Abstract
The first detection of Fascioloides (F.) magna in northeastern Bavaria in 2011 was presumably correlated to natural migration movements of free ranging wild ruminants originating from the neighboring Czech Republic, where high infection rates have been reported frequently. To gain more data on the continuing spreading and current occurrence of the giant liver fluke in surrounding regions, 700 livers of cloven-hoofed game originating from eleven different northeastern Bavarian counties were investigated for the presence of F. magna and accompanying liver parasites in the hunting season 2019–2020. Macroscopically altered liver tissue was further investigated by pathohistological and parasitological examination. F. magna was detected in 5.9% (38/640) of livers from red deer (0.7% < 1 year, 9.8% > 1 year; p < 0.05) whereas none of the investigated livers of wild boar, roe, and fallow deer was infected (n=60). Mild pathological alterations of the liver tissue were documented in 15, moderate in 14, and major in 9 of all F. magna-positive cases. Histologically, the fluke-specific pigment haematin, large trematode eggs, and periportal fibrosis were detected in the liver tissue of infected animals. In 9% of all investigated livers, parasitic stages of other parasites, i.e., Dicrocoelium dendriticum (6.0%), Fasciola hepatica (0.3%), Taenia spp. (1.7%), and Echinococcus multilocularis (1.0%), were found. According to the results of this study, F. magna is not restricted to formerly known affected regions of Upper Franconia, but is also present in the military training ground Grafenwöhr, Upper Palatinate, and the Bavarian Forest National Park, Lower Bavaria, with estimated prevalences of 3.6 and 16.4%, respectively, and thus seems to spread in suitable habitats in northeastern Bavaria.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the introduction of the giant liver fluke, Fascioloides (F.) magna (Bassi 1875) from North America, the trematode has established itself in numerous European countries starting off in Italy (Bassi 1875), the Czech Republic, and Slovakia (Erhardová-Kotrlá 1971; Kasny et al. 2012; Rajsky et al. 2002) spreading out to Austria (Sattmann et al. 2014), Croatia (Janicki et al. 2005), Hungary (Majoros et al. 1994; Nagy et al. 2018), and just recently to Germany (Plötz et al. 2015, Rehbein et al. 2021). Using different water snails of the family Lymnaeidae as intermediate hosts, the highly pathogenic fluke infects not only wild ruminants but also a variety of other mammals showing a variable pathogenicity depending on the affected definitive host species (Erhardová-Kotrlá 1971). Severe pathologic liver changes with fatal course of disease have been described for infected roe deer (Capreolus capreolus), sheep (Ovis aries), and goats (Capra hircus) whereas red deer (Cervus elaphus) and fallow deer (Dama dama) might carry high numbers of the trematode correlated with a considerable destruction of liver tissue without showing any clinical signs (Ursprung et al. 2006). After the arrival of F. magna in northeastern Bavaria in 2011 (Rehbein et al. 2021), high infection rates have recently been reported in the red deer population in the Veldensteiner forest, Upper Franconia (König et al. 2019), leading to the assumption that the trematode might also be present in wild ungulates living in bordering counties. The increasing prevalence for F. magna in red deer living in certain regions in Germany might rise a possible threat not only for the adjacent roe deer population but also for sheep herds grazing on pastures close to affected forest sections (Foreyt 1990). According to reports of experimental infections of ovines with Fascioloides-metacercariae leading to fatal casualties caused by a small number of immature migrating flukes within a short period of time, it should be avoided to raise sheep in regions with evidence of the giant liver fluke (Foreyt 1996, Foreyt and Todd 1976). Thus, the frequent tracking of Fascioloides-positive wild ungulates as previously realized in Austria seems to be of major importance in order to avert possible related economic damages (Ursprung et al. 2006). Since detailed information on the current spreading of the giant liver fluke due to natural migrations of wild ruminants in northeastern Bavaria is still scarce, the present study was performed to gain data on the occurrence and geographical expansion of F. magna and other liver parasites in cloven-hoofed game from 14 counties in Upper and Middle Franconia, Upper Palatinate, and Lower Bavaria.
Methods
Sample origin
From October 2019 to February 2020, a total of 700 shot pieces of cloven-hoofed game was investigated for the presence of F. magna and accompanying liver parasites at the Bavarian Health and Food Safety Authority (LGL), Erlangen, Germany. Livers were obtained from 640 pieces of red deer (Cervus elaphus; 251 male, 389 female), 24 pieces of roe deer (Capreolus capreolus; four male, 20 female), one piece of fallow deer (Dama dama; male), and 35 pieces of wild boar (Sus scrofa; 17 male, 18 female).
