Abstract
Parasite and pathogen surveillance is crucial for understanding trends in their distributions and host spectra, as well as to document changes in their population dynamics. Nevertheless, continuous surveillance is time-consuming, underfunded due to the non-charismatic nature of parasites/pathogens, and research infrastructure is usually limited to short-term surveillance efforts. Species observation data provided by the public can contribute to long-term surveillance of parasites using photographic evidence of infections shared on community science platforms. Here, we used public photo repositories to document the occurrence across space and time of Lucilia spp. (Diptera: Calliphoridae), a parasite inducing nasal toad myiasis in the European toad Bufo bufo (Anura: Bufonidae). We found a total of 262 toad myiasis observations on iNaturalist (n = 132), on GBIF (n = 86), on Flickr (n = 41), and on observation.org (n = 3). Our results indicate that the distribution of toad myiasis is regionally limited, despite its host being widely distributed and abundant across a wide region in Europe. Observations were found in 12 countries with relatively low prevalence, including Belgium (3.90%, CI 2.44–6.18), Denmark (1.26%, CI 0.89–1.80), France (0.45%, CI 0.14–1.38), Germany (1.27%, CI 0.92–1.75), Lithuania (0.50%, CI 0.13–1.98), Luxembourg (1.30%, CI 0.42–3.95), the Netherlands (2.71%, CI 1.61–4.52), Poland (0.89%, CI 0.34–2.35), Russia (Kaliningrad Oblast) (4.76%, CI 0.67–27.14), Switzerland (NA), Ukraine (0.87%, CI 0.12–5.91), and in the UK (0.45%, CI 0.28–0.72). Nevertheless, the number of uploaded observations of both parasite infection and host presence indicates a stable increase likely due to the growing popularity of community science websites. Overall, community science is a useful tool to detect and monitor certain wildlife diseases and to recognize potential changes in disease dynamics through time and space.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Amphibians are among the most threatened vertebrates globally. Approximately 39% of described amphibian species are currently classified as near threatened (NT), vulnerable (VU), endangered (EN), or critically endangered (CR) by the International Union for Conservation of Nature (IUCN; IUCN 2022). Recent declines in amphibian populations have been observed globally and are often attributed to various anthropogenic or environmental pressures, including commercial trade of amphibians, climate change, habitat loss, and emerging infectious diseases (Sodhi et al. 2008; Collins 2010). For instance, amphibian-associated diseases (e.g., chytrid fungi Batrachochytrium spp.) are well documented to cause severe population declines and are of critical global conservation concern (Fisher and Garner 2020). Batrachochytrium dendrobatidis was found on ~ 42% of tested amphibian species, globally (Olson et al. 2013), and their presence is occasionally associated with 100% mortality in some populations (Berger et al. 2005). Additionally, amphibians are important “habitats” of a wide variety of obligate parasites, including nematodes, mites, ticks, and dipterans (Campião et al. 2015; Legett et al. 2018; Santos et al. 2018; Mendoza-Roldan et al. 2020). Several dipteran species have been reported to cause myiasis in amphibians globally (Hagman et al. 2005; Kraus 2007; Eaton et al. 2008; Eizemberg et al. 2008; De Mello-Patiu and De Luna-Dias 2010; Glaw et al. 2014; de Souza-Pinto et al. 2015). In Europe, two dipterans are believed to induce myiasis in toads. One of these is the toad fly Lucilia bufonivora, a highly host-specific dipteran parasite, which causes nasal myiasis in European toads (Bufo bufo), but has been occasionally reported to also infect co-occurring frog species (Brumpt 1934a, b; Lada 2009). The presence of L. bufonivora in North America has recently been confirmed with both morphological and molecular methods. However, contrary to observations across the Palearctic, here it causes non-nasal myiasis in local amphibians (Tantawi and Whitworth 2014; Arias-Robledo et al. 2019b; Whitworth et al. 2021). Furthermore, the molecular analysis of infected individuals demonstrated a relatively large genetic distance between the European and North American L. bufonivora populations, suggesting that the presence of this species across the Nearctic region is unlikely to be the result of a recent introduction (Arias-Robledo et al. 2019b). Infections by L. bufonivora are relatively rarely observed throughout its distribution range (with some local exceptions), showing prevalence that ranges from 0 to 12%, depending also on host age and sex (Strijbosch 1980). Infection by L. bufonivora is generally biased toward female toads, and hosts with larger body sizes or older ages, and is usually fatal to hosts (Strijbosch 1980).
