Abstract
Supplementary feeding is a commonly used wildlife management practice, but while it may bring benefits and fulfil management goals, it can also result in unintended negative consequences. In the temperate zone, winter supplementary feeding may reduce movement of some ungulate species, leading to increased utilization of feeding sites by individuals and, consequently, animal aggregation. However, the intensity of feeding site utilization can vary depending on various extrinsic factors, e.g. habitat type, population density or weather conditions. Here, we predicted that the index of feeding sites utilization, expressed as the distance of European bison (Bison bonasus) to feeding sites, would be positively associated with the severity of winter conditions; thus, bison will be closer to feeding sites on colder days and in the presence of snow cover. We analysed winter (December to March) tracking data of 43 VHF- and GPS-collared European bison (24 males and 19 females) collected from 2005 to 2012 in Białowieża Primeval Forest (NE Poland), where bison are supplementally fed with different intensity throughout winter. Female bison were closer to feeding sites than males throughout winter, and regardless of sex, bison were the closest to feeding sites in mid-winter (January to February) and on colder days independently of the time of the season. Additionally, the distances of bison to feeding sites were significantly related to snow cover and depth; i.e. bison were closer to the feeding sites on days with present snow cover and deeper snow. Hence, the winter area occupied by bison differed with changing weather severity — being 4 and 28 times larger in the warmest periods compared to the coldest days with snow cover (for females and males, respectively). This may have direct and indirect ecological consequences for the ecosystem due to potential impact on nutrient cycling, seed dispersal, interspecific competition, vegetation growth, forest succession and carrion distribution. Given these ecological impacts of bison and weather-dependent utilization of supplementary fodder, we recognize the possible need in the future to revise and adapt winter supplementary feeding to annual and seasonal variation in winter severity to meet management goals while optimizing the costs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Humans provide forage to ungulates both intentionally, by delivering it to feeding sites, and unintentionally, by leaving food leftovers at compost piles and rubbish dumps (Smith 2001; Sorensen et al. 2014) or not protecting agricultural crops (Oro et al. 2013; Milner et al. 2014). Winter supplementary feeding, which involves providing forage to feeding sites during winter, is a widely practiced management approach for ungulates in temperate ecosystems of Europe and North America (Milner et al. 2014). In temperate latitudes, during winter, ungulates are exposed to higher thermoregulatory and mobility costs due to harsh weather conditions expressed in low ambient temperature and snow cover (Jensen et al. 1999; Arnold et al. 2006; Signer et al. 2011; Ossi et al. 2015). However, since supplementary fodder is an alternative and easily accessible food resource in winter, it allows ungulates to maintain high body condition, thereby enhancing their reproduction, survival (Krasińska and Krasiński 2013; Peterson and Messmer 2007) and trophy quality (Putman and Staines 2004; Cooper et al. 2006). Nevertheless, winter supplementary feeding of ungulates is also implemented for various other management goals. For instance, diversionary feeding aims at modification of ungulate movement and spatial behaviour to decrease their impact on forest and agricultural crops (van Beest et al. 2010a; Smith et al. 2001; Náhlik et al. 2005; Katona et al. 2014; Krasińska and Krasiński 2013; Jaroszewicz et al. 2017), limit ungulate-vehicle collisions (Wood and Wolfe 1988; Andreassen et al. 2005) or prevent disease transmission between livestock and wildlife (Brook 2008; Sorensen et al. 2014).
Undeniably, supplementary and diversionary feeding practices demontrated various benefits (e.g. reduced number of mule deer Odocoileus hemionus collisions along highways in Utah, USA (Wood and Wolfe 1988); increased winter survival (Krasińska and Krasiński 2013)), however, these practices have often failed to achieve their intended management goals (Cooper et al. 2006; van Beest et al. 2010b; Kowalczyk et al. 2011; Felton et al. 2017). Moreover, supplementary feeding can lead to unintended outcomes, such as aggregation around feeding sites, which may increase probability of intra- and interspecific disease and parasite transmission (Sorensen et al. 2014; Kołodziej-Sobocińska et al. 2016b), elevate impact of ungulates on vegetation composition and structure near feeding sites (Dunkley and Cattet 2003; Côté et al. 2004; Putman and Staines 2004; Mathisen and Skarpe 2011) and increase stress level caused by competitive feeding behaviour (Ceacero et al. 2012). Furthermore, by consuming supplementary forage prepared from sources from outside of the targeted area (i.e. hay), wild herbivores can contribute to seed dispersal of non-native vegetation seeds (Jaroszewicz et al. 2017).
Although in many places supplementary feeding was continuously delivered to feeding sites during periods of natural forage shortage, their utilization exhibited high spatio-temporal variation. Ungulates were more prone to visit feeding sites that provided high-quality fodder (Rajský et al. 2008) or those that have been present in the environment for longer duration and they were familiar with (Gundersen et al. 2004; Ranc et al. 2021). In addition, the visitation frequency to feeding sites by ungulates in winter was also positively associated with the severity of weather conditions. However, the effect of weather conditions on feeding sites visitation was mostly pronounced in small-bodied species or in young which body condition and survival are strongly dependent on weather conditions in winter (Gaillard et al. 1993; Loison and Langvatn 1998). For example, roe deer (Capreolus capreolus) used the supplemental feeding sites during harsh environmental conditions (Ossi et al. 2017; Ranc et al. 2021) and decreased utilization of artificial fodder in the time of spring green-up (Ossi et al. 2020), whereas red deer (Cervus elaphus) intensified the intake of supplementary fodder as soon as snow appeared (Náhlik et al. 2005).
