Abstract
Purpose
This study was designed to determine whether the injection of muscle-derived stem cells into the anal sphincter can improve functional properties in a fecal incontinence rat model.
Methods
Cryoinjured rats were utilized as a fecal incontinence model. The gastrocnemius muscles of normal three-week-old female Sprague-Dawley rats were used for the purification of the muscle-derived stem cells. The experimental group was divided into three subgroups: normal control; cryoinjured; and muscle-derived stem cells (3 × 106 cells) injection group of cryoinjured rats. All groups were subsequently employed in contractility experiments using muscle strips from the anal sphincter, one week after preparation.
Results
Contractility in the cryoinjured group was significantly lower than in the control after treatment with acetylcholine and KCl. In the muscle-derived stem cells injection group, contraction amplitude was higher than in the cryoinjured group but not significantly (20.5 ± 21.3 vs. 17.3 ± 3.4 g per gram tissue, with acetylcholine (10−4 mol/l); 31 ± 14.2 vs. 18.4 ± 7.9 g per gram tissue, with KCl (10−4 mol/l)). PKH-26-labeled transplanted cells were detected in all of the grafted sphincters. Differentiated muscle masses stained positively for alpha smooth muscle actin and myosin heavy chain at the muscle-derived stem cells injection sites.
Conclusions
This is the first study reporting that autologous muscle-derived stem cell grafts may be a tool for improving anal sphincter function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Fecal incontinence (FI) remains a socially isolating condition, which can have a profound impact on all aspects of quality of life. A systematic review on the prevalence of FI in community-dwelling adults has shown that the estimated prevalence varied from 0.4 to 18 percent.1 The most common cause of FI is damage to the anal sphincter or nerve complex resulting from disease process or traumas, including obstetric injuries and anorectal surgery. One surgical approach to the treatment of FI has been bulking agents for the augmentation of muscle bulk and to restore sphincter anatomy in sites in which it is deficient, or circumferentially in cases in which the whole muscle was degenerate. Since the initial report in 1993,2 several agents have been used in the treatment of FI with glutaraldehyde cross-linked collagen,3 autologous fat,4 injectable silicone,5–8 and carbon beads.9–11 Injection therapy improved the patient’s symptoms2–11 in nonrandomized studies, although the reported improvements in resting or squeeze pressure remain controversial. However, the efficacy of injectable agents is limited by several factors, including reabsorption, allergic reactions, and migration, even when injection therapy is utilized for the treatment of urinary incontinence.10 Clearly, “living” contractile bulking agents, which improve sphincter muscle function, would be ideal, because conventional bulking agents exert only a “bulk” effect, without effecting any improvements in anal sphincter contractility.
There are several reports about muscle-derived stem cells, but the definition of muscle-derived stem cells (MDSC) is still unclear. Satellite cells play a central role in regeneration of skeletal muscle, and a recent study showed that satellite cells are a heterogeneous population composed of stem cells and committed progenitors.12,13 The injection of MDSC into the periurethral sphincter was effective in a rat urinary incontinence model.14–20 Recently, Strasser et al. demonstrated that injection therapy using autologous myoblasts and fibroblasts yielded better outcomes than collagen injection for the treatment of stress urinary incontinence in women in a randomized, controlled trial.21 However, the question as to whether these MDSC are bulking the urethra or promoting the functional recovery of the injured sphincter remains to be satisfactorily answered.
In the coloproctology field, some authors have demonstrated that autologous stem cells obtained from a lipoaspirate may be utilized for the treatment of fistulas in Crohn’s disease without any adverse effects.22,23 Shafik4 has shown that perianal fat injection was effective in the treatment of partial fecal incontinence with no fat migration or embolism. Until recently, no studies have been conducted to determine the functional and histologic effects that occur after the injection of stem cells into the anal sphincter in a FI animal model. In the present study, we have attempted to determine whether injected MDSC can effect improvements in the functional properties of a rat FI model.
Materials and Methods
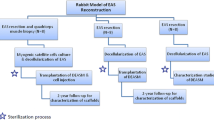
The Animal Care and Use Committee of Seoul National University Bundang Hospital approved all procedures performed on animals (Protocol No. 64-2007-028). All experiments were conducted using normal three-week-old (200–250 g) female Sprague-Dawley (SD) rats. Three groups were used: a normal control group that received no injections; a cryoinjured group; and a MDSC injection group of cryoinjured rats. The MDSC injection group was subjected to cryoinjury and MDSC injection under single anesthetic conditions. All groups were subsequently subjected to contractility experiments one week later.
