Abstract
European wild rabbit (Oryctolagus cuniculus) and red-legged partridge (Alectoris rufa) are main small game species of conservation concern in Mediterranean ecosystems. To date, their presence in wild boar’s (Sus scrofa) diet and factors driving their consumption have been little investigated. A genetic metabarcoding approach was used on 80 wild boar faeces collected from four hunting estates devoted to small game hunting during two different seasons. Abundances of wild boar, rabbits and partridges were first estimated. Results showed DNA of seventeen chordate species. The highest frequency of occurrence (FO) corresponded to mammals and birds, with 77.6 and 22.3%, respectively. DNA of game species was detected in 39/71 (FO = 55.0%) samples, highlighting the presence of European wild rabbit in 27 (FO = 38.0%) and red-legged partridge in eight (FO = 11.3%). Dietary composition varied between seasons and estates, being rabbit the main responsible (explaining 35.26% and 39.45% of differences, respectively). Rabbit FO in the diet was positively related to the abundance of wild boar and rabbit density on the estate. It was greater in autumn and in estates where rabbits were hunted. Regarding red-legged partridge, a significant and positive relationship between its population density and its diet FO was observed, without significant differences between seasons or estates. Overall, our results suggest wild boar as an opportunistic species whose diet is largely determined by the relative availability of different food resources. Its ecological role concerning small game species in Mediterranean agroecosystems seems to be more related to consumption of carrion during the hunting season than to direct predation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The impact of land-use changes on game species has been the subject of increasing concern and, consequently, research (e.g. Sotherton 1998; Jiménez-García et al. 2006; Delibes-Mateos et al. 2009; Petersson et al. 2019). Agricultural intensification has been reported as a critical constraint for small game populations (e.g. Smith et al. 2004), which may explain the decline of small game species in some regions (Panek 1997; Buenestado et al. 2008). However, these species have suffered a marked decline (Edwards et al. 2000; Bro et al. 2001; Delibes-Mateos et al. 2008, 2009; Díaz-Fernández et al. 2012; Guerrero-Casado et al. 2016; Gée et al. 2018), while large game species have substantially increased their abundance and distribution (Herruzo and Martinez-Jauregui 2013; Carpio et al. 2021), especially in the case of wild boar (Sus scrofa; Massei et al. 2015; Wehr 2021).
Previous studies have shown a clear spatial segregation between small and large game species, where small game species are mainly linked to agroecosystems and large game species to woodlands (Vargas et al. 2007; Delibes-Mateos et al. 2009). However, the wild boar is colonising and spreading rapidly throughout agricultural areas (Hearn et al. 2014; Morelle et al. 2016) and urban and peri-urban scenarios (Castillo-Contreras et al. 2018); they are now present in quasi-entire countries in the Northern Hemisphere (e.g. Bencatel et al. 2019). In agroecosystems, hunting traditionally targets small game species (i.e. rabbits, hares, partridges). Consequently, the return of wild boar to agroecosystems is not considered positive, and this species is therefore mostly hunted to reduce crop damage (e.g. Morelle et al. 2016) and avoid predation on small game species (e.g. Carpio et al. 2014a). In this context, previous studies have shown a negative relationship between wild boar and population abundances of small game species, which was explained either by competition or direct predation (Carpio et al. 2014a, 2014b; Barros et al. 2020). However, most of these studies have been carried out in forested areas devoted to large game species, where the abundance of small game species is low and wild boar reaches a high population abundance (Virgós et al. 2011). Nevertheless, little is known about the relevance of small game species in the diet of wild boar and the factors driving their consumption in newly colonised agroecosystems.
In the specific case of the red-legged partridge (Alectoris rufa) and other birds that nest on the ground, previous studies have shown the wild boar as an important predator of nests (Carpio et al. 2014a; Oja et al. 2017; Mori et al. 2021). Furthermore, when dead birds become available seasonally, such as by releasing farm-bred partridges in autumn to increase game yield (Alonso et al. 2005), which usually experience a high mortality rate (Gortázar et al. 2000; Díaz-Fernández et al. 2012), wild boar scavenging on bird carrion has been reported (Cellina 2008; Ballari et al. 2015). In the case of European wild rabbits (Oryctolagus cuniculus), the potential effect of wild boar as a predator is little known (see a review by Schley and Roper 2003), although wild boar could depredate young or diseased rabbits (Virgós et al. 2011). In this respect, Herrero et al. (2006) found that vertebrates only represent 1.3% of the volume in the diet of wild boar in agroecosystems in north-eastern Spain, but with a frequency of occurrence of 48.2% (depicting mammals and birds 0.96% and 0.15% of volume and 27% and 16% of frequency, respectively). Similarly, Schley and Roper (2003) reported a frequency of the occurrence of animal matter (including hunted species such as Alectoris rufa, Columba palumbus, Phasianus colchicus, Turdus sp., Lepus europaeus, Oryctolagus cuniculus and Cervus elaphus) of between 47 and 88% in the agroecosystems of Western Europe. However, the role of factors that drive the consumption of small game species in agroecosystems (e.g. hunting, densities) is mostly unknown (Wilcox and Van Vuren 2009; Tobajas et al. 2021a, 2021b).
Genetic tools allow considerably more precise taxonomic identification of the animals/plants consumed compared to morphological analyses of animal remains (Oja et al. 2017). Robeson et al. (2018) assessed the utility of metabarcoding, showing that this technique is revolutionising diet studies by adding sensitivity to species identification (de Sousa et al. 2019), even though this method does not allow us to distinguish direct predation from carrion consumption (Wilcox and van Vuren 2009; Oja et al. 2017). Therefore, the aim of this study is to assess the factors driving the consumption of small game species by wild boar in agroecosystems. Specifically, the objectives were as follows: (i) to characterise the chordate communities in the diet of wild boar, (ii) to determine the influence of the season and hunting pressure on the spatiotemporal changes in the composition of the chordate communities in the diet and (iii) to evaluate the potential effect of hunting on the frequency of occurrence of wild rabbit and red-legged partridge in the diet as the main small game species in the study area.