Even though the factors affecting winter supplementary feeding in various ungulate species have been broadly studied (Milner et al. 2014), there has been no studies on this problem for European bison (Bison bonasus) which future existence depends on adequate management actions. With our study, we tried to bridge this gap, and additionally, we wanted to highlight the issue of wildlife supplementary feeding in the context of ongoing climate change by studying the winter space use of European bison that are supplementally fed in Białowieża Forest (NE Poland). This is especially important because weather conditions have undergone substantial changes in recent decade due to global warming. Increasing temperature in winter has caused elongation of the growing season and prolonged availability of natural forage for ungulates from northern latitudes (Oechel and Billings 1992).
European bison were reintroduced to Białowieża Forest in 1953 after their extinction in the wild in 1919 (Kowalczyk et al. 2011, 2013). The conservation plan recommended winter supplementary feeding as one of the practices that aimed at maintaining body condition (Kowalczyk et al. 2013) and mitigating conflicts with humans by decreasing the damage made by bison to tree stands and farm crops (Krasińska and Krasiński 2013; Jaroszewicz et al. 2017). Hence, European bison in Białowieża Forest have been supplementally fed at feeding sites throughout the winter period (December-March). We expect this management practice to have a significant effect on the winter space use of bison since the majority of the Białowieża Forest population utilizes these feeding sites during winter (Krasińska and Krasiński 2013). However, the utilization of feeding sites by bison should be associated with the variation in winter conditions. We predicted that (1) bison would concentrate their space use at feeding sites in the middle of the winter season (January and February) when natural forage is remarkably limited and weather conditions are the most severe (low temperatures, presence of a snow cover); (2) independently of the day of winter, bison will be closer to feeding sites on colder days with snow cover due to higher energy requirements and decreased natural food accessibility. However, we expected that (3) the effect of harsh winter conditions should be more pronounced in males than in females. Since females aggregate in large herds in winter, they require abundant food resources, therefore, they should be strongly dependent on utilization of feeding sites regardless of weather conditions. In turn, male utilization of feeding sites should be more weather-dependent because bulls roam solitary or in small groups and, during mild weather, they can utilize less abundant and dispersed food resources (small hay stacks, meadows, anthropogenic food).
Materials and methods
Study area
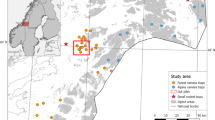
The study was conducted in the Polish part of the Białowieża Primeval Forest (BPF, 625 km2) located in north-eastern Poland (Fig. 1). BPF is one of the best-preserved temperate forests in Europe (Melis et al. 2007) and one of the very few forests on the European Plain where fragments of close to primeval forest remained (Sabatini et al. 2018; Jaroszewicz et al. 2019). BPF is covered by a mosaic of forest communities (coniferous, fresh and wet deciduous forest, and early successional stands) (Jędrzejewska and Jędrzejewski, 1998) dominated by Norwegian spruce Picea abies (25%), black alder Alnus glutinosa (21%), Scots pine Pinus sylvestris (19%), European hornbeam Carpinus betulus (11%), birch Betula sp. (7%), oaks Quercus sp. (6%) and small-leaved lime Tilia cordata (5%) (Modzelewska et al. 2020). Open habitats (6% of BPF area) include river valleys, forest meadows and glades with human settlements (Fig. 1; Sokołowski 2004; Kowalczyk et al. 2013, 2021). BPF is inhabited by five ungulate species, roe deer, red deer (Cervus elaphus), moose (Alces alces), European bison and wild boar (Sus scrofa), as well as two large predators: grey wolf (Canis lupus) and Eurasian lynx (Lynx lynx). The most abundant species is red deer, while other ungulates occur at lower densities (Borowik et al. 2013; Bubnicki et al. 2019), and the estimated number of European bison in the BPF in 2020 was 715 individuals (Raczyński 2021). European bison in the BPF are supplementally fed during winter (from December to March; Krasińska and Krasiński 2013), which leads to their aggregation around feeding sites (up to 100 individuals; Hayward et al. 2011; Kołodziej-Sobocińska et al. 2016a). Supplementary fodder primarily consists of hay, hay silage and occasionally beetroots (Kowalczyk et al. 2011). The hay for European bison is brought to feeding sites in summer and stored in ruffed haystacks (hay stacked around a tall, central pole) with free access to bison. The hay is usually consumed by bison between November and January, and supplemented by managers until March with different frequency, depending on the location. In the centre of the forest, where the largest herds of bison occur, hay is provided 3–5 times a week while in more distant locations on the edges of the forest usually once or twice a week (Radwan et al. 2010).
The climate in the BPF is transitional between continental and Atlantic types, with clearly marked warm and cold seasons. During the study period (2005–2012), the temperature ranged from –24.4 to 10.6 °C, and for 174 (47%) days, the temperature was below 0 °C. The snow persisted per 139 days a year on average, and its depth varied between 0.01 and 0.55 m. (Czarnecki et al. 2020; IMGW 2021, accessed via “climate” package).
Data collection and processing
In the study, we used tracking data of 43 European bison (24 males and 19 females) equipped with GPS or VHF collars during winter season (from 1 December to 31 March) between 2005 and 2012. Bison were immobilized from a car or hides with a dart gun (Dan-Inject) and etorphine (Arnemo 2003).