MDSC Purification and Characterization
MDSC were harvested from the gastrocnemius muscles of rats and purified via the preplate technique.24 The muscle biopsy was obtained from the hind limb and was minced into a coarse slurry with successively smaller needles. The cells were then enzymatically dissociated via the addition of collagenase type XI 0.2 percent (Sigma, St. Louis, MO) for 1 hour at 37°C, 240 units of Grade II dispase (Gibco-BRL, Grand Island, NY) for 45 minutes, and trypsin 0.1 percent (Gibco-BRL) for 30 minutes. The dissociated cells were then preplated on collagen and poly-D-lysin (Sigma, St. Louis, MO)-coated flasks. The early preplated cells and fibroblasts were observed to adhere to the first flask. The early plated cells were discarded because they do not proliferate well, and they survive poorly after transplantation. All cells that failed to adhere to the flask were then transferred to another flask for approximately one hour and were serially transferred and incubated.24 The cells used in this experiment were obtained from the last preplate. For the characterization of cultured cells, predetermined optimal amounts of rat anti-mouse monoclonal antibodies (CD-34, CD-45, Desmin) were directly added to each tube for 30 minutes. The proliferation medium used to grow the cells was Dulbecco’s modified Eagle medium (Gibco-BRL) supplemented with 10 percent fetal bovine serum (Gibco-BRL), 10 percent horse serum (Gibco-BRL), 0.5 percent chick embryo extract (Sera Laboratories International Ltd, UK), and 1 percent penicillin/streptomycin (Gibco-BRL). For the labeling of the MDSC, PKH-26 dye (an aliphatic fluorescent chromophore, Sigma) was used to label the cells before their injection, which is a lipophilic fluorescent dye that binds irreversibly to the cell membrane and is not transferable to other cells.
Cryoinjury in Anal Sphincter of Rat and Autologous MDSC Injection
The cryoinjured group and the MDSC injection group were administered ketamine (80 mg/kg), and after appropriate induction, cryoinjury of the anal sphincter was induced using an aluminum rod with a diameter of 2 mm, which was chilled with liquid nitrogen. The chilled probe was positioned against the right hemisphere of the anal sphincter for 30 seconds. This mode of injury is the modification of a previously described procedure.25
Approximately five minutes after the injury, MDSC suspended in Hanks’ balanced salt solution (Gibco-BRL; 3 × 106 cells per 100 μl) was injected manually into the middle anal sphincter of the MDSC group rats using a 50 μl Hamilton microsyringe (25 gauge needle; Hamilton, Reno, NV) with microscopic guidance. Three injections per rat (30 μl each) were performed on the right hemisphere of the anal sphincter, which had been subjected to cryoinjury.
Preparation of Muscle Strips and Contractility Test
Each group underwent contractility experiments at one week after surgery. After anesthetic induction in all groups, the anal sphincter was trimmed and transferred into a bath perfused with phosphate-buffered saline. The transverse loop of the sphincter was incised on the left side, and each strip measured approximately 1 × 1 × 10 mm (width and length). The muscle strips were tied with 3-0 silk sutures to a glass hook, and the other end was connected to a Grass Force Transducer (FT03, Grass Instruments, Quincy, MA), which was connected to an amplifier (P-122, Grass). The strips were mounted in a 20 ml bath containing Tyrode’s solution (116 mM NaCl, 5 mM KCl, 5 mM HEPES, 1 mM MgCl2, 24 mM NaHCO3, 2 mM CaCl2, and 11.5 mM glucose) at 37°C and were equilibrated with constant bubbling with a mixture of 95 percent O2-5 percent CO2.