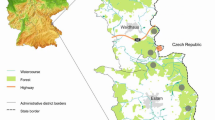
The majority of samples (554/700) was collected during driven hunts at the military training grounds Grafenwöhr (counties Amberg-Sulzbach/Neustadt a.d. Waldnaab) and Hohenfels (counties Neumarkt/Amberg-Sulzbach). The second largest collection region was the Bavarian Forest National Park (counties Regen/Freyung-Grafenau) with 110 samples. The remaining livers (36/700) originated from animals shot in the framework of private hunts in eight different counties in northeastern Bavaria (Fig. 1). All samples were investigated directly within 24 to 48 h after arrival in Erlangen or frozen at −20 °C until further processing.
Fourteen selected counties (marked in yellow, green, and red) for sample collection in Upper and Middle Franconia, Upper Palatinate, and Lower Bavaria, namely, Amberg-Sulzbach (AS), Bayreuth (BT), Cham (CHA), Forchheim (FO), Freyung-Grafenau (FRG), Hof (HO), Kulmbach (KU), Neustadt a. d. Waldnaab (NEW), Neumarkt (NM), Nürnberger Land (LAU), Regen (REG), Schwandorf (SAD), Tirschenreuth (TIR), and Wunsiedel (WUN). Counties marked in red: sample submissions with detection of F. magna; counties marked in green: sample submissions without detection of F. magna; counties marked in yellow: selected for sample collection, but without sample submissions (no data available). Streaked circle 1: Veldensteiner forest; streaked circles 2–4: origin of 95% of investigated samples (2 military training ground Grafenwöhr, 3 military training ground Hohenfels, 4 Bavarian Forest National Park). Inset: location of included counties in Germany
Pathological and histological investigations
During a first macroscopic examination, the surfaces of all livers were screened for any pathological alterations before the organs were cut into slices with a maximum thickness of 1 cm in order to evaluate the liver parenchyma in detail. Any pathological-anatomical changes of the livers were documented, and in case of a suspected infection with F. magna, a categorization of the alterations was conducted into “absent” (–), “minor” (+), “moderate” (++), or “major extent” (+++) according to Plötz et al. (2015). Altered liver tissue was cut into small pieces, fixed in 4% neutral-buffered formaldehyde for at least 24 h and processed routinely for the histological investigation. After hematoxylin-eosin (HE)-staining, the slides were examined microscopically with 25 × to 630 × magnification using a light microscope.
Parasitological investigations
All livers were screened for liver parasites macroscopically. Number and size of adult parasites or developmental parasitic stages were documented followed by a morphological species determination (Naem et al. 2012). Liver slices of altered organs were covered with water over night for a sedimentation process. After 12–24 h, the liver tissue was dumped into a sieve and rinsed with tap water. The sieve residual was examined for adult or juvenile parasitic stages. The rinsing liquid was sedimented for an hour followed by several sedimentation periods of 15 min until excess particles were removed. The final sediment was examined for parasite eggs in a petri dish with 100 × magnification using a light microscope.
Molecular investigations
Parasitic material (adults, juvenile stages, or eggs) as well as liver tissue with morphological alterations of categories (+)–(+++) was frozen at −20 °C until further investigation by PCR. Tissue samples were taken at four representative locations per F. magna-suspicious liver. DNA was extracted using the QIAamp® DNA Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s recommended protocol for tissues with initial incubation of specimens in buffer ATL and Proteinase K at 56 °C overnight. Isolated DNA was stored at −20 °C until further use. For the amplification of partial ITS-2 regions of F. magna, F. hepatica, or D. dendriticum, an individual set of specific primers was used (Králová-Hromadová et al. 2008, Bazsalovicsová et al. 2010, Table 1). Developmental stages of cestodes detected in liver material of red deer and wild boar were differentiated by multiplex PCR according to Trachsel et al. (2007), targeting the genes nad1 of E. multilocularis and rrnS of E. granulosus/Taenia spp., respectively (Table 1). PCR reactions with trematode and liver samples were performed in a total volume of 50 µl, containing 5 µl of template DNA, 0.5 µM of each primer, 1.5 mM MgCl2, 1U Invitrogen PlatinumTM Taq DNA Polymerase (Thermo Fisher Scientific, Darmstadt, Germany), and 0.1 mM dNTP-Mix (Roche, Mannheim, Germany) using a BIOMETRA T3 thermocycler (Analytic Jena AG, Germany). Multiplex PCR of cestode samples was performed using the QIAGEN Multiplex PCR Kit with a reaction volume of 50 µl, containing 25 µl Qiagen master mix, 5 µl of template DNA, and 5 µl of primer mix (2 µM of each primer Cest1-4 and 16 µM of primer Cest5; Table 1). PCR products were visualized on 1.5% agarose gels, purified using the QIAquick PCR purification kit (Qiagen, Hilden, Germany) according to the manufacturer’s recommendations and sequenced in two directions by MWG Eurofins Genomics sequencing services (Ebersberg, Germany). Forward and reverse sequences were complemented and aligned using online tools (Reverse Complement, http://www.bioinformatics.org/sms/rev_comp.html; ClustalOmega, https://www.ebi.ac.uk/Tools/msa/clustalo). Database searches and sequence comparisons were performed with BLAST provided by the National Center for Biotechnology Information (BLAST, http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Data analysis
The occurrences of F. magna and other liver parasites for different origins, age, and sex groups were compared with a χ2-test. p-values < 0.05 were considered to be significant.