For the tracking data of European bison in BPF, we had to consider that 1) tracking duration of each bison was different; 2) during the same time period, both GPS and VHF collars were used to track different bison; and 3) for single individuals, the usage of collars during tracking period was mixed (i.e. only VHF or GPS collar used, transition either from VHF to GPS or from GPS to VHF). Therefore, bison locations (even for the same individuals) were collected with different intensity (VHF: from 1 to 12 locations per day; GPS: 24 locations per day). Moreover, VHF telemetry was conducted only during daylight hours, while GPS tracking was done 24 hours a day. To correct for possible circadian difference in the use of feeding sites by bison, we extracted locations fixed exclusively during daylight hours (between 08:00 am and 04:00 pm) — the period for which both VHF and GPS locations were available (19,906 locations: December to March, 2005–2012). Next, to unify the number of locations collected daily by GPS and VHF collars, for each bison and day we extracted one random location from all locations available for considered daylight hours (5386 bison locations in total).
Then, we georeferenced all bison winter feeding sites (n = 38) in Białowieża Forest and calculated distances between each bison location and the closest feeding site in QGIS software ver. 3.4.15 (QGIS Development Team 2021) (Fig. 1); thus, the number of distances was equal to the number of locations (n = 5386). Since part of the studied individuals could have been members of the same winter herds, their locations could be spatially autocorrelated. To deal with this problem, we decided to perform our analyses on population rather than individual level. For each unique day in the data, we calculated the mean and 95% quantile of available distances, separately for sexes. The calculations of mean and 95% quantile were restricted to days when at least 5 distances of bison to the nearest feeding site were available (range: from 1 to 13 distances per day) for a given sex (males: n = 107 days; females: n = 342 days; in total: n = 449).
Statistical analyses
To verify the association between considered explanatory variables and the mean distance of bison to feeding sites in winter, we fitted a generalized additive model (GAM1) in the R package “mgcv” (Wood 2011). We chose the GAM framework because for different predictors, we expected either linear or non-linear relationship with a dependent variable and GAM allows for flexible testing of these two types of relationships (Wood 2011). We used gamma family with a logarithmic link to conform to the distribution of continuous response variable: mean bison distance to winter feeding sites (n = 449). In GAM1, we included the main effect of the following explanatory variables: day of winter (continuous variable; from 1 December to 31 March for each considered winter period), presence of snow cover (categorical variable; 1, snow present; 0, snow absent), residuals of mean daily temperature (continuous variable) and sex (categorical variable). We also considered interactive effects between day of winter and sex and between residuals of mean daily temperature and sex. As the correlation between considered explanatory variables was moderate (R < 0.49), all variables were retained in the analyses.
Because we expected the day of winter to be nonlinearly related to mean bison distance to feeding sites, we fitted this variable with a spline (smoothing function), while other explanatory variables were fitted as linear (parametric) predictors. We expected limited amount of curvilinearity in the relationship between the day of winter and mean distance of bison to feeding sites; thus, we considered models using splines with a low number of knots (from 3 to 5) and compared them with AIC. The model with knot number equalling 4 had the lowest AIC values and this model was chosen as the best one. As the majority of the variation in mean daily temperature is expressed by seasonal dynamics (higher temperatures in the beginning and the end of winter and lowest in the middle of winter), we removed the seasonal component from the mean daily temperature by extracting residuals from the second-order polynomial regression model testing the relationship between the day of winter and mean daily temperature (LeBreton et al. 2013; García et al. 2020). Extracted residuals were then used as a variable in GAM1 as a residual mean daily temperature. This way we mitigated non-linear collinearity between the day of winter and mean daily temperature and obtained new interpretation of the mean daily temperature. Therefore, we could explain whether higher or lower temperature than expected on any day, based on predictions from the second-order polynomial model, related to the mean distance of bison to the feeding sites. Furthermore, we added the number of bison distances to closest feeding site in each unique day (range: from 5 to 13) as a weight to the GAM1, to give higher importance for mean bison distances which were calculated based on a larger sample size. Due to limited number of location in the majority of the studied winter seasons, we were not able to test the inter-seasonal variation in the mean bison distance to the feeding sites; thus, we added winter season (2004/2005–2011/2012) as a categorical random factor (penalized regression term) to GAM1 (Wood 2011).
Next, we fitted a generalized additive model with gamma error structure (GAM2) with 95% quantile of bison distance to feeding sites as a response variable to predict maximum distance of male and female bison to feeding sites in each considered month (December to March) under warm and cold scenarios. To fit GAM2, we used the same variables and model settings as in GAM1. To predict 95% quantile of bison distance to feeding sites, we used estimates of the GAM2 parameters. Then, we made predictions of 95% quantile for the 15th day of each month (from December till March; 15th, 46th, 76th and 105th day of winter respectively). In (1) warm scenario, the mean daily temperature was set to the maximum residual of mean daily temperature that was registered in a given month and for snow absent, whereas in (2) cold scenario, we used the minimum residual of mean daily temperature in respective month and presence of snow. Predictions for both scenarios were made separately for males and females and averaged over winter seasons. Predicted distances were used as radii for drawing buffers around feeding sites in the study area. Finally, we calculated the area covered by merged buffers to estimate maximum area utilized by male and female European bison in BPF under warm and cold conditions.
Finally, we investigated the relationship between mean bison distance to feeding sites and snow depth. From the dataset used in GAM1-2, we extracted days for which snow depth was at least 1 cm (n = 243), and for this dataset, we fitted a generalized additive model with gamma error structure (GAM3). We set the mean distance of bison to feeding sites as response variables and main and interactive effect of snow depth (continuous variable) and sex (categorical variable) as explanatory (parametric) predictors. Winter season was fitted as a random factor.
To evaluate the assumptions of GAM1-3, we inspected plots presenting distribution of models (deviance) residuals against the fitted values. We did not find any pattern in residual distribution; thus, we considered our models to be fitted correctly. All statistical analyses were done in R ver 4.0.5 (R Core Team 2021).