The second fly species documented to cause myiasis in amphibians is the common toad fly L. silvarum. This species mostly induces non-nasal myiasis in amphibians across North America. However, no evidence has presented its involvement of toad myiasis in Europe (Arias-Robledo et al. 2019a). The saprophagous behavior of L. silvarum has been documented in the Palearctic region (Zumpt 1956). In addition, a recent study on a broad European scale concluded that L. silvarum is not involved in toad myiasis in the Netherlands, Switzerland, and the UK; instead, L. bufonivora was responsible for all documented infections in these countries (Arias-Robledo et al. 2019b). Overall, there is no current evidence that L. silvarum causes nasal myiasis in European amphibians, including in Bufo bufo; therefore, European dipteran-induced toad nasal myiasis cases are currently assumed to be induced by L. bufonivora, exclusively. Nevertheless, L. silvarum occurs through the Palearctic (Williams et al. 2016), and its involvement in toad myiasis cannot be excluded in this region. Across Europe, both toad-associated fly species, L. bufonivora and L. silvarum, and/or toad myiasis have been observed in multiple countries (Richards 1926; Brumpt 1934a; Rognes 1980; Huijbregts 2002; Kaczorowska 2006; Karpa 2008; Fremdt et al. 2012; Sulakova et al. 2013; Gil Arriortua et al. 2014; Pohjoismäki and Kahanpää 2014; Feddern et al. 2018; Arias-Robledo et al. 2019b). Infection patterns of L. silvarum are largely unknown as infection of toads by this species is rarely observed.
European toads have suffered a population decline in the last two decades (Carrier and Beebee 2003; Bosch and Martínez-Solano 2006; Petrovan and Schmidt 2016). Consequently, it is crucial that conservation efforts also recognize the potential threats their host-specific pathogens and parasites might face (Rózsa and Vas 2015; Carlson et al. 2020). While presence/absence as well as density data are often collected for vertebrate species, these are virtually non-existent for most of their metazoan parasites. Such data are however essential to establish population sizes and distribution ranges, as well as to identify potential population declines. Occurrence data of parasite-induced host myiasis can provide a good approximation of distribution, occurrence, and density of amphibian-associated myiasis-inducing parasites (Lucilia spp.) throughout their range. Due to the advances in and the growing accessibility of photographic technology and to the increasing popularity of community science websites, the observations recorded on these sites have become increasingly important and recognized in parasite and disease monitoring/data collection during the past decade (Crowl et al. 2008; Dickinson et al. 2010; Bartel et al. 2011; Lawson et al. 2015).
In this study, we explore the highly specialized dipteran parasites, Lucilia spp. (Diptera: Calliphoridae), that induce nasal myiasis (i.e., infestation by dipterous larvae of live hosts’ tissues) in amphibian hosts. We aimed to document the distribution, host spectra, as well as population trends of these parasites, due to the highly limited knowledge on these. We rely on public photo repositories and emphasize the growing importance of community-based projects in assessing trends in parasitic geographic ranges and densities. Additionally, observations of dipteran-induced nasal myiasis of toads were analyzed to determine how effectively community science can be applied to access the current distribution and infection rate of these amphibian-specific parasites and associated diseases.
Materials and methods
Toad fly–induced myiasis shows a characteristic infection in the nasal cavity of the host; therefore, it is easily recognizable from photos and can be used to detect dipteran parasitism in these hosts. Infection data were collected from three online photo and data repositories (Flickr, GBIF, and iNaturalist). Additionally, the date and locality coordinates of observations were recorded whenever provided by the observer (either on the website or upon request). Uploaded photos of the target host species were screened for signs of nasal myiasis infection to find parasite occurrence data on all three platforms. Additionally, relevant photos were searched using a combination of different keywords: toad fly AND toad AND parasite OR infection OR disease OR larvae OR Lucilia, to find further occurrence data in secondary host species. Toad fly infections were recorded when the photographed amphibians showed signs of nasal myiasis and the presence of dipteran larvae could be established. Consequently, the infestation rate established here is conservative due to the (presumable) exclusion of hosts with early-stage toad fly infections. Additionally, myiasis presence of only alive toads was considered, excluding visibly deceased individuals. Images duplicated across platforms were considered only once. Given that iNaturalist (unlike Flickr or GBIF) allows the extraction of the total number of observations for specific search criteria, it allows the estimation of prevalence. Therefore, we report all results both specifically to iNaturalist and for combining the three repositories separately. Infection occurrence data was visualized in QGIS (version 3.20.1) (QGIS Development Team 2019) and in R (version 4.0.3) (RTeam 2020), using ggplot2 (Wickham and Chang 2016).