Materials and methods
Study area and period
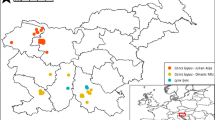
The present study was carried out on four hunting estates (two where rabbits are hunted and two where they are not) in South-Central Spain during the early autumn of 2020 and early spring of 2021 (Fig. 1). This region has a Mediterranean climate with a continental influence, which means that it has a very warm and dry summer (from June to September, but with the considerable annual variations typical of the Mediterranean climate). The habitat is characterised by different land uses and management scenarios, with small patches of evergreen oak forests dominated by Quercus sp. and scrublands (Cystus sp., Pistacia sp., Rosmarinus sp., Erica sp. and Phyllirea sp.) alternating with rainfed crops (especially cereal, sunflowers, or legumes), woody crops (olive groves, vineyards, or almonds) and with scattered pastures that form dehesas (Joffre et al. 1999) (Supplementary Information).
European wild rabbit, red-legged partridge and wild boar are present at all the study sites. These estates are traditionally dedicated to the hunting of small game species with management characterised by the supply of water and food (especially in summer) mainly for rabbits and red-legged partridges and selective control of predators, mainly red fox (Vulpes vulpes) and magpie (Pica pica). The hunting season extends from October to January. However, estates A and B are within the Iberian lynx project, and so the rabbit is not hunted in these areas (https://www.castillalamancha.es/sites/default/files/documentos/20120511/linceiberico.pdf), while the rabbit is hunted on estates C and D.
Estimating wild rabbit and red-legged partridge population densities
Rabbit and red-legged partridge population densities were estimated on each hunting estate. Each survey occurred on three consecutive days, in clear weather conditions, and avoiding days when hunting was permitted (Barrio et al. 2010). The estimates were performed in two seasons (early autumn (between 15/09/2020 and 05/10/2020) and early spring (09/03/2021 and 18/04/2021)) on each hunting estate by a driver and an observer inside a vehicle, travelling at 10 − 15 km/h, at sunrise (between 7.00 and 10.00 a.m.). Rabbits are active during twilight and at night, with two activity peaks that coincide with sunrise and sunset (Díez et al. 2005). In the same way, red-legged partridges are also active during the first three hours after dawn (Borralho et al. 1996). Each transect was an average of 16.76 km ± 1.95 (SE) in length. The distance from the observer to the animals was measured using a telemeter (range 15 − 1100 m; precision ± 1 m ± 0.1%), and compass bearings were taken to determine the angle between the animals and the transect line.
Rabbit and red-legged partridge densities were estimated using the distance sampling method (Buckland 2004) with Distance 7.3 software (Thomas et al. 2010). Two models per species were run (one for spring and one for autumn), and the analyses were stratified by hunting estate to estimate density. Briefly, half-normal, uniform and hazard rate models for the detection function were fitted against the data using cosine, Hermite polynomial and simple polynomial adjustment terms, which were fitted sequentially. The selection of the best model and adjustment term was based on Akaike’s information criterion (AIC) (Akaike 1974).
Estimation of relative wild boar abundance and the collection of faeces for diet analyses
Wild boar relative population abundance was estimated based on the frequency of faecal dropping (FBII) on a walked transect index (Acevedo et al. 2007). Three transects of 4 km per hunting estate were performed on the same dates as the rabbit and red-legged partridge estimations. Each transect count consisted of 40 segments of 100 m in length and 1 m in width; each segment was divided into 10 sectors of 10 m in length. Sign frequency, as a relative abundance index, was defined as the average number of 10-m sectors containing droppings per 100-m transect and was calculated per estate and season. We also obtained an FBII by counting carnivore scats in the abovementioned three 4-km-long transects.
The search and collection of fresh wild boar excrements were carried out along transects (Ebert et al. 2012). An active search was also performed in bedding sites to collect fresh faecal droppings (one per bed to ensure the independence of data) in each estate per season (i.e. 20 faeces per hunting estate, approximately). Faeces were put into plastic bags and stored at − 80 °C before analysis to prevent further degradation of DNA (Oja et al. 2017).
Molecular analyses
DNA was extracted from each faecal sample using the E.Z.N.A tissue DNA kit (Omega Bio-Tek, GA, USA), following the manufacturer’s protocol but employing an additional initial digestion step with a lysis wash buffer as described by Maudet et al. (2004). DNA extractions were performed in a room exclusively dedicated to this purpose to reduce the risk of DNA contamination. Negative controls were also added in each manipulation to monitor DNA contamination. The diet was analysed using the previously published primers MICOlintF (5′-GGWACWGGWTGAACWGTWTAYCCYCC-3′) (Leray et al. 2013) and PolyShortCoiR (5′-CCNCCTCCNGCWGGRTCRAARAA-3′) (Carr et al. 2011). These primers amplified a ~ 200 − 300 bp fragment of the mitochondrial-encoded cytochrome oxidase subunit I (COI) of metazoan taxa (Robeson et al. 2018). Primers were modified to contain Illumina adaptors at the 5′ end of the sequence (forward primers: 5′‐TCGTCGGCAGCGTCAGATGTGTATAAGAGACAG‐3′, reverse primers: 5′‐GTCTCGTGGGCTCGGAGATGTGTATAAGAGACAG‐3′). Due to the lack of specificity of the Sus scrofa CO1 near the amplification site, a dual priming oligomer 5′-ACCCACCTTTAGCTGGAAACTTAGCCCATGCAGGAGCTTCAGTTGATCTAACAAIIIICTCCCTACACCTT-C3-3′ (Robeson et al. 2018) was employed to block host sequence amplification. A 10:1 ratio of blocking primers to amplification primers was used as suggested by Robeson et al. (2018). PCR reactions were carried out in volumes of 10 μl, comprising 5 μl of QIAGEN Multiplex PCR Master Mix, 0.3 μl of 10 mM MICOlintF and PolyShortCoiR primers, 0.3 μl of 100 nM blocking primer, 3.1 μl of ultra‐pure water and 1 μl of DNA extract. The thermocycling program used initial denaturing at 95 °C for 15 min, followed by 35 cycles of denaturing at 95 °C for 30 s, annealing at 55 °C for 30 s and extension at 72 °C for 30 s, with a final extension at 72 °C for 5 min. Three replicates of each PCR reaction were performed. Amplification success was confirmed by visually inspecting 2 μl of each PCR product on a 2% gel-stained agarose. A second-round PCR was performed to attach the unique dual P5 and P7 indexes selected to each sample/replicate, using the PCR product diluted ten times to reduce the amount of initial template and guarantee the complete incorporation of indexes in the library. Indexing reactions were carried‐out in volumes of 10 μl, comprising 5 μl 2 × KAPA HiFi HotStart ReadyMix (Kapa Biosystems, Cape Town, South Africa), 1 μl of mixed indexing primer (5 μM stock; Gansauge and Meyer 2013), 2 μl ultrapure water and 2 μl of diluted first‐round PCR product. The indexing thermocycling program used initial denaturing at 95 °C for 3 min, followed by 10 cycles of 95 °C for 30 s, 55 °C for 30 s and 72 °C for 30 s, with an extension of 72 °C for 5 min. Indexing PCR success was evaluated through electrophoresis, and the final sample libraries were purified using 1.2 × AMPure ® XP beads. Each sample library was quantified with Epoch (Biotec, USA) and diluted to 15 nM. These sample libraries were then pooled and quantified using qPCR (KAPA Library Quantification Kit for Illumina platforms) and normalised to 4 nM. The final library was sequenced in an Illumina MiSeq System using approximately three-quarters a lane of MiSeq V2 500‐cycle reagent kit (Illumina, California, USA) with an expected average of 30,000 paired‐end reads per replicate (90,000 per sample).