Results
Pathological findings
A macroscopically visible hepatic damage indicating an infection with F. magna was present in 38 samples originating from red deer. Mild pathological alterations of the liver tissue were documented in 15, moderate in 14, and major in nine cases, respectively (Fig. 2a–c). Livers exhibiting Fascioloides specimens (23 cases, Fig. 2d–e) showed black pigmented streaks as well as pseudocysts lined with connective tissue (up to 5 cm in diameter) filled with one up to 66 F. magna specimens (median value of 3) with a body length ranging from 1.5 to 8 cm (Fig. 2a–e). Similar hepatic alterations were present in livers without Fascioloides specimens except that migration tracks and pseudocysts were filled with yellowish to dark brown material of fluid, pasty, pulpy, or firm consistency.
Acute fresh infections were marked by solitary migration tracks filled with fresh blood and liquid liver tissue (Fig. 2f). In chronic cases, the entire liver was indurated with connective tissue and partly minimized in size (Fig. 2c). In 68.4% of F. magna-positive livers, slight to severe perihepatitis villosa was present (Fig. 2h).
Histological alterations in F. magna-positive livers
Histologically, the trematode-specific black blood pigment of hemoglobinic origin “haematin” was present in 37 of 38 Fascioloides-suspicious livers as well as in single liver lymph nodes (Fig. 3a–d). In most cases, black pigmented migration tracks were formed within the hepatic parenchyma whereas tightly clustered pigment accumulations marked the ground part of fibrous capsules. Additionally, large thin-walled trematode eggs were found in variable quantity in livers populated with mature flukes (Fig. 3b). Further histopathological results typical for F. magna were moderate to high-grade periportal fibrosis, lymphoplasmacellular inflammatory reactions (Fig. 3c), focal hemorrhage, and tissue necrosis. The severity of all mentioned alterations varied depending on the infectious stage of the affected animals.
Parasitological and molecular investigations
Mature or immature specimens of the giant liver fluke were detected and morphologically identified in 23 out of 38 Fascioloides-suspicious livers. Fluke material was further examined by F. magna-specific PCR (Table 1) to proof the results of the morphologically based determinations. All DNA samples produced amplicons of the expected size (152 bp) with sequence homologies between 99 and 100% to GenBank accession no. DQ683545.1 (partial F. magna 5.8S ribosomal RNA gene and internal transcribed spacer 2). Large trematode eggs of the type Fascioloides/Fasciola were present in 31 sediments of rinsing liquid of liver tissue (Table 2). In 21 cases, those eggs could be identified as F. magna by PCR. In two samples, it was possible to identify the eggs as Fasciola hepatica with 100% sequence homology to GenBank accession no. MT423007.1. Both cases could be classified as single infections with the latter trematode. In eight samples, the extraction of DNA from eggs was insufficient for a determination of the fluke species by PCR.
In 63 (9%) of all 700 investigated livers, other liver parasites besides F. magna were detected and further investigated by PCR for species identification. Namely, parasitic stages of Dicrocoelium dendriticum (40 pieces of red deer and two roe deer, 6%), Fasciola hepatica (two pieces of red deer, 0.3%), Taenia hydatigena (11 pieces of red deer and one wild boar, 1.7%), and Echinococcus multilocularis (7 pieces of wild boar, 1.0%) were found. Coinfections were restricted to four cases in red deer harboring F. magna and D. dendriticum simultaneously. A single coinfection with T. hydatigena and E. multilocularis occurred in the liver of one wild boar.