Results
Observations across primary and secondary host species
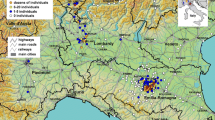
A total of 129 observations of dipteran-induced myiasis in B. bufo were detected on iNaturalist, and 115 observations on other platforms, including Flickr (n = 30) and GBIF (n = 85). Furthermore, seven individuals of three additional (secondary host) species showed signs of myiasis in their nasal cavities, including B. spinosus (Spain, n = 4), Pelophylax sp. (the Netherlands, n = 1), and Rana temporaria (East European Region of Russia, n = 2) (Fig. 1).
Distribution of dipteran-induced nasal myiasis in B. bufo. Green dots indicate iNaturalist; white dots indicate Flickr and GBIF observations. Countries where toad myiasis is confirmed through community science observations are indicated in green, whereas its absence is indicated in dark gray (historically present). Secondary host use (other than B. bufo) observations represented with red stars
Geographical distribution of dipteran-induced nasal myiasis in toads across Europe
The presence of nasal myiasis in B. bufo was observed in 12 countries, including Belgium, Denmark, France, Germany, Lithuania, Luxembourg, the Netherlands, Poland, Russia (East European Region), Switzerland, Ukraine, and UK (Fig. 1, Table 1). Based on the iNaturalist database, toad myiasis was most frequent in Germany (n = 37), in Denmark (n = 30), and in Belgium (n = 17). However, when data from all three repositories were combined, most observations were reported from the Netherlands (n = 87) (Table 1). The highest prevalence was observed in the East European Region of Russia (Kaliningrad Oblast), with 4.76% (95% CI 0.67–27.14); however, sample size was low in the region (n = 21). High prevalence (with a relatively high observation rate) was observed in Belgium and the Netherlands with 3.90% (95% CI 2.44–6.18) and 2.71% (95% CI 1.61–4.52) of individuals being infected, respectively (Table 1). No infection has been recorded in certain areas, such as Central and Southern Europe, where B. bufo observations are abundant based on community science data.
Distribution of toad myiasis between 2004 and 2021 on iNaturalist
Both host and infection observations increased between January 2004 and December 2021 (Fig. 2). The highest number of B. bufo observations was submitted in 2021 (n = 6344), a 30.61% increase from 2020 (n = 4857) (Fig. 2). Similarly, the highest number of nasal myiasis presence was observed in 2021 (n = 57), an increase of 62.85% compared to 2020 (n = 35) (Fig. 2). The highest infection rate (1.01%) was observed in 2016, including all host individuals, and excluding the years with less than 100 host observations (i.e., 2004–2014). The overall mean prevalence of toad myiasis between 2015 and 2021 was 0.83%. Most observations were reported from June to August (Table S1), with the earliest observations in May and the latest in October (Table S1).
Number of nasal toad myiasis observations (green circles, left Y axis) and B. bufo (gray barplots, right Y axis) observations found on iNaturalist uploaded between January 1, 2004, and December 31, 2021, in countries where both infection and host occurred (see Table 1)
Discussion
Community science has already contributed tremendously to the monitoring of numerous parasitic and pathogenic organisms. For instance, information was obtained on the entomopathogenic fungi of the invasive Asian ladybirds (Harmonia axyridis) in the USA or on the infectious eye disease (caused by bacteria Mycoplasma gallisepticum) in house finches Carpodacus mexicanus, producing visible symptoms in their hosts (Altizer et al. 2004). Volunteer observers’ reports on the transmissible facial tumor disease, threatening populations of the endangered Tasmanian devil (Sarcophilus harrisii), have proved to be extremely valuable in disease tracking (Hawkins et al. 2006). Moreover, medically important arthropods, such as kissing bugs (Triatominae), the vectors of Chagas disease, which poses a high public health risk, have been tracked using insect samples collected by the public (Curtis-Robles et al. 2015). Invasive mosquito species, which can carry a wide variety of human pathogens, have also been tracked and monitored based on data provided by volunteers (Palmer et al. 2017; Walther and Kampen 2017; Bartumeus et al. 2018; Pernat et al. 2020). Community science has also been a valuable tool in documenting tick bites and in inferring tick-borne disease distribution and seasonality in several regions (Garcia-Marti et al. 2018; Lernout et al. 2019; Porter et al. 2019; Eisen and Eisen 2020). Additionally, crowd-sourced specimens and occurrence data have significantly contributed to our understanding on the geographic distribution and phylogeny of hairworm (Nematomorpha) species (Hanelt et al. 2015; Doherty et al. 2021). These and similar studies emphasize the importance of community science in the research of non-medical parasites, as well.