Results
The mean distance of bison to the feeding sites during daytime hours (between 08:00 am and 04:00 pm) (± SD) ranged from 0.03 ± 0.34 to 2.33 ± 0.34 km for females and from 0.18 ± 0.45 to 2.17 ± 0.45 km for males (Fig. 1S, Appendix 1). For both sexes, mean distance to feeding sites varied throughout the season (GAM1; Fig. 2; Table 1, females: P < 0.001; males: P < 0.05) in a non-linear pattern (GAM1; Table 1). Females were the closest to the feeding sites in the middle of winter, while at the beginning and the end of winter they roamed further from the feeding sites (Fig. 2A). For example, on the 76th day of winter (February 15), females were on average 90 m closer to the feeding sites than on the 15th day of winter (December 15) and 210 m closer than on the 105th day of winter (March 15). Males were the farthest from the feeding sites at the beginning of winter and approached feeding sites in the middle of winter (Fig. 2B). For example, on the 15th day of winter (December 15), males were on average 290 m farther from the feeding sites than on the 76th day of winter (February 15) and 360 m farther away than on the 105th day of winter (March 15). Furthermore, independently of the day of winter, distance of male and female bison from the feeding sites increased with increasing corrected mean daily temperature (GAM1; Table 1; Fig. 3; P = 0.04). With corrected mean daily temperature decreasing by 5ºC, the distance to the feeding sites of females decreased on average by 100 m, while for males, the distance to the feeding sites decreased by 140 m. Bison distances to feeding sites were negatively related to snow presence (GAM1, Fig. 3, Table 1, P < 0.001). Bison were by 120 m closer to the feeding sites when snow was present than in snowless conditions (Fig. 3).
The predictions of GAM2 indicated that 95% quantile of bison distance to feeding sites and thus, the area of Białowieża Primeval Forest occupied by bison, differed with weather conditions. The area used by European bison decreased on days when residual mean daily temperature was low and snow present (i.e. cold conditions) (Table 2). In cold conditions, males utilized larger area than females only at the beginning of winter (Table 2), while in mild weather conditions (snow absent and the highest residual mean daily temperature for this month), males utilized larger area than females throughout the whole winter (Table 2). In January, for mild weather conditions, female and male bison were predicted to utilize, respectively, more than 4 and 28 times larger area than under cold conditions (snow present and the lowest residual mean daily temperature) (Table 2; Fig. 4).
The distribution of feeding sites in Białowieża Primeval Forest with marked buffers representing the maximum area utilized by male and female European bison (Bison bonasus) under the (A) cold and (B) warm weather conditions in a given month. For more details, see the “Statistical analyses” section
Mean distance of bison to the feeding sites was significantly negatively associated with snow depth (GAM3, Table 1, P < 0.001). Males responded to increasing snow depth more strongly than females (GAM3, Fig. 5, Table 1, P < 0.05). With snow depth increasing by 5 cm, the distance to feeding sites of males decreased on average by 65 m while for females by 20 m.
Discussion
In our work, we examined the space use of European bison around winter feeding sites in Białowieża Primeval Forest under contrasting weather conditions. According to our analyses, bison were the closest to feeding sites in the middle of winter (January and February) when weather conditions were most severe and natural food resources remarkably limited. Females roamed farther from the feeding sites, mainly at the end of winter (March) when some herbaceous vegetation starts to appear in open habitats. Males approached feeding sites later (late January/early February) and remained in the vicinity of feeding sites throughout March. For females, such behaviour contradicts the previous findings which showed that ungulates kept utilization of the feeding sites even when natural vegetation is already present in spring (e.g. red deer (Bradesco 2012)). Moreover, we found that temperature affected bison space use independently of the time of season, with bison being closer to the feeding sites on days when daily temperature was below the expected for a given day of winter. Additionally, bison were also closer to the feeding sites on days when snow cover was present, especially when snow was deep. Our results showed that utilization of feeding sites by males was more related to harsh winter conditions than in females.
The severity of weather conditions affects the space use and thus, home range size of many ungulate species (Grignolio et al. 2004; Morellet et al. 2013; Borowik et al. 2020). Animals exposed to low temperature in winter reduce their activity to limit loss of energy needed for thermoregulation (white-tailed deer Odocoileus virginianus: Taillon et al. 2006; wild boar: Brivio et al. 2017). In addition, the presence of snow cover impedes animal mobility and makes movements costly in terms of energetic expenditures (Fox et al. 1992). These costs increase with increasing snow depth, as observed, for example, in mule deer (Odocoileus hemionus), elk (Cervus elaphus nelson; Parker et al. 1984), barren ground caribou (Rangifer tarandus granti, Fancy and White 1987) and roe deer (Hewison et al. 1998). Snow accumulation during winter may also limit access to vegetation, thereby increasing the amount of time required to encounter a food item (Robinson and Merrill 2012). To counteract the costs associated with severe weather conditions, ungulates utilize supplementary fodder when available, and concentrate their activity around feeding sites, which is reflected in reduced mobility and home range sizes (Guillet et al. 1996; van Beest et al. 2010a). This is especially true for European bison, which are grazers, unlike other ungulate species occurring in the same ecosystem, and their natural forage in winter consists in majority of dry grasses that are often unavailable due to thick snow cover (Krasińska and Krasiński 2013).