Occurrence of F. magna and regional distribution
In consideration of all utilized diagnostic methods (morphological identification of fluke specimens, Fascioloides-DNA or haematin in liver tissue), F. magna was detected in 5.9% (38/640) of livers from red deer (Table 2), whereas none of the investigated livers of wild boar, roe, and fallow deer was infected. Statistically, an upper limit for the occurrence in wild boar and roe deer was estimated as 8.2% and 11.7%, respectively (p = 0.05). Animals older than 1 year were significantly more often infected with the trematode compared to animals younger than 1 year (p < 0.05; Table 3). The difference of occurrence rates for F. magna between male and female pieces was statistically insignificant (p = 0.76). According to the results of the present study, the American suction worm can be detected in the military training ground Grafenwöhr and in the Bavarian Forest National Park with an estimated prevalence of 3.6% and 16.4%, respectively (Table 4). A determination of the regional occurrence of F. magna for all other included districts was not applicable due to low sample numbers.
Discussion
The evidence of mild to moderate alterations in the majority of infected livers (29/38 or 76.3%) is well known for red deer belonging to the group of definitive hosts of F. magna (Pybus 2001). Equivalent lesions are caused by limited migration of immature flukes followed by subsequent encapsulation of mature specimens which is usually associated with pressure atrophy of adjacent liver parenchyma (Pybus 2001). In general, the extent of liver damage in positive animals varied depending on the amount of flukes and age of infection in the present study. Thus, a high fluke burden within one organ resulted in higher number of fibrous capsules and a chronic course of infection was marked by increased general fibrosis of liver tissue. Positive livers containing F. magna specimens harbored a median of three flukes representing a general low infection burden in the investigated animal population. In the course of its life, F. magna might grow up to the remarkable size of 8–10 cm and reaches sexual maturity with a body length of 3–5 cm leading to patent infections marked by egg-shedding with the feces of the infected host (Ursprung et al. 2006). Accordingly, trematode eggs were only diagnosed in the sediment of livers populated by flukes with a minimum size of 3 cm in the study at hand.
In most recently conducted surveys, the diagnosis of F. magna relies on a macroscopic investigation of liver tissue, solely (Novobilisky et al. 2007; Plötz et al. 2015; Rehbein et al. 2021; Ursprung et al. 2006). Furthermore, in the framework of some of those epidemiological surveys, organs were only further investigated after the detection of suspicious alterations in the affected livers by hunters. Consequently, F. magna possibly represents an underdiagnosed parasite since slight changes in the liver tissue might be overlooked at first sight in the field. In order to collect reliable data on the occurrence of Fascioloides, the inset of additional diagnostic methods on a broad set of non-preselected samples is highly recommended. Besides the general macroscopic examination, a histological investigation of affected liver tissue seems to be a valuable supplementary diagnostic method in order to verify an infestation with F. magna since it is not always possible to detect adult stages or trematode eggs (Table 2). The migration of immature Fascioloides-flukes has been reported to be associated with the formation of characteristic haematin in hepatic tissue (Campbell 1960; Blažek and Gilka 1970). Hence, all 37 out of 38 Fascioloides-suspicious livers containing this pigment of hemoglobinic origin were considered as F. magna-positive in the framework of this study (Table 2). Thirty-three of these cases were confirmed by the detection of Fascioloides-DNA obtained from hepatic tissue, large trematode eggs, or fluke material. In four cases, the histological finding of haematin was the only indication for an infection with the large American liver fluke while neither gross Fascioloides-specific pathological alterations, nor flukes, nor trematode eggs were present. It was possible to verify two of those suspected cases by subsequent PCR. The exclusive histological investigation might reveal false negative results in chronic cases with encapsulated flukes and sample collection at the wrong location within the liver tissue (Campbell 1960). Accordingly, no haematin was detectable in the histological cut of one liver in the present survey despite the molecular verification of infection (Table 2). The investigation for F. magna-like eggs in the sediment of altered liver tissue is another complementary method to confirm the diagnosis, especially in chronic infections with macerated Fascioloides-specimens. Even though macroscopic liver alterations differ depending on the concerned trematode species, an investigation of large trematode eggs by PCR with subsequent sequencing of obtained isolates is necessary to differentiate F. magna from Fasciola hepatica whose eggs appear identical microscopically (Bazsalovicsová et al. 2010; Králová-Hromadová et al. 2008).