Bufo bufo is protected and listed in Red Data books in several European countries, as well as listed on Appendix III of the Bern Convention. However, there have been no efforts made to assess the vulnerability of the unique and highly specific parasites of this amphibian species. In this work, data collected by the public was used to broaden our understanding about the distribution and infection patterns of toad myiasis likely caused by the dipteran parasite, Lucilia bufonivora or L. silvarum.
This study demonstrates that, while toad myiasis is most frequent in B. bufo, it occasionally occurs in other species, such as B. spinosus, Pelophylax sp., and Rana temporaria. These additional infections probably represent secondary host use but may also be the result of phenotypically similar infection by another parasitic dipteran species. Infections of non-primary hosts are frequently observed, even in highly specialized parasites (Lootvoet et al. 2013; Haelewaters et al. 2017; Sándor et al. 2021), though development usually fails (Tompkins and Clayton 1999). Currently, there is no data about the developmental success of L. bufonivora and L. silvarum in non-primary hosts. Future studies are needed, using both molecular and traditional taxonomic tools to identify which Lucilia species are involved in nasal myiasis across toad populations in Europe.
Our community science–based database revealed no reports of toad myiasis from Albania, Austria, Bulgaria, Czech Republic, Estonia, Finland, Hungary, Norway, Romania, Slovakia, and Sweden, despite toad-associated parasitic Lucilia spp. being historically present in these countries (Pape et al. 2015; de Jong 2016) and despite the relatively large number of toad observations uploaded from this region. With the growing popularity of community science projects, more host observations are expected in the future, improving data resolution and our ability to document the presence/prevalence of toad myiasis globally. Additionally, community science datasets will likely serve as incredibly valuable long-term data sources that will contribute to improving our understanding on changes in geographic ranges, population sizes, or prevalences of parasitic organisms in the future.
Detecting the presence of parasites and signs of diseases through photos shared by the public may provide valuable information about the occurrence of current and newly introduced pathogens and parasites, improving surveillance efforts. Several wildlife diseases and parasites show characteristic infection symptoms, and therefore their potential occurrence can be detected using community science data. Besides photos uploaded by the public on platforms used here, online databases of camera trap photos (e.g., https://www.wildlifeinsights.org/) could be used as additional sources of parasite/disease surveillance. Camera trap photos have already been used to characterize the distribution and seasonality of sarcoptic mange (caused by the mite Sarcoptes scabiei) in carnivores (Carricondo-Sanchez et al. 2017). Moreover, the presence and abundance of dermal pathogens and parasites of aquatic mammals have also been monitored using photography-based tools (Hunt et al. 2013). Pseudogymnoascus destructans (the causative agent of white-nose syndrome) shows visible symptoms on the dermal tissue of infected bats and can be recognized from photos (Puechmaille et al. 2011). Botfly infection also frequently appears on photographs submitted to community science sites (e.g., tree squirrel botfly Cuterebra emasculator, infecting its host, the grey squirrel Sciurus carolinensis). While reliable species-level identification of parasites requires morphological and/or molecular examination, these observations provide a good indication of the presence and abundance of certain parasite genera in their hosts and at different locations. Using photographs to access the presence, distribution, and seasonality of pathogens is regularly applied, not only in animal-associated pathogens or parasites, but also in research on phytopathogenic species. For instance, aerial photographs can provide data on wide-scale infection presence in various plant species (Mohanty et al. 2016; Castelao Tetila et al. 2017; Wiesner-Hanks et al. 2019).
Biodiversity conservation, including the conservation of rare and vulnerable parasites, is increasingly recognized, and needs more research focus both now and in the future (Dougherty et al. 2016; Cizauskas et al. 2017; Carlson et al. 2020; Szentiványi et al. 2020). More studies are needed to better understand which environmental factors drive the distribution of vulnerable parasite species and to better protect their habitats to ensure their future survival. Overall, community science is an increasingly important tool in parasite and disease surveillance, such as in the case of the current study, and may gain further importance in the detection of rarely observed parasite species, contributing to conservation efforts.