Bioinformatics
The bioinformatic processing of sequencing reads was conducted using OBITools (Boyer et al. 2016). Initially, paired‐end reads were aligned using the command ‘illuminapairedend’ and discarded if the overlapping quality was < 40 (Taberlet et al. 2018). Unaligned sequences were also removed using the “obigrep” function. The sequences were dereplicated into unique sequences across samples using “obiuniq”, and primer sequences were removed using “ngsfilter”. Sequences with 250–300 bp were kept and sequences with reading counts lower than 10 were removed. Finally, we used “obiclean” with a 50% threshold for the abundance ratio to clean sequences from PCR or sequencing errors. The resulting exact sequence variants (ESVs) obtained were compared with the reference nucleotide database (NCBI) using the BLASTN algorithm (McGinnis and Madden 2004). A full taxonomy for each blast result was further obtained using the “taxonomizr” package in R (https://cran.r-project.org/web/packages/taxonomizr/index.html). Thereafter, a python script was used to condense the taxonomic identifications assigned to each ESV. Clustering considers the assignment to a species level to have a percentage identity of at least 98%, while lower identity thresholds were fixed for assignments to Order or lower taxonomic levels (< 92% of identity), Family (≥ 92%) and Genus (≥ 95%) levels. The number of reads of resulting taxonomic identifications was added per sample, and taxa that exhibited less than 10 reads per sample were excluded from further analyses. Statistical analyses were performed using only sequencing reads assigned to the Species level within Chordata phyla, given the focus of this study on small game species as prey items for wild boar.
Statistical analyses
To characterise the dietary composition, the frequency of occurrence (FO, i.e. the number of faeces containing DNA of each chordate species divided by the total number of faeces examined) was calculated per species/group of species. To test dissimilarity in diet composition (presence/absence of each chordate species) between seasons (two levels: autumn and spring) and hunting estates (two levels: rabbit hunting allowed/forbidden), we used the permutational multivariate analysis of variance (PERMANOVA). The type III (partial) sum of squares was used, and all of the tests were performed with 999 permutations of residuals in a reduced model (Anderson and Ter Braak 2003). The factors were considered to be statistically significant if P < 0.05. The advantage of the permutation approach is that the resulting test is “distribution-free” and is unconstrained by many of the typical assumptions of parametric statistics (Walters and Coen 2006). The percentage similarity procedure (SIMPER) evaluates similarity and dissimilarity between pairs of groups (spring vs. autumn and hunting vs. no hunting rabbits on the estate) and analyses the influence of each taxon (Species, in the present study) regarding similarity/dissimilarity. Here, SIMPER was employed to identify species that mostly contribute to the dissimilarity between seasons and between hunting management. Those species whose FO was less than 5% were omitted in the statistical analyses because rare species may negatively influence multivariate analyses and add little to dissimilarity measurement (Rowe and Holland 2013). These multivariate analyses were performed using Primer, version 6 (Clarke and Gorley 2006), including the PERMANOVA + add-on package (Anderson et al. 2008).
Finally, to determine the factors driving the consumption of small game species by wild boar, two generalised linear mixed models (GzLMM) were performed using the frequency of occurrence of rabbit (FOR) (model 1) and the frequency of occurrence of red-legged partridge (FOP) (model 2) as response variables. Both models were fitted with a binomial distribution and a logit link function. The variables, abundance of wild boar, abundance of carnivores, rabbit density, and red-legged partridge density, were included as explanatory variables in both models, while season (two levels) and hunting management (two levels) were included as factors. The interaction between season and hunting management was also considered. The hunting estate (four levels) was considered a random effect factor. The selection of the most plausible models was carried out by using Akaike’s information criterion (AIC) (Burnham and Anderson 2002) and following a backward stepwise procedure (Zuur et al. 2009). We compared the AIC for small sample sizes (AICc value). We, therefore, selected all the models in which Δi < 2 about the best model. These analyses were carried out using the InfoStat software program (Di Rienzo et al. 2011).
Results
Wild boar and carnivore relative abundance and wild rabbit and red-legged partridge densities
In the study areas, rabbit density (mean ± SD) was 0.66 ± 0.96 rabbits/ha and 0.30 ± 0.41 rabbits/ha in autumn and spring, respectively. Coefficients of the variation of the estimates (mean ± SD) were 34% ± 17% and 22.5% ± 4% in autumn and spring, respectively. Similarly, red-legged partridge density (mean ± SD) was 0.67 ± 0.46 partridges/ha and 0.34 ± 0.42 partridges/ha in autumn and spring, respectively. Coefficients of the variation of the estimates (mean ± SD) were 28% ± 15% and 23.8% ± 15% in autumn and spring, respectively. Carnivore frequency index (mean ± SD) was 0.22 ± 0.07 and 0.16 ± 0.08, while the wild boar frequency index (mean ± SD) was 0.33 ± 0.07 and 0.23 ± 0.14 in autumn and spring, respectively (Table 1).