The macroscopic examination of livers revealed other liver parasites besides F. magna in almost 9% (n=63) of all included animals. Unexpectedly, coinfections of the invasive species F. magna and Fasciola hepatica were not present even though both flukes share the same intermediate host and F. hepatica is a common endoparasite in cervids (Böhm et al. 2006). It is also worth mentioning that infections with Fascioloides were nineteen times more frequent in the investigated material than infections with Fasciola. This might indicate that the giant liver fluke is even better adapted to habitats, intermediate, and final hosts than its indigenous counterpart. One out of three animals shedding eggs of both Dicrocoelium dendriticum and F. magna showed a chronic cholangitis which is pathognomonic for the small liver fluke. The other two livers had lesions compatible with a Fascioloides-infection, confirming that Dicrocoelium infections are generally less apparent than those caused by other flukes (Otranto and Traversa 2003). Even though information on the occurrence and pathological impact of metacercariae of Taenia hydatigena in cloven hoofed game in Germany is limited, the finding of hepatic Cysticercus tenuicollis in wild ungulates has already been reported from Turkey (Cengiz et al. 2019), Poland (Filip et al. 2019), Argentina (Flueck et al. 2012), Slovac Republic (Letková et al. 2008), Hungary (Murai et al. 1979), Canada (Pybus 1990), Iran (Solaymani-Mohammadi et al. 2003), and Russia (Tretyakov et al. 2022). The verification of a genetic similarity between metacestodes from Polish moose and adult thin-necked bladderworms from wolves across the German border allows the assumption of a possible correlation with the recolonization of wolves in Central Europe as possible final hosts of Taenia hydatigena (Filip et al. 2019). Accordingly, eggs of the latter parasite were present in 13% of fecal samples investigated in the framework of a survey on the endoparasite fauna of wild European grey wolves from Germany (Bindke et al. 2019). Moreover, the detection of Cysticercus tenuicollis cysts seems to be a common incidental finding in wild boar from Europe (Filip et al. 2019; Sgroi et al. 2019 and 2020). Additionally, seven of 35 wild boars in this study showed typical alterations for degenerated calcified metacestodes of the fox tapeworm (E. multilocularis) which have already been described in feral pigs as dead-end hosts not constituting any influence on the epidemiology of the generally zoonotic cestode (Boucher et al. 2005; Pfister et al. 1993).
Since the first record of F. magna in wild ungulates in northeastern Bavaria, Germany, in 2011, the imported American trematode has successfully established further habitats including the two districts Upper Palatinate and Upper Franconia (Plötz el al. 2015; Rehbein et al. 2021; König et al. 2019). Especially, the Veldensteiner forest (Upper Franconia) has recently been identified as a hot spot of the giant liver fluke colonizing game, with reported local prevalence rates of 40–71% in red deer (König et al. 2019). The latter forest is located next to the military training ground Grafenwöhr (Fig. 1) harboring important populations of game species susceptive to the parasite. Even though prevalence data based on area-wide investigations of fecal or tissue samples originating from different final hosts is still lacking, there have been single reports of the occurrence of F. magna in certain other Bavarian regions to date. During a sampling period from 2011 to 2014, the investigation of 89 livers originating from wild boar, sika deer (Cervus nippon), fallow, roe, and red deer hunted down in one geographical area in the Upper Palatinate forest revealed characteristic gross lesions of F. magna infection in 40.6% (Rehbein et al. 2021). Moreover, within the framework of a subsequent regional survey of 1,280 fecal samples from fallow and red deer kept in a deer farm located in the northeast of Upper Palatinate close to the border to the Czech Republic from 2012 to 2013, Fascioloides-eggs were present in 4.9% (Plötz el al. 2015). According to the results of the present study, F. magna was present in 38 of 640 livers (5.9%) originating from red deer considering all diagnostic methods. This comparatively low occurrence might be explained by the fact that a broad number of non-selected organs were examined within the course of driven hunts independent of macroscopically visible liver alterations raising suspicion of trematode-infections. The other investigated animal species wild boar and roe deer both harbored no giant liver flukes even though infections with the latter parasite have already been described (Foreyt at al. 1975; Rehbein et al. 2021). Likewise, no Fascioloides specimens were detected in the livers of 33 pieces of wild boar from Italy during a sampling period of three years by Balbo et al. (1989). However, in the present study, the combined sample number (59 livers of feral pigs and roe deer) was an order of magnitude lower compared to red deer (640 livers) and thus a possible occurrence of approximately 10% per species might be reasonable. In general, a direct comparison of occurrence data with previously conducted surveys remains difficult due to divergent investigation matrices (for example, feces versus livers), sampling strategies (selected versus unselected samples), investigated animal population (wild ranging versus game kept in reserves), and applied diagnostic methods.