The strips were equilibrated for 30 minutes at 2 g resting tension for the recording of isometric tension in superfusion organ baths. The strips were then stimulated for 20 seconds at 32 Hz with 1 millisecond pulses at 80 V using an S48 (Grass Instruments) stimulator. After electrical field stimulation, acetylcholine (Ach) (10−4 mol/l) or KCl (10−4 mol/l) was added to the organ baths to determine the contractile activities of the anal sphincter. A small air bubble was introduced as each drug was administered to ensure minimal mixing and/or dilution of the drug. For KCl treatment after Ach, the strips were washed three times in fresh buffer. Twitch contractions were determined with strain-gauge transducers and recorded on a Model 7D Polygraph. The analog signals obtained from the Force Transducer were converted to digital signals at a 3 Hz sampling rate (Polyview, Grass) and were stored in a computer database for further analysis.
Immunohistochemical Staining
Immediately after the recording of isotonic tension, the strips were weighed and frozen in liquid nitrogen. Serial 5 μm thickness cryostat sections were obtained around each of the injection sites, stained with hematoxylin/eosin (H/E), examined microscopically and additionally via fluorescence microscopy for the detection of PKH-26-labelled cells, and photographed. The cytoplasm of PKH-26-labeled cells showed red fluorescence as detected by fluorescence microscopy, whereas the nucleus remained unstained. PKH-26-labeled cells are usually considered viable because the dye leaves the cell membrane after cell death.26 Myonuclei were visualized with 4′,6-diamidine-2-phenylindole (DAPI). Immunohistochemical staining also was conducted for alpha-smooth muscle actin (SMA) monoclonal antibody (diluted to 1:100; Sigma) and myogenin monoclonal antibody (diluted 1:200; InVitroGen). To evaluate the immune-privileged behavior of injected cells in MDSC rats, immunohistochemical staining to detect CD3 marker was performed with a positive control of a normal rat. Muscle strips of the anal sphincter underwent CD3 staining after harvest. The primary antibody was a mouse monoclonal antibody against CD3 that was incubated for one hour at room temperature. The CD3 is a pan-T-cell marker that is positive for CD4 and CD8 lymphocytes.
Statistical Analysis
Contractility data are expressed as means ± standard deviations. Paired t-tests were used for comparison between data at one week. P < 0.05 was considered to be statistically significant. All calculations were conducted by using SPSS® 11.0 software (SPSS, Inc., Chicago, IL).
Results
Characterization of MDSC
We isolated MDSC from rat gastrocnemius muscles, and the cells were characterized via immunostaining. The MDSC were determined to evidence stem-cell characteristics (CD34+) and were not of hematogenous (CD45-) but rather myogenic origin (desmin+).14,27 They were used for further experiments as MDSC.
Contractility
The maximal contraction amplitude of the anal sphincter strip in the cryoinjured group was statistically significantly lower than in the normal control group after Ach (10−4 mol/l) and KCl (10−4 mol/l) treatment (17.3 ± 3.4 vs. 35.1 ± 6.2 g per gram tissue, P = 0.001; 18.4 ± 7.9 vs. 46.9 ± 14.1 g per gram tissue, P = 0.006, respectively; Table 1). Representative examples of anal sphincter contractility studies after Ach (10−4 mol/l) treatment are shown in Fig. 1a-c. In the MDSC injection group, the contraction of the sphincter strip was higher than in the cryoinjured group after treatment with Ach (10−4 mol/l) and KCl (10−4 mol/l) treatment, but this was not significantly different (20.5 ± 21.3 vs. 17.3 ± 3.4 g per gram tissue, P = 0.748; 31 ± 14.2 vs. 18.4 ± 7.9 g per gram tissue, P = 0.120, respectively).
Histologic Findings
The anal sphincters of the normal control group harbored both smooth and skeletal muscle (Fig. 2a). A layer of outer striated muscle fibers encircles the smooth muscle layers. The cryoinjured group showed damaged muscle fibers with cytoplasmic vacuolization and focal interstitial inflammatory cell infiltration (Fig. 2b). Regenerating muscle fibers with enlarged nuclei were observed in the variable orientation at the muscle-derived stem cell injection site (Fig. 2c). PKH-26-labeled transplanted cells were detected in all of the grafted sphincters at seven days after injection (Fig. 3). Regenerating myofibers of smooth and skeletal muscle stained positively for alpha-SMA and myosin heavy-chain (MHC) at the MDSC injection sites. There was no observed immune response, such as lymphocytic infiltration, vasculitis, or single-cell apoptosis, around the site of MDSC injection. CD3 staining was not observed at MDSC injection sites at seven days (Fig. 4).