Chordate diversity in the diet of wild boar
Seventy-one out of the 80 faeces analysed were identified as wild boar, with an average number of reads of 38,823 (ranging between 12,150 and 69,986). One sample was not successfully amplified, and the remaining eight samples were misidentified in the field and belong to mouflon (Ovis orientalis) (n = 3), fallow deer (Dama dama) (n = 2), red fox (Vulpes vulpes) (n = 2) and dog (Canis lupus familiaris) (n = 1). The DNA from 17 chordate species was detected, including one amphibian, two reptiles, five birds and nine mammal species (Table 2). The FO of mammals is clearly predominant as it is present in 77.6% of the droppings, followed by birds in 22.4%, reptiles in 4.2% and amphibians in 1.4% (Fig. 2).
At the species level within mammals, the presence of rabbit and Algerian mouse (Mus spretus) is shown in 38% and 14% of the wild boar droppings, respectively, while the birds, red-legged partridge and common wood pigeon (Columba palumbus), are present in 11.3% and 8.5% of the droppings, respectively (Table 2). In the case of small game species, 39 droppings had the presence of at least one game species (FO = 55%). The relative frequency of these 17 chordate species represents only a small fraction of the total number of reads observed (mean ± SE = 6.37 ± 1.79) and ranges from 0 to 89.50%. This percentage was higher in autumn (7.10% ± 2.41) than in spring (5.03% ± 2.51).
Seasonal variation in the composition of the diet
The results of PERMANOVA showed significant differences in the composition of species present in the wild boar diet depending on the season and hunting management (Table 3).
The SIMPER results showed a dissimilarity in the diet of wild boar of 91.55% between autumn and spring; rabbit and red deer (Cervus elaphus) were the main cause of these differences (Table 4). Also, the results showed a variance in the diet of 90.1% between those estates with rabbit hunting and those without, in which once again rabbit was the main cause of these differences (Table 4).
Variables involved in the frequency of occurrence of wild rabbit (FOR) and red-legged partridge (FOP)
Regarding the variables that can determine the FOR in the diet, the results showed a significant and positive relationship between the abundance of wild boar and rabbit density (Table 5). In addition, and independently of the density of rabbits, there was an effect of the season, where FOR was greater in autumn. The frequency of occurrence was also higher on estates where rabbit hunting is allowed, although this effect is modulated by the interaction with the season and is higher in autumn on estates with rabbit hunting (Fig. 3). Regarding the red-legged partridge, the results showed a significant and positive relationship between the density of partridge and the FOP. The amount of deviance explained in the rabbit model was D = 15.69%, while in the partridge model, it was D = 9.73%.
Predicted mean values (± S.E.) of frequency of occurrence of rabbit (FOR) in different seasons (spring vs. autumn) and on estates with different hunting management systems (hunting vs. no hunting). Capital letters indicate significant differences (P < 0.05) between season and hunting management according to Fisher LSD tests
Discussion
Overall, our results indicate that wild boar is an opportunistic species whose diet is largely determined by the relative availability of different food types (Schley and Roper 2003). In the context of agroecosystems, wild boar feed on a rich community of animal species (including game and livestock species). Wild boar consume carrion (Inagaki et al. 2020; Tobajas et al. 2021a), although the overall relative proportion of scavenged vs. preyed-upon vertebrate foods is frequently unknown (Taylor and Hellgren 1997) because it is often impossible to know whether an animal was killed deliberately or ingested as carrion (Wilcox and Van Vuren 2009). Therefore, facultative scavengers such as wild boar are expected to have no direct effect on taxa they consume as carrion, although they will have a direct negative impact on prey taxa that they consume via predation (Wilson and Wolkovich 2011). Our results suggest differences in diet between seasons and estates with and without rabbit hunting; wild rabbit is the main driver of those differences (Table 4). The models showed that, independently of the wild rabbit population density, FOR was higher in autumn and on estates where the rabbit is hunted, but this was not evidenced for red-legged partridges. Overall, our results suggest an opportunistic consumption of small game species in Mediterranean environments and indicate scavenging behaviour as the main source of wild rabbit consumption, due to the higher FOR observed during the hunting season on hunting estates, although we cannot rule out the role that predation has about FOR. Rabbits and probably partridges are not the preferred resources for wild boar but they may provide an additional font of animal protein when individuals are easily available and when boar populations are high.
Chordate species in the diet and seasonal variation in the composition of the diet
According to our results, the FO of chordate species in the diet was 77.6%, within the percentage range described for Spain (84.5% by Giménez Anaya et al. 2008; 83.8% by Herrero et al. 2004; 50.7% by Herrero et al. 2005; 90.5% by Herrero et al. 2006 or 90.7% by Irizar et al. 2004). However, the FO was slightly higher in autumn (80%) than in spring (73%). Autumn coincides with the hunting season for small game species (e.g. rabbit, Turdus sp., red-legged partridge, red fox, wood pigeon or magpie). In this sense, many of these are game species and, when analysed, the seasonal differences increase (56% and 42% for autumn and spring, respectively). In the case of rabbits, in addition to hunting, another natural cause of mortality is the presence of diseases, like myxomatosis or rabbit haemorrhagic disease (RHD), which may also increase the availability of carcasses. Myxomatosis outbreaks occur during the summer and autumn months, coinciding with the greater presence of competent vectors, as well as the higher density of susceptible hosts (higher density of young rabbits) (Calvete et al. 2002), while RHD appears mainly during spring and winter (Villafuerte et al. 1995). The FOR observed in autumn (FOR = 48%) could be due to hunting and myxomatosis and in spring (FOR = 23%) could be related to outbreaks of RHD or predation on burrows (Schley and Roper 2003). In the case of red-legged partridges, some hunting estates release farm-bred partridges in autumn to increase game yield (Alonso et al. 2005; Díaz-Fernández et al. 2013), which usually experience a high mortality rate (Gortázar et al. 2000; Díaz-Fernández et al. 2012). This could explain the high FOP (21%) found in one of the study areas in autumn. In this context, where other scavengers are scarce or absent (e.g. top predators or vultures; Mateo-Tomás et al. 2015), wild boar can dominate carrion use through their significant body size and adaptations to find and use carrion efficiently (Tobajas et al. 2021a, b). This could indicate some ability of generalists to replace other key vertebrates in the delivery of ecosystem functions and services (e.g. compensated through an increase in the abundance of individuals; Mateo-Tomas et al. 2017). Therefore, the wild boar is more likely to be providing an ecosystem service rather than acting as a true predator of small game species; however, more studies are needed to discern what percentage of the diet may be scavenged and how much is due to predation.