With respect to the geographical origin of positive animals, the presence of the giant liver fluke was confirmed in red deer originating from the military training ground Grafenwöhr, Upper Palatinate with an estimated prevalence of 3.6%. A total of 16.4% of all investigated red deer from the Bavarian Forest National Park were infected with F. magna representing the first report of F. magna in Lower Bavaria. A possible explanation for the effective dispersion of the parasite in Germany is the migration of infected cloven hoofed game from highly endemic regions in the Czech Republic to bordering German counties harboring suitable habitats for appropriate intermediate hosts enabling a completion of the infection cycle (Rehbein et al. 2021). Within the last decades, numerous endemic F. magna foci have been reported from the Czech Republic with prevalences ranging up to 95% in populations of both free and game ranging cervids (Erhardova-Kotrla 1971; Kasny et al. 2012; Novobilský et al. 2007). As early as 2007, so-called enzootic wet regions in the west of the country providing ideal living conditions not only for freshwater snails as intermediate hosts but also for deer as final hosts have already been assumed to support the spread of the giant liver fluke to adjacent regions in southeastern Germany (Novobilský et al. 2007).
Whereas the impact of the sex turned out to be insignificant, the age of Fascioloides-infected animals of the study was statistically significant in the present study. Accordingly, animals older than 1 year were 13 times more often infected compared to animals younger than 1 year. Those findings are in accordance with Rehbein et al. (2021) and Plötz et al. (2015) and might be due to the wide range of variations concerning the prepatent period of the giant liver fluke in ruminant hosts ranging from three up to 7 months (Foreyt and Todd 1976). Thus, most infected animals do not show typical liver lesions in their first year of life because of the long development time of immature flukes.
To summarize the major results of the present study, the giant liver fluke should be considered as an emerging endoparasite in red deer from northeastern Bavaria. The detection of novel habitats of the fluke in Lower Bavaria raises the assumption that neighboring counties of recent Fascioloides hotspots also harbor large populations of the giant liver fluke. Since high mortality rates in connection with a so-called Fascioloides-malnutrition syndrome have been described in infected game ranging red deer, increasing numbers of affected animals should be taken seriously with regard to the preservation of a good health status of the cloven-hoofed game population in the affected counties (Balbo et al. 1989). Consequently, a continuation of the monitoring in northeastern Bavaria is advisable for the successive hunting seasons. Regarding the composition of samples, a higher number of livers originating from roe deer and sheep as endangered aberrant hosts should be included in order to get information on the spreading of F. magna through infected red deer populations. The outcome of the comparative investigations of the present study emphasizes the importance to apply different diagnostic methods besides a macroscopic examination of livers for a reliable detection of F. magna.
Availability of data and material
One gene sequence (partial ITS-2-gene) of an adult Fascioloides magna specimen which was identical with all Fascioloides-sequences obtained within the framework of the present study was uploaded exemplarily to GenBank Ac. No. OK093210.
References
Bassi R (1875) Sulla cachessia ittero-verminosa, o marciacia dei Cervi, causata dal Distomum magnum. Il Medico Veterinario 4:497–515
Balbo T, Rossi L, Meneguz PG (1989) Integrated control of Fascioloides magna infection in Northern Italy. Parassitologia 31:137–144
Bazsalovicsová E, Králová-Hromadová I, Špakulová M, Reblánová M, Oberhauserová K (2010) Determination of ribosomal internal transcribed spacer 2 (ITS2) interspecific markers in Fasciola hepatica, Fascioloides magna, Dicrocoelium dendriticum and Paramphistomum cervi (Trematoda), parasites of wild and domestic ruminants. Helminthologia 47:76–82. https://doi.org/10.2478/s11687-010-0011-1