References
Altizer S, Hochachka WM, Dhondt AA et al (2004) Seasonal dynamics of mycoplasmal conjunctivitis in Eastern North American house finches. J Anim Ecol 73:309–322
Arias-Robledo G, Stark T, Wall RL, Stevens JR (2019a) The toad fly Lucilia bufonivora: its evolutionary status and molecular identification. Med Vet Entomol 33:131–139. https://doi.org/10.1111/mve.12328
Arias-Robledo G, Wall R, Szpila K et al (2019b) Ecological and geographical speciation in Lucilia bufonivora: the evolution of amphibian obligate parasitism. Int J Parasitol Parasites Wildl 10:218–230. https://doi.org/10.1016/j.ijppaw.2019.09.005
Bartel RA, Oberhauser KS, De Roode JC, Altizer SM (2011) Monarch butterfly migration and parasite transmission in eastern North America. Ecology 92:342–351. https://doi.org/10.1890/10-0489.1
Bartumeus F, Oltra A, Palmer JRB (2018) Citizen science: a gateway for innovation in disease-carrying mosquito management? Trends Parasitol 34:727–729. https://doi.org/10.1016/j.pt.2018.04.010
Berger L, Marantelli G, Skerratt LF, Speare R (2005) Virulence of the amphibian chytrid fungus Batrachochytrium dendrobatidis varies with the strain. Dis Aquat Organ 68:47–50
Bosch J, Martínez-Solano I (2006) Chytrid fungus infection related to unusual mortalities of Salamandra salamandra and Bufo bufo in the Peñalara Natural Park, Spain. Oryx 40:84–89. https://doi.org/10.1017/S0030605306000093
Brumpt E (1934a) Recherches expérimentales sur la biologie de la Lucilia bufonivora. Ann Parasitol Hum Comparée 12:81–97. https://doi.org/10.1051/parasite/1934122081
Brumpt E (1934b) Spécificité parasitaire et déterminisme de la ponte de la mouche Lucilia bufonivora. Comptes Rendus Hebd Des Séances L’académie Des Sci 198:124–126
Campião KM, De Aquino Ribas AC, Morais DH et al (2015) How many parasites species a frog might have? Determinants of parasite diversity in South American anurans. PLoS ONE 10:1–12. https://doi.org/10.1371/journal.pone.0140577
Carlson CJ, Hopkins S, Bell KC et al (2020) A global parasite conservation plan. Biol Conserv 250:108596. https://doi.org/10.1016/j.biocon.2020.108596
Carricondo-Sanchez D, Odden M, Linnell JDC, Odden J (2017) The range of the mange: spatiotemporal patterns of sarcoptic mange in red foxes (Vulpes vulpes) as revealed by camera trapping. PLoS ONE 12:1–16. https://doi.org/10.1371/journal.pone.0176200
Carrier JA, Beebee TJC (2003) Recent, substantial, and unexplained declines of the common toad Bufo bufo in lowland England. Biol Conserv 111:395–399. https://doi.org/10.1016/S0006-3207(02)00308-7
Castelao Tetila E, Brandoli Machado B, Belete NADS et al (2017) Identification of soybean foliar diseases using unmanned aerial vehicle images. IEEE Geosci Remote Sens Lett 14:2190–2194. https://doi.org/10.1109/LGRS.2017.2743715
Cizauskas CA, Carlson CJ, Burgio KR et al (2017) Parasite vulnerability to climate change: an evidence-based functional trait approach. R Soc Open Sci. https://doi.org/10.1098/rsos.160535
Collins JP (2010) Amphibian decline and extinction: what we know and what we need to learn. Dis Aquat Organ 92:93–99. https://doi.org/10.3354/dao02307
Crowl TA, Crist TO, Parmenter RR et al (2008) The spread of invasive species and infectious disease as drivers of ecosystem change. Front Ecol Environ 6:238–246. https://doi.org/10.1890/070151
Curtis-Robles R, Wozniak EJ, Auckland LD et al (2015) Combining public health education and disease ecology research: using citizen science to assess Chagas disease entomological risk in Texas. PLoS Negl Trop Dis 9:1–12. https://doi.org/10.1371/journal.pntd.0004235
de Jong, Y. et al. (2014) Fauna Europaea - all European animal species on the web. Biodiversity Data Journal 2: e4034. https://doi.org/10.3897/BDJ.2.e4034
De Mello-Patiu CA, De Luna-Dias C (2010) Myiasis in the neotropical amphibian Hypsiboas beckeri (Anura: Hylidae) by a new species of Lepidodexia (Diptera: Sarcophagidae). J Parasitol 96:685–688. https://doi.org/10.1645/GE-2423.1