We also indicated the sex-specific differences in the effect of weather condition and day of winter on the distance of bison to feeding sites. Females were closer to feeding sites for the most of winter, which may suggest that females utilized feeding sites more often than males, while males approached feedings sites mainly during adverse weather conditions. These differences can be caused by sex-segregation observed in bison during this time of the year, which is associated with a high dimorphism in body size of European bison (Krasińska and Krasiński 2002). According to activity budget hypothesis, larger males in dimorphic ungulates should spend less time foraging and spent more time walking than females (Ruckstuhl and Neuhaus 2002). Therefore, European bison males in winter are usually solitary or move in small male groups while females with calves create large aggregations (Krasińska and Krasiński 1995). Moreover, males can find sufficient food supplies also outside the feeding sites (e.g. natural vegetation in forest glades or river valleys), while female herds, due to high forage requirements, are forced to utilize feeding sites or migrate outside the forest to feed on winter crops (Hofman-Kamińska and Kowalczyk 2012; Kowalczyk et al. 2013).
Although we did not use direct measure of the utilization of feeding sites by bison, and the efficiency of applied index (i.e. distance of bison to feeding sites) in describing supplementary forage use by bison remained unknown, we claim that the different distances of bison to the feeding sites in winter are expressing the variation in feeding site utilization. We did not know any other factor associated with studied feeding sites, or surrounding area, that could additionally affect differences in bison proximity to the feeding sites. However, it would be beneficial to confirm the observed relationships by the results of the studies made on direct measures of supplementary fodder use, as well as supplementing those analyses with the variables describing the quantity and quality of consumed supplementary forage and spatio-temporal variation in natural food supply.
In the presence of fast-occurring climate change and global warming, we expect a further decrease in winter severity; thus, its limiting effect on the mobility of ungulates will no longer occur at such scale and intensity as in the past. Increasing winter temperature and shorter persistence of snow cover will cause earlier vegetation green-up and ultimately, significant changes in the time of natural forage availability to herbivores (Barichivich et al. 2013; Keenan et al. 2014). Milder winters may also reduce changes in body condition and lower energetic requirements of animals, thus increasing independence from fodder supplementation. In the case of European bison, we can expect a limited intake of supplementary fodder and a larger area of the BPF utilized by bison during milder weather conditions. Such changes can have a diverse impact on the ecosystem functioning through their influence on nutrient cycling (Melis et al. 2007; Jaroszewicz et al. 2013), vegetation growth, forest succession (Kowalczyk et al. 2021) and the availability and distribution of carrion (Selva et al. 2003). Furthermore, increased bison mobility seems to be also favourable because smaller bison aggregations may decrease parasite and disease transmission (Kołodziej-Sobocińska 2016a, b) and diffuse browsing from feeding sites areas (Jaroszewicz et al. 2017). On the other hand, greater mobility and utilization of larger areas by bison during milder winter may also have negative impacts. For instance, bison may disperse non-forest plant species consumed with hay that is delivered to feeding sites which in consequence, may lead to changes in the composition of natural vegetation and plant diversity (Jaroszewicz et al. 2008, 2013). Greater mobility also increases probability of bison moving more frequently outside of BPF into farmland areas surrounding the forest, causing conflicts with farmers through damaging crops.
To summarize, our results showed that the utilization of feeding sites by bison was associated with winter weather conditions. Therefore, there is a high probability that ongoing global warming will cause further changes in supplementary forage use by European bison, which may have far-reaching ecological consequences for the ecosystem of the BPF. However, to address appropriately the problem of supplementary feeding under changing weather conditions, we urgently need further studies which will more precisely evaluate optimal supplementary feeding strategies under different winter severity scenarios, allowing to meet management or conservation goals while optimizing the costs. This is especially important in case of European bison which was restored after extinction in the wild (Plumb et al. 2020), as its future depends on appropriate conservation practices aimed on both, preservation and counteracting the actual and potential threats, and ensuring that population functioning is shaped by natural processes as much as possible.
References
Andreassen HP, Gundersen H, Storaas T (2005) The effect of scent-marking, forest clearing, and supplemental feeding on moose-train collisions. J Wildl Manag 69(3):1125–1132
Arnemo JM, Kreeger TJ, Soverti T (2003) Chemical immobilization of free-ranging moose. Alces 39:243–253