Hematoxylin-eosin staining. a. Normal histology of the anal sphincter in normal control rats. Internal smooth muscle (arrowhead) and external skeletal muscle (arrow) (magnification, ×10). b. Damaged muscle fibers with cytoplasmic vacuolization and focal interstitial inflammatory cell infiltration in the cryoinjured group (magnification, ×400). c. Regenerating muscle fibers in the variable orientation with enlarged nuclei at the muscle-derived stem cell injection site (magnification, ×400).
Muscle formation in the cryoinjured anal sphincter following muscle-derived stem cell injection. a-c. Normal control group shows a thick smooth muscle layer encircled by a layer of outer striated muscle fibers. d-f. Cryoinjured group shows weakened smooth muscle and skeletal muscle layer. G. Muscle-derived stem cell injection group shows the variable orientation of the new muscle fiber. h. Differentiated smooth muscle (yellow) shown in colocalization of injected muscle-derived stem cell injection (PKH-26-staining, red) and smooth muscle via the immunostaining of alpha smooth muscle actin (green). i. Differentiated skeletal muscle (yellow) shown in the colocalization of injected muscle-derived stem cell injection (PKH-26-staining, red) and skeletal muscle via immunostaining of myosin heavy-chain (green). Hematoxylin-eosin staining (a, d, g), immunostaining of alpha smooth muscle actin (b, e, h), and myosin heavy-chain (c, f, i) (magnification, ×100).
Discussion
Injection therapy for the treatment of anal sphincter contractility defects is a minimally invasive procedure, which can be readily and rapidly performed by an experienced colorectal surgeon/physician in an outpatient setting. However, the injectable agents have potential disadvantages, including the need for multiple injections, allergic reactions caused by foreign agents, and migration. Stem-cell therapy as a “living” contractile bulking agent may be an ideal tool, which is unlikely to result in migration problems, as have been associated with Teflon, Silicon, and Durasphere.10
In the present study, we used a highly specific population of cells that evidence stem cell features compared with myoblasts. These cells were highly purified MDSC, which were identified via immunohistochemical staining as CD34+, CD45-, and desmin+.13,28,29 These cells have been reported to evidence an improved survival rate posttransplantation because they do not trigger immune reactions as do regular myoblasts after injection.28 In this study, injection of autograft MDSC was immunologically inert. MDSC do not fuse quickly with each other and have been shown to be capable of proliferating more efficiently. In the current study, we attempted to determine whether injected MDSC could survive in the anal sphincter, differentiate into myofibrils, and ultimately enhance contractility. The injected PKH-26-labeled MDSC have shown improved survival compared with myoblasts, which do not survive beyond 48 hours after injection.28 MDSC are reported to be durable and have been demonstrated to survive in the lower urinary tract for up to six months postinjection.30
We demonstrated that the MDSC had integrated within the muscle layer of the cryoinjured anal sphincter and that new muscle fiber had formed at the sphincter injection sites, as evidenced by histologic examination. This study verified that differentiated muscle cells originate from transplanted cells via the colocalization of injected muscle-derived stem cell with PKH-26-staining and muscles, as shown by the immunostaining of MHC or alpha-SMA. It means that autologous skeletal cell grafts can switch to slow-twitch fibers.31
In this study, the FI rat model was validated because the contraction amplitudes of the cryoinjured rats were significantly lower than those of normal control rats. Using this cryoinjured animal model, we evaluated the effects of MDSC injected into cryoinjured anal sphincter tissue via measurements of the contractility of the muscle strips. In the current study, MDSC were considered to have potential as a functional bulking agent on the basis of contractility tests. However, the effects of MDSC did not improve significantly. This insignificant result may be attributed to the following points. First, this study involved small volume injections of 3 × 106 MDSC. We believe that more MDSC should be injected for FI, which differs from urinary incontinence with regard to muscle bulk. Second, contractility is representative of the functioning of the active components of the anal sphincter. It remains unclear as to whether MDSC autografting would restore sphincter muscle function or simply function as a simple bulking agent. It should be determined in the future whether injected MDSC develop into functional tissue, thus resulting in increased contractility. Third, it remains to be determined whether injected MDSC differentiate into myofibers for contraction with reinnervation. The results of some animal studies have indicated that muscle reinnervation may be possible via electrical stimulation, or the addition of fibroblasts or Schwann cells.15,32,33 Yiou et al. 16 also demonstrated that the maturation of muscle precursor cells activated nerve regeneration and restored functional motor units. Although adult skeletal muscle includes a population of progenitor stem cells that can generate cells of the myogenic lineage and cells with neurogenic properties,34 it remains to be determined whether stem cells can induce the functional reinnervation of regenerated muscles. Fourth, MDSC injection therapy may have certain disadvantages, including migration and reabsorbtion, as is the case with other commercial injection agents. Fifth, the insignificance could simply be a Type II error, with only five rats in each group, although the trends were actually quite strong. Sixth, Ach or KCl was added to the organ baths to determine the contractile activities of the anal sphincter in this study. We think that some agents may induce a greater effective stimulation of muscle other than KCl or Ach, which might give a significant improvement in muscle contractility.