de Souza-Pinto FC, França IF, de Mello-Patiu CA (2015) Brief description of myiasis cases in three amphibian species from Atlantic Forest located in the central region of the State of Minas Gerais, Brazil. Herpetol Notes 8:287–290
Dickinson JL, Zuckerberg B, Bonter DN (2010) Citizen science as an ecological research tool: challenges and benefits. Annu Rev Ecol Evol Syst 41:149–172. https://doi.org/10.1146/annurev-ecolsys-102209-144636
Doherty JF, Filion A, Bennett J et al (2021) The people vs science: can passively crowdsourced internet data shed light on host-parasite interactions? Parasitology 148:1313–1319. https://doi.org/10.1017/S0031182021000962
Dougherty ER, Carlson CJ, Bueno VM et al (2016) Paradigms for parasite conservation. Conserv Biol 30:724–733. https://doi.org/10.1111/cobi.12634
Eaton BR, Moenting AE, Paszkowski CA, Shpeley D (2008) Myiasis by Lucilia silvarum (Calliphoridae) in amphibian species in boreal Alberta, Canada. J Parasitol 94:949–952. https://doi.org/10.1645/GE-1373.1
Eisen L, Eisen RJ (2020) Benefits and drawbacks of citizen science to complement traditional data gathering approaches for medically important hard ticks (Acari: Ixodidae) in the United States. J Med Entomol. https://doi.org/10.1093/jme/tjaa165
Eizemberg R, Sabagh LT, Mello RDS (2008) First record of myiasis in Aplastodiscus arildae (Anura: Hylidae) by Notochaeta bufonivora (Diptera: Sarcophagidae) in the Neotropical area. Parasitol Res 102:329–331. https://doi.org/10.1007/s00436-007-0767-5
Feddern N, Amendt J, Schyma C et al (2018) A preliminary study about the spatiotemporal distribution of forensically important blow flies (Diptera: Calliphoridae) in the area of Bern, Switzerland. Forensic Sci Int 289:57–66. https://doi.org/10.1016/j.forsciint.2018.05.022
Fisher MC, Garner TWJ (2020) Chytrid fungi and global amphibian declines. Nat Rev Microbiol 18:332–343. https://doi.org/10.1038/s41579-020-0335-x
Fremdt H, Szpila K, Huijbregts J et al (2012) Lucilia silvarum Meigen, 1826 (Diptera: Calliphoridae)-a new species of interest for forensic entomology in Europe. Forensic Sci Int 222:335–339. https://doi.org/10.1016/j.forsciint.2012.07.013
Garcia-Marti I, Zurita-Milla R, Harms MG, Swart A (2018) Using volunteered observations to map human exposure to ticks. Sci Rep 8:1–10. https://doi.org/10.1038/s41598-018-33900-2
Gil Arriortua M, Martínez de Pancorbo M, Bordas MIS (2014) Presence of Lucilia bufonivora (Diptera, Calliphoridae) confirmed in the Basque Country (northern Spain). Boletín La Asoc Española Entomol 38:25–31
Glaw F, Morinìere J, Glaw K, Doczkal D (2014) Myiasis bei der Erdkröte (Bufo bufo) verursacht durch die Schmeißfliege Lucilia ampullacea. Z Feldherpetol 21:83–95
Haelewaters D, Pfliegler WP, Szentiványi T et al (2017) Parasites of parasites of bats: Laboulbeniales (Fungi: Ascomycota) on bat flies (Diptera: Nycteribiidae) in central Europe. Parasit Vectors 10:96
Hagman M, Pape T, Schulte R (2005) Flesh fly myiasis (Diptera, Sarcophagidae) in Peruvian poison frogs genus Epipedobates (Anura, Dendrobatidae). Phyllomedusa 4:69–73. https://doi.org/10.11606/issn.2316-9079.v4i1p69-73
Hanelt B, Schmidt-Rhaesa A, Bolek MG (2015) Cryptic species of hairworm parasites revealed by molecular data and crowdsourcing of specimen collections. Mol Phylogenet Evol 82:211–218. https://doi.org/10.1016/j.ympev.2014.09.010
Hawkins CE, Baars C, Hesterman H et al (2006) Emerging disease and population decline of an island endemic, the Tasmanian devil Sarcophilus harrisii. Biol Conserv 131:307–324. https://doi.org/10.1016/j.biocon.2006.04.010
Huijbregts H (2002) Nederlandse bromvliegen (Diptera: Calliphoridae). Entomol Ber 62:82–89
Hunt KE, Moore MJ, Rolland RM et al (2013) Overcoming the challenges of studying conservation physiology in large whales: a review of available methods. Conserv Physiol 1:cot006–cot006. https://doi.org/10.1093/conphys/cot006
IUCN (2022) The IUCN Red List of Threatened Species. Version 2022-1. https://www.iucnredlist.org. Accessed on 4 Mar 2022
Kaczorowska E (2006) Blowflies (Diptera : Calliphoridae) in the saline habitats of the Polish Baltic coast. Polish J Entomol 75:11–27