Other important species detected in faeces were domestic (goat and sheep) and wild ungulates (red deer and mouflon), with probability related to the consumption of carcasses (Arrondo et al. 2019). Wild boar consumes wild ungulates or cattle remains (Carrasco-Garcia et al. 2018); since some carcasses are abandoned in the field, 6.3% of farmers in Spain declared that they leave livestock carcasses in the countryside (even when recognising this as an illegal practice; Gigante et al. 2021). In the case of small mammals (such as Crocidura russula or Mus spretus) as well as reptiles such as Blanus cinereus found in wild boar diet are often fossorial or semi-fossorial species, which could be opportunistically predated during rooting behaviour (McDonough et al. 2022).
Frequency of occurrence of rabbit and partridge
The models showed how the FO is directly related to the density of prey species, which confirms what was mentioned above about the opportunistic role of the wild boar. Previous studies based on a correlative approach support the negative effect of wild boar as a predator/competitor on rabbit distribution and abundance (Virgos et al. 2011; Carpio et al. 2014b; Barasona et al. 2021). Although hunters and managers consider wild boar as effective predators of young or diseased rabbits, our results do not make it possible to differentiate predation from carrion consumption, since the maximum consumption occurs in the season of highest density and hunting on the estates. However, an increase in consumption is observed between estates with and about those without rabbit hunting (Fig. 3), which may be due to the presence of carcasses derived from hunting activity (Tobajas et al. 2021b). The abundance of wild boar also showed a positive relationship with the FOR, which could be explained by the attraction of wild boar to areas of higher rabbit density, since rabbits have an aggregated distribution (Lombardi et al. 2003). On the other hand, in the case of red-legged partridge, only the abundance of this species was positively related to FOP. For this species, previous studies showed that high wild boar densities may negatively affect species abundance, mediated by a reduction in food availability and nest predation (Carpio et al. 2014a, 2015; Mori et al. 2021). The predation of nests or chicks, together with the possible predation or mortality rate of released partridges that could be consumed as carrion, could explain their FO in the diet. However, in the case of the red-legged partridge, we did not find a seasonal effect on the FOP (Table 5), so it is not possible to suggest that it is due to scavenging. For this species, wild boar consumes more on the hunting estates and during seasons in which the density is higher. The results could indicate that rabbit and red-legged partridge consumption is opportunistic, consuming more where and when there are more animals.
This study is, to the best of our knowledge, the first approximation concerning the role of rabbit and red-legged partridge in the diet of wild boar in small game estates. The main limitation is the difficulty in distinguishing predation from carrion consumption. However, the use of complementary analyses (such as isotopes), together with the integration of eDNA and population data, might help differentiate predation from scavenging. Our findings highlight how these species constitute an important part of the wild boar diet in these agroecosystems (as well as other game species) when densities are high, indicating the opportunistic nature of wild boar. Studies on a broader temporal scale are also necessary to evaluate the effect of the wild boar on the population dynamics of these species in these agroecosystems.
Availability of data and material
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Acevedo P, Vicente J, Höfle U, Cassinello J, Ruiz-Fons F, Gortázar C (2007) Estimation of European wild boar relative abundance and aggregation: a novel method in epidemiological risk assessment. Epidemiol Infect 135:519–527. https://doi.org/10.1017/S0950268806007059
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Autom Control 19:716–723. https://doi.org/10.1109/TAC.1974.1100705
Alonso ME, Pérez JA, Gaudioso VR, Díez C, Prieto R (2005) Study of survival, dispersal and home range of autumn-released red-legged partridges (Alectoris rufa). Br Poult Sci 46:401–406. https://doi.org/10.1080/00071660500180968
Anderson M, Braak CT (2003) Permutation tests for multi-factorial analysis of variance. J Stat Comput Simul 73:85–113. https://doi.org/10.1080/00949650215733
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for Primer. Guide to Software and Statistical Methods. Plymouth, UK
Arrondo E, Morales-Reyes Z, Moleón M, Cortés-Avizanda A, Donázar JA, Sánchez-Zapata JA (2019) Rewilding traditional grazing areas affects scavenger assemblages and carcass consumption patterns. Basic Appl Ecol 41:56–66. https://doi.org/10.1016/j.baae.2019.10.006
Ballari SA, Cuevas MF, Ojeda RA, Navarro JL (2015) Diet of wild boar (Sus scrofa) in a protected area of Argentina: the importance of baiting. Mammal Res 60:81–87. https://doi.org/10.1007/s13364-014-0202-0
Barasona JA, Carpio A, Boadella M, Gortazar C, Pineiro X, Zumalacárregui C, Vicente J, Vinuela J. (2021) Expansion of native wild boar populations is a new threat for semi-arid wetland areas. Ecol Indic 125:107563. https://doi.org/10.1016/j.ecolind.2021.107563
Barrio IC, Acevedo P, Tortosa FS (2010) Assessment of methods for estimating wild rabbit population abundance in agricultural landscapes. Eur J Wildl Res 56:335–340. https://doi.org/10.1007/s10344-009-0327-7