Bindke JD, Springer A, Janecek-Erfurth E, Böer M, Strube C (2019) Helminth infections of wild European gray wolves (Canis lupus Linnaeus, 1758) in Lower Saxony, Germany, and comparison to captive wolves. Parasitol Res 118:701–706. https://doi.org/10.1007/s00436-018-6181-3
Blažek K, Gilka F (1970) Contribution on the knowledge of the pigment found in infection with Fascioloides magna. Folia parasitological (Praha) 17:165
Boucher JM, Hanosset R, Augot D, Bart JM, Morand M, Piarroux R, Pozet-Bouhier F, Losson B, Cliquet F (2005) Detection of Echinococcus multilocularis in wild boars in France using PCR techniques against larval form. Vet Parasitol 129(2005):259–266
Böhm C, White PC, Daniels MJ, Allcroft DJ, Munro R, Hutchings MR (2006) The health of wild red and sika deer in Scotland: an analysis of key endoparasites and recommendations for monitoring disease. The Veterinary Journal 171(2):287–294. https://doi.org/10.1016/j.tvjl.2004.10.020
Campbell WC (1960) Nature and possible significance of the pigment in Fascioloidiasis. J Parasitol 46(6):769–775
Cengiz G, Tenekeci GY, Bilgen N (2019) Molecular and morphological characterization of Cysticercus tenuicollis in red deer (Cervus elaphus) from Turkey. Acta Parasitol 64:652–657. https://doi.org/10.2478/s11686-019-00085-1
Erhardová-Kotrlá B (1971) The occurrence of Fascioloides magna (Bassi, 1875) in Czechoslovakia. Publishing House of the Czechoslovak Academy of Sciences, Prague, p 155
Filip KJ, Pyziel AM, Jeżewski W, Myczka AW, Demiaszkiewicz AW, Laskowski Z (2019) First molecular identification of Taenia hydatigena in wild ungulates in Poland. EcoHealth 16(1):161–70. https://doi.org/10.1007/s10393-019-01392-9
Flueck T and Smith-Flueck AD (2012) Diseases of introduced red deer in Patagonia and implications for native ungulates. Anim Prod Sci 52.766 – 773. https://doi.org/10.1071/AN11342
Foreyt WJ, Todd AC, Foreyt K (1975) Fascioloides magna (Bassi, 1875) in feral swine from southern Texas. J Wildl Dis 11:554–558. https://doi.org/10.7589/0090-3558-11.4.554
Foreyt WJ and Todd AC (1976) Development of the large American liver fluke Fascioloides magna in white-tailed deer, cattle and sheep J Parasitol 62: 26–32. https://doi.org/10.2307/3279036
Foreyt WJ (1990) Domestic sheep as rare definitive host of the large American Liver Fluke Fascioloides magna. J Parasitol 76(5):736–739
Foreyt WJ (1996) Susceptibility of bighorn sheep (Ovis canadiensis) to experimentally-induced Fascioloides magna infections. Journal of Wildlife Diseases 32(3):556–559
Janicki Z, Konjevic D, Severin K (2005) Monitoring and treatment of Fascioloides magna in semi-farm red deer husbandry in Croatia. Vet Res Comm 29:83–8. https://doi.org/10.1007/s11259-005-0027-z
Kasny M, Beran L, Siegelova V, Siegel T, Leontovyc R, Berankova K, Pankrac J, Kostakova M, Horak P (2012) Geographical distribution of the giant liver fluke (Fascioloides magna) in the Czech Republic and potential risk of its further spread. Vet Med. 57:101–9
König A, Ehrmantraut C (2019) Appearance of the American liver fluke in northern Bavaria. 34rd International Union of Game Biologists (IUCB, Congress 2019 Kaunas, Lithuania – 26–30 August 2019)
Králová-Hromadová I, Špakulová M, Horáčková E, Turčeková L, Novobilský A (2008) Sequence analysis of ribosomal and mitochondrial genes of the giant liver fluke Fascioloides magna (Trematoda: Fasciolidae): intraspecific variation and differentiation from Fasciola hepatica. J Parasitol 94(1):58–67. https://doi.org/10.1645/GE-1324.1
Letková V, Lazar P, Soroka J, Goldová M, Čurlík J (2008) Epizootiology of cysticercosis of wild cervids. Nat Croat. 17(4):311–318
Majoros G, Sztojkov V (1994) Appearance of the American liver fluke Fascioloides magna (Bassi, 1875) (Trematoda: Fasciolata) in Hungary. Parasit Hung 27:27–38
Murai É, Sugár L (1979) Taeniid species in Hungary (Cestoda, Taeniidae). I. Cysticercosis, cenurosis and hydatidosis in wild ungulates. Parasit Hung 12:41–52
Novobilský A, Horáčková E, Hirtová L, Modrý D, Koudela B (2007) The giant liver fluke Fascioloides magna (Bassi 1875) in cervids in the Czech Republic and potential of its spreading to Germany. Parasitol Res 100:549–553. https://doi.org/10.1007/s00436-006-0299-4
Naem S, Budke C, Craig TM (2012) Morphological characterisation of adult Fascioloides magna (Trematoda: Fascioloidae): first SEM report. Parasitol Res 110:971–978. https://doi.org/10.1007/s00436-011-2582-2