Karpa A (2008) Catalogue of Latvian flies (Diptera: Brachycera). Latv Entomol 46:4–43
Kraus F (2007) Fly parasitism in Papuan frogs, with a discussion of ecological factors influencing evolution of life-history differences. J Nat Hist 41:1863–1874. https://doi.org/10.1080/00222930701511875
Lada GA (2009) On amphibian parasite Lucilia bufonivora (Insecta, Diptera, Calliphoridae) in Tambov region. Curr Stud Herpetol 9:62–64
Lawson B, Petrovan SO, Cunningham AA (2015) Citizen science and wildlife disease surveillance. EcoHealth 12:693–702. https://doi.org/10.1007/s10393-015-1054-z
Legett HD, Baranov VA, Bernal XE (2018) Seasonal variation in abundance and diversity of eavesdropping frog-biting midges (Diptera, Corethrellidae) in a neotropical rainforest. Ecol Entomol 43:226–233. https://doi.org/10.1111/een.12492
Lernout T, De Regge N, Tersago K et al (2019) Prevalence of pathogens in ticks collected from humans through citizen science in Belgium. Parasit Vectors 12:1–11. https://doi.org/10.1186/s13071-019-3806-z
Lootvoet A, Blanchet S, Gevrey M et al (2013) Patterns and processes of alternative host use in a generalist parasite: insights from a natural host-parasite interaction. Funct Ecol 27:1403–1414. https://doi.org/10.1111/1365-2435.12140
Mendoza-Roldan J, Ribeiro SR, Castilho-Onofrio V et al (2020) Mites and ticks of reptiles and amphibians in Brazil. Acta Trop 208:105515. https://doi.org/10.1016/j.actatropica.2020.105515
Mohanty SP, Hughes DP, Salathé M (2016) Using deep learning for image-based plant disease detection. Front Plant Sci 7:1–10. https://doi.org/10.3389/fpls.2016.01419
Olson DH, Aanensen DM, Ronnenberg KL et al (2013) Mapping the global emergence of Batrachochytrium dendrobatidis, the amphibian chytrid fungus. PLoS ONE. https://doi.org/10.1371/journal.pone.0056802
Palmer JRB, Oltra A, Collantes F et al (2017) Citizen science provides a reliable and scalable tool to track disease-carrying mosquitoes. Nat Commun 8:1–12. https://doi.org/10.1038/s41467-017-00914-9
Pape T, Beuk P, Pont AC et al (2015) Fauna Europaea: Diptera - Brachycera. Biodivers Data J 3:1–31. https://doi.org/10.3897/BDJ.3.e4187
Pernat N, Kampen H, Jeschke JM, Werner D (2020) Citizen science versus professional data collection: comparison of approaches to mosquito monitoring in Germany. J Appl Ecol. https://doi.org/10.1111/1365-2664.13767
Petrovan SO, Schmidt BR (2016) Volunteer conservation action data reveals large-scale and long-term negative population trends of a widespread amphibian, the common toad (Bufo bufo). PLoS ONE 11:1–12. https://doi.org/10.1371/journal.pone.0161943
Pohjoismäki J, Kahanpää J (2014) Checklist of the superfamilies oestroidea and hippoboscoidea of Finland (insecta, Diptera). Zookeys 408:383–408. https://doi.org/10.3897/zookeys.441.7252
Porter WT, Motyka PJ, Wachara J et al (2019) Citizen science informs human-tick exposure in the Northeastern United States. Int J Health Geogr 18:1–14. https://doi.org/10.1186/s12942-019-0173-0
Puechmaille SJ, Wibbelt G, Korn V et al (2011) Pan-European distribution of white-nose syndrome fungus (Geomyces destructans) not associated with mass mortality. PLoS ONE. https://doi.org/10.1371/journal.pone.0019167
QGIS Development Team (2019) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
Richards OW (1926) Notes on the British species of Lucilia (Diptera). Trans R Entomol Soc London 255–260
Rognes K (1980) The blow-fly genus Lucilia Robineau-Desvoidy (Diptera, Calliphoridae) in Norway. Fauna Nor Nor J Entomol Ser B 39–52
Rózsa L, Vas Z (2015) Co-extinct and critically co-endangered species of parasitic lice, and conservation-induced extinction: should lice be reintroduced to their hosts? Oryx 49:107–110. https://doi.org/10.1017/S0030605313000628
Sándor AD, Mihalca AD, Domşa C et al (2021) Argasid ticks of Palearctic bats: distribution, host selection, and zoonotic importance. Front Vet Sci. https://doi.org/10.3389/fvets.2021.684737