Barros AL, Curveira-Santos G, Marques TA, Santos-Reis M (2020) Accounting for detection unveils the intricacy of wild boar and rabbit co-occurrence patterns in a Mediterranean landscape. Sci Rep 10:1–9. https://doi.org/10.1038/s41598-020-63492-9
Bencatel J, Sabino-Marques H, Álvares F, Moura AE, Barbosa AM (2019) Atlas de Mamíferos de Portugal. University Press, Évora
Borralho R, Rego F, Pinto PV (1996) Is driven transect sampling suitable for estimating red-legged partridge Alectoris rufa densities? Wildl Biol 2:259–268. https://doi.org/10.2981/wlb.1996.029
Boyer F, Mercier C, Bonin A, Le Bras Y, Taberlet P, Coissac E (2016) Obitools: A unix-inspired software package for DNA metabarcoding. Mol Ecol Resour 16:176–182. https://doi.org/10.1111/1755-0998.12428
Bro E, Reitz F, Clobert J, Migot P, Massot M (2001) Diagnosing the environmental causes of the decline in Grey Partridge Perdix perdix survival in France. Ibis 143:120–132. https://doi.org/10.1111/j.1474-919X.2001.tb04176.x
Buckland ST (2004) Advanced distance sampling. University Press, Oxford
Buenestado FJ, Ferreras P, Delibes-Mateos M, Tortosa FS, Blanco-Aguiar JA, Villafuerte R (2008) Habitat selection and home range size of red-legged partridges in Spain. Agric Ecosyst Environ 126:158–162. https://doi.org/10.1016/j.agee.2008.01.020
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, NY
Calvete C, Estrada R, Villafuerte R, Osácar JJ, Lucientes J (2002) Epidemiology of viral haemorrhagic disease and myxomatosis in a free-living population of wild rabbits. Vet Rec 150:776–782. https://doi.org/10.1136/vr.150.25.776
Carpio AJ, Guerrero-Casado J, Tortosa FS, Vicente J (2014a) Predation of simulated red-legged partridge nests in big game estates from South Central Spain. Eur J Wildl Res 60:391–394. https://doi.org/10.1007/s10344-013-0786-8
Carpio AJ, Guerrero-Casado J, Ruiz-Aizpurua L, Vicente J, Tortosa FS (2014b) The high abundance of wild ungulates in a Mediterranean region: is this compatible with the European rabbit? Wildl Biol 20:161–166. https://doi.org/10.2981/wlb.13113
Carpio AJ, Oteros J, Vicente J, Tortosa FS, Guerrero-Casado J (2015) Factors affecting red-legged partridge Alectoris rufa abundance on big-game hunting estates: implications for management and conservation. Ardeola 62:283–297. https://doi.org/10.13157/arla.62.2.2015.283
Carpio AJ, Apollonio M, Acevedo P (2021) Wild ungulate overabundance in Europe: contexts, causes, monitoring and management recommendations. Mammal Rev 51:95–108. https://doi.org/10.1111/mam.12221
Carr CM, Hardy SM, Brown TM, Macdonald TA, Hebert PD (2011) A tri-oceanic perspective: DNA barcoding reveals geographic structure and cryptic diversity in Canadian polychaetes. PLoS ONE 6:22232. https://doi.org/10.1371/journal.pone.0022232
Carrasco-Garcia R, Barroso P, Perez-Olivares J, Montoro V, Vicente J (2018) Consumption of big game remains by scavengers: a potential risk as regards disease transmission in central Spain. Front Vet Sci 5:4. https://doi.org/10.3389/fvets.2018.00004
Castillo-Contreras R, Carvalho J, Serrano E, Mentaberre G, Fernández-Aguilar X, Colom A, González-Crespo C, Lavín S, López-Olvera JR (2018) Urban wild boars prefer fragmented areas with food resources near natural corridors. Sci Total Environ 615:282–288. https://doi.org/10.1016/j.scitotenv.2017.09.277
Cellina S (2008) Effects of supplemental feeding on the body condition and reproductive state of wild boar Sus scrofa in Luxembourg. Dissertation, University of Sussex
Clarke KR, Gorley RN (2006) PRIMER v6: User Manual/Tutorial. Plymouth, UK
Delibes-Mateos M, Ferreras P, Villafuerte R (2008) Rabbit populations and game management: the situation after 15 years of rabbit haemorrhagic disease in central-southern Spain. Biodivers Conserv 17:559–574. https://doi.org/10.1007/s10531-007-9272-5
Delibes-Mateos M, Farfán MA, Olivero J, Márquez AL, Vargas JM (2009) Long-term changes in game species over a long period of transformation in the Iberian Mediterranean landscape. Environmen Manag 43:1256–1268. https://doi.org/10.1007/s00267-009-9297-5
de Sousa LL, Silva SM, Xavier R (2019) DNA metabarcoding in diet studies: unveiling ecological aspects in aquatic and terrestrial ecosystems. Environmen DNA 1:199–214. https://doi.org/10.1002/edn3.27
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo YC (2011) InfoStat versión 2011. Grupo InfoStat. http://www.infostat.com.ar. Accessed 12 September 2022
Díaz-Fernández S, Viñuela J, Arroyo B (2012) Harvest of red-legged partridge in central Spain. J Wildl Manag 76:1354–1363. https://doi.org/10.1002/jwmg.391
Díaz-Fernández S, Arroyo B, Casas F, Martinez-Haro M, Viñuela J (2013) Effect of game management on wild red-legged partridge abundance. PLoS One 8:e66671. https://doi.org/10.1371/journal.pone.0066671
Díez Valle C, Pérez Garrido JA, Prieto Martin R, Alonso De La Varga ME, Olmedo Olmedo JA (2005) Activity patterns of wild rabbit (Oryctolagus cuniculus) under semi-freedom conditions, during autumn and winter. Wildl Biol Pract 1:41–46
Ebert C, Knauer F, Spielberger B, Thiele B, Hohmann U (2012) Estimating wild boar Sus scrofa population size using faecal DNA and capture-recapture modelling. Wildl Biol 18:142–152. https://doi.org/10.2981/11-002
Edwards PJ, Fletcher MR, Berny P (2000) Review of the factors affecting the decline of the European brown hare, Lepus europaeus (Pallas, 1778) and the use of wildlife incident data to evaluate the significance of paraquat. Agric Ecosyst Environ 79:95–103. https://doi.org/10.1016/S0167-8809(99)00153-X
Gée A, Sarasa M, Pays O (2018) Long-term variation of demographic parameters in four small game species in Europe: opportunities and limits to test for a global pattern. Anim Biodivers Conserv 41:33–60. https://doi.org/10.32800/abc.2018.41.0033