Nagy E, Jócsák I, Csivincsik A, Zsolnai A, Halász T, Nyúl A, Plucsinszki Z, Simon T, Szabó S, Turbók J, Nemes C, Sugár L, Nagy G (2018) Establishment of Fascioloides magna in a new region of Hungary: case report. Parasitol Res 117:3683–3687. https://doi.org/10.1007/s00436-018-6099-9
Otranto D, Traversa D (2003) Dicrocoeliosis of ruminants: a little known fluke disease. Trends Parasitol 19(1):12–15. https://doi.org/10.1016/S1471-4922(02)00009-0
Pfister T, Schad V, Schelling U, Lucius R, Frank W (1993) Incomplete development of larval Echinococcus multilocularis (Cestoda: Taeniidae) in spontaneously infected wild boars. Parasitol Res 79:617–618
Plötz C, Rehbein S, Bamler H, Reindl H, Pfister K, Scheuerle MC (2015) Fascioloides magna – epizootiology in a deer farm in Germany. Berl Munch Tierarztl Wochenschr 128:177–182. https://doi.org/10.2376/0005-9366-128-177
Pybus MJ (1990) Survey of hepatic and pulmonary helminths of wild cervides in Alberta. Canada. J of Wildl Dis 26(4):453–459
Pybus MJ (2001) Liver flukes. In: Samuel WM, Pybus MJ, Kocan AA (eds) Parasitic diseases of wild mammals, 2nd edn. Iowa State Univ. Press, Iowa, pp 121–149
Rajsky D, Čorba J, Várady M, Špakulová R, Cabada J (2002) Control of fascioloidosis (Fascioloides magna Bassi, 1875) in red deer and roe deer. Helminthologia 39(2):67–70
Rehbein S, Visser M, Hamel D, Reindl H (2021) Occurrence of the giant liver fluke, Fascioloides magna, in sympatric wild ungulates in one area in the Upper Palatinate Forest (northeastern Bavaria, Germany). Parasitol Res 120:553–561. https://doi.org/10.1007/s00436-020-06996-7
Sattmann H, Hörweg C, Gaub L, Feix A, Haider M, Walochnik J, Rabitsch W, Prosl H (2014) Wherefrom and whereabouts of an alien: the American liver fluke Fascioloides magna in Austria: an overview. Wien klin Wochenschr 126:23–31. https://doi.org/10.1007/s00508-014-0499-3
Sgroi G, Varcasia A, D’Alessio N, Varuzza P, Buono F, Amoroso MG, Boufana B, Fioretti A, Veneziano V (2019) Massive Taenia hydatigena cysticercosis in a wild boar (Sus scrofa) from Italy. Acta Parasitol 64:938–941. https://doi.org/10.2478/s11686-019-00110
Sgroi G, Varcasia A, Dessì G, D’Alessio N, Pacifico L, Buono F, Neola B, Fusco G, Santoro M, Toscano M, Fioretti A, Veneziano V (2020) Taenia hydatigena cysticercosis in wild boar (Sus scrofa) from southern Italy: an epidemiological and molecular survey. Parasitology 147:1636–1642. https://doi.org/10.1017/S0031182020001559
Solaymani-Mohammadi S, Mobedi I, Rezaian M, Massoud J, Mohebali M, Hooshyar H, Ashrafi K, Rokni MB (2003) Helminth parasites of wild boar, Sus scrofa, in Luristan province, western Iran and their public health significance. J. Helminthol. 77:263–267. https://doi.org/10.1079/JOH2003168
Trachsel D, Deplazes P, Mathis A (2007) Identification of taeniid eggs in the faeces from carnivores based on multiplex PCR using targets in mitochondrial DNA. Parasitology 134:911–920. https://doi.org/10.1017/S0031182007002235
Tretyakov AM, Kiriltsov EV, Chernih VG, Boryaev GI (2022) Biodiversity conservation of commercially hunted ungulates in Transbaikalia ensuring effective veterinary measures. IOP Conf. Ser.: Earth Environ Sci. 953. https://doi.org/10.1088/1755-1315/953/1/012042
Ursprung J, Joachim A, Prosl H (2006) Vorkommen und Bekämpfung des Amerikanischen Riesenleberegels, Fascioloides magna, in einer Schalenwildpopulation in den österreichischen Donauauen. Berl Munch Tierarztl Wochenschr 119:316–323
Acknowledgements
We thank the technical team of the Department for Pathology, Parasitology, and Bee Diseases for the great support processing the livers. Further thanks go to the staff members of the German forestry agency for assistance with the sample collection during driven hunts at the military training areas Grafenwöhr and Hohenfels.
Funding
Open Access funding enabled and organized by Projekt DEAL. The Bavarian Hunting Association (BJV) bore part of the costs for the liver examinations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sommer, M.F., Drdlicek, J., Müller, M. et al. Fascioloides magna and other liver parasites in cloven-hoofed game from northeastern Bavaria, Germany: occurrence and pathological findings with special emphasis on red deer (Cervus elaphus). Eur J Wildl Res 68, 73 (2022). https://doi.org/10.1007/s10344-022-01616-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-022-01616-4