Arnold W, Ruf T, Kuntz R (2006) Seasonal adjustment of energy budget in a large wild mammal, the Przewalski horse (Equus ferus przewalskii) II. Energy expenditure. J Exp Biol 209(22):4566–4573
Barichivich J, Briffa KR, Myneni RB, Osborn TJ, Melvin TM, Ciais P, Piao S, Tucker C (2013) Large-scale variations in the vegetation growing season and annual cycle of atmospheric CO2 at high northern latitudes from 1950 to 2011. Glob Change Biol 19:3167–3183. https://doi.org/10.1111/gcb.12283
Borowik T, Cornulier T, Jędrzejewska B (2013) Environmental factors shaping ungulate abundances in Poland. Acta Theriol 58(4):403–413. https://doi.org/10.1007/s13364-013-0153-x
Borowik T, Ratkiewicz M, Maślanko W, Duda N, Kowalczyk R (2020) Too hot to handle: summer space use shift in a cold-adapted ungulate at the edge of its range. Landscape Ecol 35(6):1341–1351. https://doi.org/10.1007/s10980-020-01018-4
Bradeško U (2012) Effects of supplemental feeding on annual, seasonal and circadian spatial distribution of red deer (Cervus elaphus L.) in Slovenia: graduation thesis - university studies, Ljubljana, VIII, 102 str., ilustr. https://repozitorij.uni-lj.si/IzpisGradiva.php?id=16128. [COBISS.SI-ID 3503526]
Brivio F, Grignolio S, Brogi R, Benazzi M, Bertolucci C, Apollonio M (2017) An analysis of intrinsic and extrinsic factors affecting the activity of a nocturnal species: the wild boar. Mamm Biol 84(1):73–81. https://doi.org/10.1016/j.mambio.2017.01.007
Brook RK (2008) Elk-agriculture conflicts in the greater Riding Mountain ecosystem: building bridges between the natural and social sciences to promote sustainability. Ph.D. dissertation, Department of Environment and Geography, The University of Manitoba, Winnipeg
Bubnicki JW, Churski M, Schmidt K, Diserens TA, Kuijper DP (2019) Linking spatial patterns of terrestrial herbivore community structure to trophic interactions. ELife 8:e44937. https://doi.org/10.7554/eLife.44937
Ceacero F, García AJ, Landete-Castillejos T, Bartošová J, Bartoš L, Gallego L (2012) Benefits for dominant red deer hinds under a competitive feeding system: food access behavior, diet and nutrient selection. PLoS ONE 7:e32780
Cooper SM, Owens MK, Cooper RM, Ginnett TF (2006) Effect of supplemental feeding on spatial distribution and browse utilization by white-tailed deer in semi-arid rangeland. J Arid Environ 66(4):716–726. https://doi.org/10.1016/j.jaridenv.2005.11.015
Côté SD, Rooney TP, Tremblay JP, Dussault C, Waller DM (2004) Ecological impacts of deer overabundance. Annual review of ecology, evolution, and systematics 113–147
Czarnecki B, Głogowski A, Nowosad J (2020) Climate: an R package to access free in-situ meteorological and hydrological datasets for environmental assessment. Sustainability 12:394. https://doi.org/10.3390/su12010394
Dunkley L, Cattet MRL (2003) A comprehensive review of the ecological and human social effects of artificial feeding and baiting of wildlife. Newsletters & Publications, Canadian Cooperative Wildlife Health Centre, p 21
Fancy SG, White RG (1987) Energy expenditures for locomotion by barren-ground caribou. Can J Zool 65:122–128. https://doi.org/10.1139/z87-018
Felton AM, Felton A, Cromsigt JP, Edenius L, Malmsten J, Wam HK (2017) Interactions between ungulates, forests, and supplementary feeding: the role of nutritional balancing in determining outcomes. Mammal Research 62(1):1–7. https://doi.org/10.1007/s13364-016-0301-1
Fox JL, Sinha SP, Chundawat RS (1992) Activity patterns and habitat use of ibex in the Himalayan Mountains of India. J Mammal 73(3):527–534. https://doi.org/10.2307/1382018
Gaillard JM, Delorme D, Boutin J-M, Van Laere G, Boisaubert B, Pradel R (1993) Roe deer survival patterns: a comparative analysis of contrasting populations. J Anim Ecol 62:778–791
García CB, Salmerón R, García C, García J (2020) Residualization: justification, properties and application. J Appl Stat 47(11):1990–2010
Grignolio S, Rossi I, Bassano B, Parrini F, Apollonio M (2004) Seasonal variations of spatial behaviour in female Alpine ibex (Capra ibex ibex) in relation to climatic conditions and age. Ethol Ecol Evol 16(3):255–264. https://doi.org/10.1080/08927014.2004.9522636
Guillet C, Bergström R, Cederlund G (1996) Size of winter home range of roe deer Capreolus capreolus in two forest areas with artificial feeding in Sweden. Wildl Biol 2(3):107–111. https://doi.org/10.2981/wlb.1996.039
Gundersen H, Andreassen HP, stein Storaas, T. (2004) Supplemental feeding of migratory moose Alces alces: forest damage at two spatial scales. Wildl Biol 10(1):213–223. https://doi.org/10.2981/wlb.2004.027
Hayward MW, Kowalczyk R, Krasiński ZA, Krasińska M, Dackiewicz J, Cornulier T (2011) Restoration and intensive management have no effect on evolutionary strategies. Endangered Species Research 15(1):53–61. https://doi.org/10.3354/esr00371
Hewison AM, Vincent JP, Reby D (1998) Social organisation of European roe deer. The biology of success, Scandinavian University Press, The European roe deer
Hofman-Kamińska E, Kowalczyk R (2012) Farm crops depredation by European bison (Bison bonasus) in the vicinity of forest habitats in northeastern Poland. Environ Manage 50(4):530–541
IMGW (2021) Meteorological data from year 2005–2012, available online: https://danepubliczne.imgw.pl/data/dane_pomiarowo_obserwacyjne/dane_meteorologiczne/. Accessed 24 Feb 2021