Gigante FD, Santos JP, López-Bao JV, Olea PP, Verschuuren B, Mateo-Tomás P (2021) Farmers’ perceptions towards scavengers are influenced by implementation deficits of EU sanitary policies. Biol Conserv 259:109166. https://doi.org/10.1016/j.biocon.2021.109166
Giménez-Anaya A, Herrero J, Rosell C, Couto S, García-Serrano A (2008) Food habits of wild boars (Sus scrofa) in a Mediterranean coastal wetland. Wetlands 28:197–203. https://doi.org/10.1672/07-18.1
Gortázar C, Villafuerte R, Martín M (2000) Success of traditional restocking of red-legged partridge for hunting purposes in areas of low density of northeast Spain Aragón. Z Jagdwiss 46:23–30. https://doi.org/10.1007/BF02240661
Hearn R, Watkins C, Balzaretti R (2014) The cultural and land use implications of the reappearance of the wild boar in North West Italy: a case study of the Val di Vara. J Rural Stud 36:52–63. https://doi.org/10.1016/j.jrurstud.2014.06.004
Herrero J, Couto S, Rosell C, Arias P (2004) Preliminary data on the diet of wild boar living in a Mediterranean coastal wetland. Galemys 16:115–123. ISSN: 1137–8700
Herrero J, Irizar I, Laskurain NA, García-Serrano A, García-González R (2005) Fruits and roots: wild boar foods during the cold season in the southwestern Pyrenees. Ital J Zool 72:49–52. https://doi.org/10.1080/11250000509356652
Herrero J, García-Serrano A, Couto S, Ortuño VM, García-González R (2006) Diet of wild boar Sus scrofa L. and crop damage in an intensive agroecosystem. Eur J Wildl Res 52:245–250
Herruzo C, Jáuregui MM (2013) Trends in hunters, hunting grounds and big game harvest in Spain. For Syst 22:114–122. https://doi.org/10.1007/s10344-006-0045-3
Inagaki A, Allen ML, Maruyama T, Yamazaki K, Tochigi K, Naganuma T, Koike S (2020) Vertebrate scavenger guild composition and utilization of carrion in an East Asian temperate forest. Ecol Evol 10:1223–1232. https://doi.org/10.1002/ece3.5976
Irizar I, Laskurain NA, Herrero J (2004) Wild boar frugivory in the Atlantic Basque Country. Galemys 16:125–134. ISSN: 1137–8700
Jiménez García D, Martínez Pérez JE, Peiró Clavell V (2006) Relationship between game species and landscape structure in the SE of Spain. Wildl Biol Pract 2:48–62. https://doi.org/10.2461/wbp.2006.2.7
Joffre R, Rambal S, Ratte JP (1999) The dehesa system of southern Spain and Portugal as a natural ecosystem mimic. Agroforest Syst 45:57–79. https://doi.org/10.1023/A:1006259402496
Gansauge MT, Meyer M (2013) Single-stranded DNA library preparation for the sequencing of ancient or damaged DNA. Nat Protoc 8:737–748. https://doi.org/10.1038/nprot.2013.038
Guerrero-Casado J, Carpio AJ, Tortosa FS (2016) Recent negative trends of wild rabbit populations in southern Spain after the arrival of the new variant of the rabbit hemorrhagic disease virus RHDV2. Mammal Biol 81:361–364. https://doi.org/10.1016/j.mambio.2016.03.006
Leray M, Yang JY, Meyer CP, Mills SC, Agudelo N, Ranwez V, Boehm JT, Machida RJ (2013) A new versatile primer set targeting a short fragment of the mitochondrial COI region for metabarcoding metazoan diversity: application for characterizing coral reef fish gut contents. Front Zool 10:1–14. https://doi.org/10.1186/1742-9994-10-34
Lombardi L, Fernández N, Moreno S, Villafuerte R (2003) Habitat-related differences in rabbit (Oryctolagus cuniculus) abundance, distribution, and activity. J Mammal 84:26–36. https://doi.org/10.1644/1545-1542(2003)084%3c0026:HRDIRO%3e2.0.CO;2
Maudet C, Luikart G, Dubray D, Von Hardenberg A, Taberlet P (2004) Low genotyping error rates in wild ungulate feces sampled in winter. Molec Ecol Not 4:772–775. https://doi.org/10.1111/j.1471-8286.2004.00787.x
Massei G, Kindberg J, Licoppe A, Gačić D, Šprem N, Kamler J, Baubet E, Hohmann U, Monaco A, Ozoliņš J, Cellina S (2015) Wild boar populations up, numbers of hunters down? A review of trends and implications for Europe. Pest Manag Sci 71:492–500. https://doi.org/10.1002/ps.3965
Mateo-Tomás P, Olea PP, Moleón M, Vicente J, Botella F, Selva N, Viñuela J, Sánchez-Zapata JA (2015) From regional to global patterns in vertebrate scavenger communities subsidized by big game hunting. Divers Distribut 21:913–924. https://doi.org/10.1111/ddi.12330
Mateo-Tomás P, Olea PP, Moleón M, Selva N, Sánchez-Zapata JA (2017) Both rare and common species support ecosystem services in scavenger communities. Glob Ecol Biogeogr 26:1459–1470. https://doi.org/10.1111/geb.12673
McDonough MT, Ditchkoff SS, Smith MD, Vercauteren KC (2022) A review of the impacts of invasive wild pigs on native vertebrates. Mammal Biol 1–12. https://doi.org/10.1007/s42991-022-00234-6
McGinnis S, Madden TL (2004) BLAST: at the core of a powerful and diverse set of sequence analysis tools. Nucleic Acids Res 32:20–25. https://doi.org/10.1093/nar/gkh435
Morelle K, Fattebert J, Mengal C, Lejeune P (2016) Invading or recolonizing? Patterns and drivers of wild boar population expansion into Belgian agroecosystems. Agric Ecosyst Environ 222:267–275. https://doi.org/10.1016/j.agee.2016.02.016
Mori E, Lazzeri L, Ferretti F, Gordigiani L, Rubolini D (2021) The wild boar Sus scrofa as a threat to ground-nesting bird species: an artificial nest experiment. J Zool 314:311–320. https://doi.org/10.1111/jzo.12887
Oja R, Soe E, Valdmann H, Saarma U (2017) Non-invasive genetics outperforms morphological methods in faecal dietary analysis, revealing wild boar as a considerable conservation concern for ground-nesting birds. PloS One 12:e0179463. https://doi.org/10.1371/journal.pone.0179463
Panek M (1997) The effect of agricultural landscape structure on food resources and survival of grey partridge Perdix perdix chicks in Poland. J Appl Ecol 787–792. https://doi.org/10.2307/2404923