The current study was for a short-term period. Although we have demonstrated engraftment of MDSC in the injury site, it remains to be determined whether MDSC would continue to differentiate over time. The risk of tumor formation or adequate functional improvement may be determined in the future via a long-term study. However, we considered that a long-term period, in which sphincter regeneration may be induced by the intrinsic satellite cells, may be a bias factor. In this study, the FI rat model was validated, although the animal model utilized herein is not perfect. This is the first study reporting that autologous MDSC grafts may be used as a tool for improving anal sphincter contractility.
References
Macmillan AK, Merrie AE, Marshall RJ, Parry BR. The prevalence of fecal incontinence in community-dwelling adults: a systematic review of the literature. Dis Colon Rectum 2004;47:1341–9.
Shafik A. Polytetrafluoroethylene injection for the treatment of partial fecal incontinence. Int Surg 1993;78:159–61.
Kumar D, Benson MJ, Bland JE. Glutaraldehyde cross-linked collagen in the treatment of faecal incontinence. Br J Surg 1998;85:978–9.
Shafik A. Perianal injection of autologous fat for treatment of sphincteric incontinence. Dis Colon Rectum 1995;38:583–7.
Malouf AJ, Vaizey CJ, Norton CS, Kamm MA. Internal anal sphincter augmentation for fecal incontinence using injectable silicone biomaterial. Dis Colon Rectum 2001;44:595–600.
Chan MK, Tjandra JJ. Injectable silicone biomaterial (PTQ) to treat fecal incontinence after hemorrhoidectomy. Dis Colon Rectum 2006;49:433–9.
Tjandra JJ, Lim JF, Hiscock R, Rajendra P. Injectable silicone biomaterial for fecal incontinence caused by internal anal sphincter dysfunction is effective. Dis Colon Rectum 2004;47:2138–46.
Kenefick NJ, Vaizey CJ, Malouf AJ, Norton CS, Marshall M, Kamm MA. Injectable silicone biomaterial for faecal incontinence due to internal anal sphincter dysfunction. Gut 2002;51:225–8.
Davis K, Kumar D, Poloniecki J. Preliminary evaluation of an injectable anal sphincter bulking agent (Durasphere) in the management of faecal incontinence. Aliment Pharmacol Ther 2003;18:237–43.
Pannek J, Brands FH, Senge T. Particle migration after transurethral injection of carbon coated beads for stress urinary incontinence. J Urol 2001;166:1350–3.
Emans PJ, Saralidze K, Knetsch ML, Gijbels MJ, Kuijer R, Koole LH. Development of new injectable bulking agents: biocompatibility of radiopaque polymeric microspheres studied in a mouse model. J Biomed Mater Res A 2005;73:430–6.
Kuang S, Kuroda K, Le Grand F, Rudnicki MA. Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell 2007;129:999–1010.
Qu-Petersen Z, Deasy B, Jankowski R, et al. Identification of a novel population of muscle stem cells in mice: potential for muscle regeneration. J Cell Biol 2002;157:851–64.
Lee JY, Paik SY, Yuk SH, Lee JH, Ghil SH, Lee SS. Long term effects of muscle-derived stem cells on leak point pressure and closing pressure in rats with transected pudendal nerves. Mol Cells 2004;18:309–13.
Cannon TW, Lee JY, Somogyi G, et al. Improved sphincter contractility after allogenic muscle-derived progenitor cell injection into the denervated rat urethra. Urology 2003;62:958–63.