Jaroszewicz B (2013) Endozoochory by European bison influences the build-up of the soil seed bank in subcontinental coniferous forest. Eur J Forest Res 132:445–452. https://doi.org/10.1007/s10342-013-0683-4
Jaroszewicz B, Cholewińska O, Gutowski JM, Samojlik T, Zimny M, Latałowa M (2019) Białowieża forest—a relic of the high naturalness of European forests. Forests 10(10):849. https://doi.org/10.3390/f10100849
Jaroszewicz B, Kwiecień K, Czortek P, Olech W, Pirożnikow E (2017) Winter supplementary feeding influences forest soil seed banks and vegetation. Appl Veg Sci 20(4):683–691. https://doi.org/10.1111/avsc.12319
Jaroszewicz B, Pirożnikow E, Sagehorn R (2008) The European bison as seed dispersers: the effect on the species composition of a disturbed pine forest community. Botany 86(5):475–484. https://doi.org/10.1139/B08-012
Jędrzejewska B, Jędrzejewski W (1998) Predation in vertebrate communities. The Białowieza Primeval Forest as a case study. Springer, Berlin Heidelberg New York
Jensen PG, Pekins PJ, Holter JB (1999) Compensatory effect of the heat increment of feeding on thermoregulation costs of white-tailed deer fawns in winter. Can J Zool 77(9):1474–1485
Katona K, Gál-Bélteki A, Terhes A, Bartucz K, Szemethy L (2014) How important is supplementary feed in the winter diet of red deer? A Test in Hungary Wildlife Biology 20(6):326–334
Keenan T, Gray J, Friedl M et al (2014) Net carbon uptake has increased through warming-induced changes in temperate forest phenology. Nat Clim Chang 4:598–604. https://doi.org/10.1038/nclimate2253
Kołodziej-Sobocińska M, Demiaszkiewicz AW, Lachowicz J, Borowik T, Kowalczyk R (2016a) Influence of management and biological factors on parasitic invasions in the wild–spread of the blood-sucking nematode Ashworthius sidemi in European bison (Bison bonasus). International Journal for Parasitology: Parasites and Wildlife 5(3):286–294. https://doi.org/10.1016/j.ijppaw.2016.09.005
Kołodziej-Sobocińska M, Pyziel AM, Demiaszkiewicz AW et al (2016b) Pattern of parasite egg shedding by European bison (Bison bonasus) in the Białowieża Primeval Forest, Poland. Mammal Research 61:179–186. https://doi.org/10.1007/s13364-016-0270-4
Kowalczyk R, Kamiński T, Borowik T (2021) Do large herbivores maintain open habitats in temperate forests?. For Ecol Manag 494. https://doi.org/10.1016/j.foreco.2021.119310
Kowalczyk R, Krasińska M, Kamiński T, Górny M, Struś P, Hofman-Kamińska E, Krasiński ZA (2013) Movements of European bison (Bison bonasus) beyond the Białowieża Forest (NE Poland): range expansion or partial migrations? Acta Theriol 58(4):391–401. https://doi.org/10.1007/s13364-013-0136-y
Kowalczyk R, Taberlet P, Coissac E, Valentini A, Miquel C, Kamiński T, Wójcik JM (2011) Influence of management practices on large herbivore diet—case of European bison in Białowieża Primeval Forest (Poland). For Ecol Manage 261(4):821–828. https://doi.org/10.1016/j.foreco.2010.11.026
Krasińska M, Krasiński Z (1995) Bisoniana 112. Composition, group size, and spatial distribution of European bison bulls in Białowieża Forest. Acta Theriologica, 40(1), 1–21
Krasińska M, Krasiński ZA (2013) European bison. The Nature Monograph. Mammal Research Institute PAS, Białowieża
LeBreton JM, Tonidandel S, Krasikova DV (2013) Residualized relative importance analysis: a technique for the comprehensive decomposition of variance in higher order regression models. Organ Res Methods 16(3):449–473
Loison A, Langvatn R (1998) Short- and long-term effects of winter spring weather on growth and survival of red deer in Norway. Oecologia 116:489–500
Mathisen KM, Skarpe C (2011) Cascading effects of moose (Alces alces) management on birds. Ecol Res 26(3):563–574. https://doi.org/10.1007/s11284-011-0815-6
Melis C, Selva N, Teurlings IJM, Skarpe C, Linnell JDC, Andersen R (2007) Soil and vegetation nutrient response to bison carcasses in Bialowieza Primeval Forest. Poland Ecological Research 22(5):807–813. https://doi.org/10.1007/s11284-006-0321-4
Milner JM, Van Beest FM, Schmidt KT, Brook RK, Storaas T (2014) To feed or not to feed? Evidence of the intended and unintended effects of feeding wild ungulates. J Wildl Manag 78(8):1322–1334. https://doi.org/10.1002/jwmg.798
Modzelewska A, Fassnacht FE, Stereńczak K (2020) Tree species identification within an extensive forest area with diverse management regimes using airborne hyperspectral data. Int J Appl Earth Obs Geoinf 84. https://doi.org/10.1016/j.jag.2019.101960
Morellet N, Bonenfant C, Börger L, Ossi F, Cagnacci F, Heurich M, Kjellander P, Linnell JDC, Nicoloso S, Sustr P, Urbano F, Mysterud A (2013) Seasonality, weather and climate affect home range size in roe deer across a wide latitudinal gradient within Europe. J Anim Ecol 82:1326–1339. https://doi.org/10.1111/1365-2656.12105
Náhlik A, Borkowski J, Király G (2005) Factors affecting the winter-feeding ecology of red deer. Wildl Biol Pract 1(1):47–52
Oechel WC, Billings WD (1992) Effects of global change on the carbon balance of arctic plants and ecosystems. Arctic ecosystems in a changing climate: an ecophysiological perspective 139–168
Oro D, Genovart M, Tavecchia G, Fowler MS, Martínez-Abraín A (2013) Ecological and evolutionary implications of food subsidies from humans. Ecol Lett 16(12):1501–1514