Petersson LK, Milberg P, Bergstedt J, Dahlgren J, Felton AM, Götmark F, Salk C, Löf M (2019) Changing land use and increasing abundance of deer cause natural regeneration failure of oaks: Six decades of landscape-scale evidence. For Ecol Manag 444:299–307. https://doi.org/10.1016/j.foreco.2019.04.037
Robeson MS, Khanipov K, Golovko G, Wisely SM, White MD, Bodenchuck M, Smyser TJ, Fofanov Y, Fierer N, Piaggio AJ (2018) Assessing the utility of metabarcoding for diet analyses of the omnivorous wild pig (Sus scrofa). Ecol Evol 8:185–196. https://doi.org/10.1002/ece3.3638
Rowe HI, Holland JD (2013) High plant richness in prairie reconstructions support diverse leafhopper communities. Restor Ecol 21:174–180. https://doi.org/10.1111/j.1526-100X.2012.00882.x
Schley L, Roper TJ (2003) Diet of wild boar Sus scrofa in Western Europe, with particular reference to consumption of agricultural crops. Mammal Rev 33:43–56. https://doi.org/10.1046/j.1365-2907.2003.00010.x
Smith RK, Jennings NV, Robinson A, Harris S (2004) Conservation of European hares Lepus europaeus in Britain: is increasing habitat heterogeneity in farmland the answer? J Appl Ecol 41:1092–1002. https://doi.org/10.1111/j.0021-8901.2004.00976.x
Sotherton NW (1998) Land use changes and the decline of farmland wildlife: an appraisal of the set-aside approach. Biol Conserv 83:259–268. https://doi.org/10.1016/S0006-3207(97)00082-7
Taberlet P, Bonin A, Zinger L, Coissac E (2018) Environmental DNA: for biodiversity research and monitoring. University Press, Oxford
Taylor RB, Hellgren EC (1997) Diet of feral hogs in the western South Texas Plains. Southwest Nat 33–39.
Thomas L, Buckland ST, Rexstad EA, Laake JL, Strindberg S, Hedley SL, Bishop JR, Marques TA, Burnham KP (2010) Distance software: design and analysis of distance sampling surveys for estimating population size. J Appl Ecol 47:5–14. https://doi.org/10.1111/j.1365-2664.2009.01737.x
Tobajas J, Descalzo E, Ferreras P, Mateo R, Margalida A (2021a) Effects on carrion consumption in a mammalian scavenger community when dominant species are excluded. Mammal Biol 101:851–859. https://doi.org/10.1007/s42991-021-00163-w
Tobajas J, Oliva-Vidal P, Piqué J, Afonso-Jordana I, García-Ferré D, Moreno-Opo R, Margalida A (2021b) Scavenging patterns of generalist predators in forested areas: the potential implications of increase in carrion availability on a threatened capercaillie population. Anim Conserv 25:259–272. https://doi.org/10.1111/acv.12735
Vargas JM, Farfán MA, Guerrero JC, Barbosa AM, Real R (2007) Geographical and environmental correlates of big and small game in Andalusia (southern Spain). Wildl Res 34:498–506. https://doi.org/10.1071/WR06012
Villafuerte R, Calvete C, Blanco JC, Lucientes J (1995) Incidence of viral hemorrhagic disease in wild rabbit populations in Spain. Mammalia 59:651–659. https://doi.org/10.1515/mamm.1995.59.4.651
Virgós E, Lozano J, Cabezas-Díaz S, Mangas JG (2011) The presence of a “competitor pit effect” compromises wild rabbit (Orcytolagus cuniculus) conservation. Anim Biol 61:319–334. https://doi.org/10.1163/157075511X584254
Walters K, Coen LD (2006) A comparison of statistical approaches to analyzing community convergence between natural and constructed oyster reefs. J Experiment Marine Biol Ecol 330:81–95. https://doi.org/10.1016/j.jembe.2005.12.018
Wehr NH (2021) Historical range expansion and biological changes of Sus scrofa corresponding to domestication and feralization. Mammal Res 66:1–12. https://doi.org/10.1007/s13364-020-00534-7
Wilcox JT, Van Vuren DH (2009) Wild pigs as predators in oak woodlands of California. J Mammal 90:114–118. https://doi.org/10.1644/08-MAMM-A-017.1
Wilson EE, Wolkovich EM (2011) Scavenging: how carnivores and carrion structure communities. Trends Ecol Evol 26:129–135. https://doi.org/10.1016/j.tree.2010.12.011
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, NY
Acknowledgements
We would like to thank J. Nesbit for his revision of the English in this manuscript and the farmers for their useful cooperation. We are also grateful to game estate keepers and owners, especially to Carlos Megía and Juan Maroto for their hospitality and assistance.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This work was supported by the Own Research Plan of UCLM, co-financed with the European Regional Development Fund (FEDER), by submitting expressions of interest (Resolution of 02/19/2020 [DOCM 02/26/2020], from the University of Castilla-La Mancha) with reference number 2020-GRIN-29072. Antonio J. Carpio is supported by a “Juan de la Cierva” contract (IJC2020-042629-I) funded by MCIN/AEI/10.13039/501100011033 and by the European Union Next Generation EU/PRTR. Saúl Jiménez-Ruiz was granted for a research mobility within the “Plan Propio” of the University of Castilla-La Mancha for the year 2022.
Author information
Authors and Affiliations
Contributions
Antonio J. Carpio, João Queirós and Pelayo Acevedo conceived and designed the study. Antonio J. Carpio, Eduardo Laguna and Saúl Jiménez-Ruiz performed the data collection. João Queirós and Paulo C. Alves performed genetic analysis. Antonio J. Carpio, Eduardo Laguna and João Queirós analysed the data and wrote the first draft of the manuscript. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Carpio, A.J., Queirós, J., Laguna, E. et al. Understanding the impact of wild boar on the European wild rabbit and red-legged partridge populations using a diet metabarcoding approach. Eur J Wildl Res 69, 18 (2023). https://doi.org/10.1007/s10344-023-01647-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-023-01647-5