Abstract
Background
In this study, we aimed to evaluate pediatric liver transplantation in terms of outcomes, complications, and long-term follow-up results.
Materials and methods
Between September 2001 and August 2015, 215 pediatric liver transplants were performed in our institution.
Results
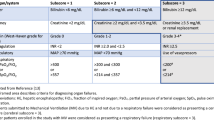
Posttransplant biliary complications were biliary leakage (16.2 %) and biliary stricture (14.8 %). Complication rates in hepatic artery and portal vein were 16.7 % and 4.6 %. Overall mortality rate was 17.8 %. The overall 5‑year survival rate was 82.3 % and 10-year survival rate was 78.9 %.
Conclusion
With improved care of younger children and the combined efforts of the parents and medical team, the number of the children receiving transplants with good outcomes will continue to increase.

Similar content being viewed by others
References
Ng VL, Alonso EM, Bucuvalas JC, et al. Health status of children alive 10 years after pediatric liver transplantation performed in the US and Canada: report of the studies of pediatric liver transplantation experience. J Pediatr. 2012;160(5):820–826. e3.
de Ville de Goyet J, Hausleithner V, Reding R, Lerut J, Janssen M, Otte JB. Impact of innovative techniques on the waiting list and results in pediatric liver transplantation. Transplantation. 1993;56(5):1130–1136.
Kasahara M, Umeshita K, Inomata Y, Uemoto S, Japanese Liver Transplantation Society. Long-term outcomes of pediatric living donor liver transplantation in japan: an analysis of more than 2200 cases listed in the registry of the japanese liver transplantation society. Am J Transplant. 2013;13(7):1830–1839.
Haberal M, Sevmis S, Karakayali H, Ozcay F, Moray G, Yilmaz U, Torgay A, Demirhan B, Arslan G. Pediatric liver transplant: results of a single center. Exp Clin Transplant. 2008;6(1):7–13.
Akdur A, Kirnap M, Ozcay F, Sezgin A, Ayvazoglu SHE, Karakayali YF, Yildirim S, Moray G, Arslan G, Haberal M. Large-for-size liver transplant: a single-center experience. Exp Clin Transplant. 2015;13(Suppl 1):108–110.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M Haberal states that there are no conflicts of interest.
Ethical standards
Informed consent was obtained from all patients for being included in the study.
Additional information
The supplement is dedicated to Jubilee World congress of International College of Surgeons 2015.
The publishing was sponsored by the Ministry of Health of the Czech Republic.
Rights and permissions
About this article
Cite this article
Haberal, M. Pediatric liver transplantation experience. Eur Surg 48 (Suppl 2), 169–171 (2016). https://doi.org/10.1007/s10353-016-0399-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-016-0399-z