Yiou R, Yoo JJ, Atala A. Restoration of functional motor units in a rat model of sphincter injury by muscle precursor cell autografts. Transplantation 2003;76:1053–60.
Kwon D, Kim Y, Pruchnic R, et al. Periurethral cellular injection: comparison of muscle-derived progenitor cells and fibroblasts with regard to efficacy and tissue contractility in an animal model of stress urinary incontinence. Urology 2006;68:449–54.
Lee JY, Cannon TW, Pruchnic R, Fraser MO, Huard J, Chancellor MB. The effects of periurethral muscle-derived stem cell injection on leak point pressure in a rat model of stress urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct 2003;14:31–7.
Yiou R, Dreyfus P, Chopin DK, Abbou CC, Lefaucheur JP. Muscle precursor cell autografting in a murine model of urethral sphincter injury. BJU Int 2002;89:298–302.
Chermansky CJ, Tarin T, Kwon DD, et al. Intraurethral muscle-derived cell injections increase leak point pressure in a rat model of intrinsic sphincter deficiency. Urology 2004;63:780–5.
Strasser H, Marksteiner R, Margreiter E, et al. Autologous myoblasts and fibroblasts versus collagen for treatment of stress urinary incontinence in women: a randomised controlled trial. Lancet 2007;369:2179–86.
Garcia-Olmo D, Garcia-Arranz M, Garcia LG, et al. Autologous stem cell transplantation for treatment of rectovaginal fistula in perianal Crohn’s disease: a new cell-based therapy. Int J Colorectal Dis 2003;18:451–4.
Garcia-Olmo D, Garcia-Arranz M, Herreros D, Pascual I, Peiro C, Rodriguez-Montes JA. A phase I clinical trial of the treatment of Crohn’s fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum 2005;48:1416–23.
Rando TA, Blau HM. Primary mouse myoblast purification, characterization, and transplantation for cell-mediated gene therapy. J Cell Biol 1994;125:1275–87.
Huard J, Yokoyama T, Pruchnic R, et al. Muscle-derived cell-mediated ex vivo gene therapy for urological dysfunction. Gene Ther 2002;9:1617–26.
Gulbins H, Pritisanac A, Anderson I, et al. Myoblasts for survive 16 weeks after intracardiac transfer and start differentiation. Thorac Cardiovasc Surg 2003;51:295–300.
Hwang JH, Yuk SH, Lee JH, et al. Isolation of muscle derived stem cells from rat and its smooth muscle differentiation. Mol Cells 2004;17:57–61.
Qu Z, Balkir L, van Deutekom JC, Robbins PD, Pruchnic R, Huard J. Development of approaches to improve cell survival in myoblast transfer therapy. J Cell Biol 1998;142:1257–67.
Beauchamp JR, Heslop L, Yu DS, et al. Expression of CD34 and Myf5 defines the majority of quiescent adult skeletal muscle satellite cells. J Cell Biol 2000;151:1221–34.
Yokoyama T, Huard J, Chancellor MB. Myoblast therapy for stress urinary incontinence and bladder dysfunction. World J Urol 2000;18:56–61.
Hagège AA, Carrion C, Menasché P, et al. Viability and differentiation of autologous skeletal myoblast grafts in ischaemic cardiomyopathy. Lancet 2003;361:491–2.
Alameddine HS, Louboutin JP, Dehaupas M, Sebille A, Fardeau M. Functional recovery induced by satellite cell grafts in irreversibly injured muscles. Cell Transplant 1994;3:3–14.
Guettier-Sigrist S, Coupin G, Warter JM, Poindron P. Cell types required to efficiently innervate human muscle cells in vitro. Exp Cell Res 2000;259:204–12.
Alessandri G, Pagano S, Bez A, et al. Isolation and culture of human muscle-derived stem cells able to differentiate into myogenic and neurogenic cell lineages. Lancet 2004;364:1872–83.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Kang, SB., Lee, H.N., Lee, J.Y. et al. Sphincter Contractility After Muscle-Derived Stem Cells Autograft into the Cryoinjured Anal Sphincters of Rats. Dis Colon Rectum 51, 1367–1373 (2008). https://doi.org/10.1007/s10350-008-9360-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10350-008-9360-y