Ossi F, Gaillard JM, Hebblewhite M, Cagnacci F (2015) Snow sinking depth and forest canopy drive winter resource selection more than supplemental feeding in an alpine population of roe deer. Eur J Wildl Res 61(1):111–124
Ossi F, Gaillard JM, Hebblewhite M, Morellet N, Ranc N, Sandfort R, Kroeschel M, Kjellander P, Mysterud A, Linnell JDC, Heurich M, Soennichsen L, Sustr P, Berger A, Rocca M, Urbano F, Cagnacci F (2017) Plastic response by a small cervid to supplemental feeding in winter across a wide environmental gradient. Ecosphere 8(1). https://doi.org/10.1002/ecs2.1629
Ossi F, Ranc N, Moorcroft P, Bonanni P, Cagnacci F (2020) Ecological and behavioral drivers of supplemental feeding use by roe deer Capreolus capreolus in a peri-urban context. Animals 10(11):2088
Parker KL, Robbins CT, Hanley TA (1984) Energy expenditures for locomotion by mule deer and elk. J Wildl Manag 48:474–488
Peterson C, Messmer TA (2007) Effects of winter-feeding on mule deer in northern Utah. J Wildl Manag 71(5):1440–1445
Plumb G, Kowalczyk R, Hernandez-Blanco JA (2020) Bison bonasus. The IUCN Red List of Threatened Species
Putman RJ, Staines BW (2004) Supplementary winter feeding of wild red deer Cervus elaphus in Europe and North America: justifications, feeding practice and effectiveness. Mammal Rev 34(4):285–306
QGIS Development Team (2021) QGIS Geographic Information System. Open Source Geospatial Foundation Project. https://qgis.osgeo.org
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 5 Apr 2022
Raczyński J (2020) European Bison Pedigree Book 2021. Białowieża National Park
Radwan J, Demiaszkiewicz AW, Kowalczyk R, Lachowicz J, Kawałko A, Wójcik JM, Pyziel MA, Babik W (2010) An evaluation of two potential risk factors, MHC diversity and host density, for infection by an invasive nematode Ashworthius sidemi in endangered European bison (Bison bonasus). Biol Cons 143(9):2049–2053. https://doi.org/10.1016/j.biocon.2010.05.012
Rajský M, Vodňanský M, Hell P, Slamečka J, Kropil R, Rajský D (2008) Influence supplementary feeding on bark browsing by red deer (Cervus elaphus) under experimental conditions. Eur J Wildl Res 54(4):701–708. https://doi.org/10.1007/s10344-008-0199-2
Ranc N, Moorcroft PR, Ossi F, Cagnacci F (2021) Experimental evidence of memory-based foraging decisions in a large wild mammal. Proc Natl Acad Sci 118(15):e2014856118
Robinson BG, Merrill EH (2012) The influence of snow on the functional response of grazing ungulates. Oikos 121(1):28–34. https://doi.org/10.1111/j.1600-0706.2011.19408.x
Ruckstuhl KE, Neuhaus P (2002) Sexual segregation in ungulates: a comparative test of three hypotheses. Biol Rev 77(1):77–96
Sabatini FM, Burrascano S, Keeton WS et al (2018) Where are Europe’s last primary forests? Divers Distrib 24(10):1426–1439. https://doi.org/10.1111/ddi.12778
Selva N, Jedrzejewska B, Jedrzejewski W, Wajrak A (2003) Scavenging on European bison carcasses in Bialowieza primeval forest (eastern Poland). Ecoscience 10(3):303–311. https://doi.org/10.1080/11956860.2003.11682778
Signer C, Ruf T, Arnold W (2011) Hypometabolism and basking: the strategies of Alpine ibex to endure harsh over-wintering conditions. Funct Ecol 25(3):537–547
Smith BL (2001) Winter feeding of elk in western North America. J Wildl Manag 65(2):173–190. https://doi.org/10.2307/3802896
Sokołowski AW (2004) Lasy Puszczy Białowieskiej [The Forests of the Białowieża Primeval Forest]. State Forests Information Centre, Warszawa [In Polish]
Sorensen A, van Beest FM, Brook RK (2014) Impacts of wildlife baiting and supplemental feeding on infectious disease transmission risk: a synthesis of knowledge. Prev Vet Med 113(4):356–363
Taillon J, Sauvé DG, CǑTÉ, S. D. (2006) The effects of decreasing winter diet quality on foraging behavior and life-history traits of white-tailed deer fawns. J Wildl Manag 70(5):1445–1454. https://doi.org/10.2193/0022-541X(2006)70[1445:TEODWD]2.0.CO;2
van Beest FM, Gundersen H, Mathisen KM, Milner JM, Skarpe C (2010a) Long-term browsing impact around diversionary feeding stations for moose in Southern Norway. For Ecol Manage 259(10):1900–1911. https://doi.org/10.1016/j.foreco.2010.02.002
van Beest FM, Loe LE, Mysterud A, Milner JM (2010b) Comparative space use and habitat selection of moose around feeding stations. J Wildl Manag 74(2):219–227. https://doi.org/10.2193/2009-109
Wood P, Wolfe ML (1988) Intercept feeding as a means of reducing deer-vehicle collisions. Wildlife Society Bulletin (1973–2006), 16(4), 376–380
Wood SN (2011) Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J R Stat Soc (b) 73(1):3–36. https://doi.org/10.1111/j.1467-9868.2010.00749.x
Acknowledgements
We are grateful to Dr. Michał Żmihorski and three anonymous reviewers whose remarks considerably improved the paper.
Funding
The study was financed by the Ministry of Science and Computerisation project no. 2P04F 011 26 and by the Frankfurt Zoological Society — Help for Threatened Wildlife and LIFE financial instrument of the European Community (project “Bison Land — European bison conservation in Białowieża Forest”, LIFE06 NAT/PL/000105 BISON-LAND).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bramorska, B., Kowalczyk, R., Kamiński, T. et al. Linking winter severity to space use of European bison around feeding sites in Białowieża Primeval Forest (NE Poland). Eur J Wildl Res 69, 66 (2023). https://doi.org/10.1007/s10344-023-01690-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-023-01690-2