetics. Sci Transl Med. 2020;12(549):eaay6570. https://doi.org/10.1126/scitranslmed.aay6570.
Sinder BP, et al. Sclerostin antibody improves skeletal parameters in a Brtl/+ mouse model of osteogenesis imperfecta. J Bone Miner Res. 2013;28(1):73–80.
Sinder BP, et al. Rapidly growing Brtl/+ mouse model of osteogenesis imperfecta improves bone mass and strength with sclerostin antibody treatment. Bone. 2015;71:115–23.
Sinder BP, et al. Adult Brtl/+ mouse model of osteogenesis imperfecta demonstrates anabolic response to sclerostin antibody treatment with increased bone mass and strength. Osteoporos Int. 2014;25(8):2097–107.
Cardinal M, et al. Sclerostin antibody reduces long bone fractures in the oim/oim model of osteogenesis imperfecta. Bone. 2019;124:137–47.
Roschger A, et al. Effect of sclerostin antibody treatment in a mouse model of severe osteogenesis imperfecta. Bone. 2014;66:182–8.
Glorieux FH, et al. BPS804 anti-sclerostin antibody in adults with moderate osteogenesis Imperfecta: results of a randomized phase 2a trial. J Bone Miner Res. 2017;32(7):1496–504.
Seefried L, et al. Efficacy of anti-sclerostin monoclonal antibody BPS804 in adult patients with hypophosphatasia. J Clin Invest. 2017;127(6):2148–58.
Raimann A, Mindler G, Kocijan R, Bekes K, Zwerina J, Haeusler G, et al. Multidisciplinary patient care in X‑linked hypophosphatemic rickets: one challenge, many perspectives. Wien Med Wochenschr. 2020;170:116–23.
Che H, et al. Impaired quality of life in adults with X‑linked hypophosphatemia and skeletal symptoms. Eur J Endocrinol. 2016;174(3):325–33.
Skrinar A, et al. The lifelong impact of X‑linked hypophosphatemia: results from a burden of disease survey. J Endocr Soc. 2019;3(7):1321–34.
Chesher D, et al. Outcome of adult patients with X‑linked hypophosphatemia caused by PHEX gene mutations. J Inherit Metab Dis. 2018;41(5):865–76.
Haffner D, et al. Clinical practice recommendations for the diagnosis and management of X‑linked hypophosphataemia. Nat Rev Nephrol. 2019;15(7):435–55.
Connor J, et al. Conventional therapy in adults with X‑linked hypophosphatemia: effects on enthesopathy and dental disease. J Clin Endocrinol Metab. 2015;100(10):3625–32.
Carpenter TO, et al. A clinician’s guide to X‑linked hypophosphatemia. J Bone Miner Res. 2011;26(7):1381–8.
Fratzl-Zelman N, et al. Alterations of bone material properties in adult patients with X‑linked hypophosphatemia (XLH). J Struct Biol. 2020;211(3):107556.
Schindeler A, Biggin A, Munns CF. Clinical evidence for the benefits of burosumab therapy for X‑linked hypophosphatemia (XLH) and other conditions in adults and children. Front Endocrinol (Lausanne). 2020;11:338.
Imel EA, et al. Burosumab versus conventional therapy in children with X‑linked hypophosphataemia: a randomised, active-controlled, open-label, phase 3 trial. Lancet. 2019;393(10189):2416–27.
Insogna KL, et al. A randomized, double-blind, placebo-controlled, phase 3 trial evaluating the efficacy of burosumab, an anti-FGF23 antibody, in adults with X‑linked hypophosphatemia: week 24 primary analysis. J Bone Miner Res. 2018;33(8):1383–93.
Portale AA, et al. Continued beneficial effects of burosumab in adults with X‑linked hypophosphatemia: results from a 24-week treatment continuation period after a 24-week double-blind placebo-controlled period. Calcif Tissue Int. 2019;105(3):271–84.
Lowe D, Sanvictores T, John S. Alkaline phosphatase. Treasure Island (FL): StatPearls Publishing LLC; 2020.
Salles JP. Hypophosphatasia: biological and clinical aspects, avenues for therapy. Clin Biochem Rev. 2020;41(1):13–27.
Vogt M, et al. Pediatric hypophosphatasia: lessons learned from a retrospective single-center chart review of 50 children. Orphanet J Rare Dis. 2020;15(1):212.
Choida V, Bubbear JS. Update on the management of hypophosphatasia. Ther Adv Musculoskelet Dis. 2019;11:1759720x19863997.
Kishnani PS, et al. Five-year efficacy and safety of asfotase alfa therapy for adults and adolescents with hypophosphatasia. Bone. 2019;121:149–62.
Bowden SA, Adler BH. Asfotase alfa treatment for 1 year in a 16 year-old male with severe childhood hypophosphatasia. Osteoporos Int. 2018;29(2):511–5.
Hofmann C, Seefried L, Jakob F. Asfotase alfa: enzyme replacement for the treatment of bone disease in hypophosphatasia. Drugs Today (Barc). 2016;52(5):271–85.
Whyte MP, et al. Asfotase alfa for infants and young children with hypophosphatasia: 7 year outcomes of a single-arm, open-label, phase 2 extension trial. Lancet Diabetes Endocrinol. 2019;7(2):93–105.
Whyte MP, et al. Asfotase alfa treatment improves survival for perinatal and infantile hypophosphatasia. J Clin Endocrinol Metab. 2016;101(1):334–42.
Genest F, et al. Physical function and health-related quality of life in adults treated with asfotase alfa for pediatric-onset hypophosphatasia. JBMR Plus. 2020;4(9):e10395.
Simon S, et al. Hypophosphatasia: from diagnosis to treatment. Curr Rheumatol Rep. 2018;20(11):69.
Magdaleno AL, et al. Adult-onset hypophosphatasia: before and after treatment with ASFOTASE ALFA. AACE Clin Case Rep. 2019;5(6):e344–e8.
Klidaras P, et al. Fracture healing in two adult patients with hypophosphatasia after asfotase alfa therapy. JBMR Plus. 2018;2(5):304–7.
Rolvien T, et al. Recovery of bone mineralization and quality during asfotase alfa treatment in an adult patient with infantile-onset hypophosphatasia. Bone. 2019;127:67–74.
Freitas TQ, Franco AS, Pereira RMR. Improvement of bone microarchitecture parameters after 12 months of treatment with asfotase alfa in adult patient with hypophosphatasia: case report. Medicine. 2018;97(48):e13210.
Seefried L, et al. Pharmacodynamics of asfotase alfa in adults with pediatric-onset hypophosphatasia. Bone. 2020; https://doi.org/10.1016/j.bone.2020.115664.
Whyte MP, et al. No vascular calcification on cardiac computed tomography spanning asfotase alfa treatment for an elderly woman with hypophosphatasia. Bone. 2019;122:231–6.
Bowden SA, Adler BH. Reappearance of hypomineralized bone after discontinuation of asfotase alfa treatment for severe childhood hypophosphatasia. Osteoporos Int. 2018;29(9):2155–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R. Kocijan previously received speaker fees, travel grants, research grants, fees for advisory-board and seminars from Alexion, UCB, Amgen and Kyowa Kirin. J. Haschka previously received speaker fees and research grants from Alexion and Amgen. J. Feurstein and J. Zwerina declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kocijan, R., Haschka, J., Feurstein, J. et al. New therapeutic options for bone diseases. Wien Med Wochenschr 171, 120–125 (2021). https://doi.org/10.1007/s10354-020-00810-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10354-020-00810-w