Santos JC, Tarvin RD, O’Connell LA et al (2018) Diversity within diversity: parasite species richness in poison frogs assessed by transcriptomics. Mol Phylogenet Evol 125:40–50. https://doi.org/10.1016/j.ympev.2018.03.015
Sodhi NS, Bickford D, Diesmos AC et al (2008) Measuring the meltdown: drivers of global amphibian extinction and decline. PLoS ONE 3:1–8. https://doi.org/10.1371/journal.pone.0001636
Strijbosch H (1980) Mortality in a population of Bufo bufo resulting from the fly Lucilia bufonivora. Oecologia 45:285–286
Sulakova H, Rognes K, Bartak M, Kubik S (2013) Calliphoridae (Diptera) of Vráž nr. Písek (Czech Republic). Proc Workshop biodiversity, Jevany, 2–3.th July, 2013 381–388. https://doi.org/10.13140/2.1.3268.3203
Szentiványi T, Markotter W, Dietrich M et al (2020) Host conservation through their parasites: molecular surveillance of vector-borne microorganisms in bats using ectoparasitic bat flies. Parasite 27:1–10. https://doi.org/10.1051/parasite/2020069
Tantawi TI, Whitworth T (2014) First record of Lucilia bufonivora Moniez, 1876 (Diptera: Calliphoridae) from North America and key to North American species of the L. bufonivora species group. Zootaxa 3881:101–124. https://doi.org/10.11646/zootaxa.3881.2.1
RStudio Team (2020) RStudio: integrated development for R. RStudio, PBC, Boston, MA. http://www.rstudio.com/
Tompkins DM, Clayton DH (1999) Host resources govern the specificity of swiftlet lice: size matters. J Anim Ecol 68:489–500. https://doi.org/10.1046/j.1365-2656.1999.00297.x
Walther D, Kampen H (2017) The citizen science project “Mueckenatlas” helps monitor the distribution and spread of invasive mosquito species in Germany. J Med Entomol 54:1790–1794. https://doi.org/10.1093/jme/tjx166
Whitworth TL, Bolek MG, Arias-Robledo G (2021) Lucilia bufonivora, not Lucilia silvarum (Diptera: Calliphoridae), causes myiasis in anurans in North America with notes about Lucilia elongata and Lucilia thatuna. J Med Entomol 58:88–92. https://doi.org/10.1093/jme/tjaa143
Wickham H, Chang W (2016) Package “ggplot2.” Cham, Switz
Wiesner-Hanks T, Wu H, Stewart E et al (2019) Millimeter-level plant disease detection from aerial photographs via deep learning and crowdsourced data. Front Plant Sci 10:1–11. https://doi.org/10.3389/fpls.2019.01550
Williams KA, Lamb J, Villet MH (2016) Phylogenetic radiation of the greenbottle flies. Zookeys 86:59–86. https://doi.org/10.3897/zookeys.568.6696
Zumpt F (1956) Calliphorinae. Die Fliegen der Palaearktischen Region. In: Lindner E (ed) Vol 11. Stuttgart, pp 1–140
Acknowledgements
We are grateful to Tomas Kay and Dr. Christian Gortázar for their constructive suggestions on the first draft of this work. Additionally, we are thankful to Alexey V. Katz, who contributed to the collection of toad myiasis observations on iNaturalist. Moreover, we would like to express our gratitude to those users who provided additional data of their observations on our request on photo repository platforms and to all who contributed with their observations on these websites.
Funding
Open access funding provided by ELKH Centre for Ecological Research. OV is supported by the National Scientific Research Fund (OTKA K143421) and by a grant of the Romanian Ministry of Research, Innovation, and Digitization (CNCS – UEFISCDI, project number PN-III-P1-1.1-TE-2021–0502).
Author information
Authors and Affiliations
Contributions
TS collected and visualized the data and conceptualized the study; both TS and OV analyzed the data and wrote and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10344_2022_1623_MOESM1_ESM.csv
Data supporting the findings of this work can be found in Table S1. Additionally, most iNaturalist data are publicly available at https://www.inaturalist.org/projects/toad-fly-lucilia-spp-myiasis-in-amphibians. (CSV 40 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szentivanyi, T., Vincze, O. Tracking wildlife diseases using community science: an example through toad myiasis. Eur J Wildl Res 68, 74 (2022). https://doi.org/10.1007/s10344-022-01623-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-022-01623-5