Abstract
Near-natural or semi-natural forests such as relatively undisturbed and old deciduous or mixed woodland are considered worth protecting and ecologically valuable habitats for bat conservation. In contrast, mono-specific forest plantations are considered ecologically less valuable; thus, decision-makers recommend these plantations as suitable locations for wind power stations and therefore want to further expand wind turbines in these habitats. This is expected to have a strong negative impact on the landscape because forests would be cleared for wind turbine pads and access roads and wind turbines rise above the trees with adverse impacts for bats. Therefore, we argue that, in light of bat conservation, the suitability of forest plantations for wind energy development is not, per se, warranted and that implications of wind power stations, even in mono-specific forest plantations, should be assessed and evaluated. We conducted long-term bat activity monitoring and recorded bat echolocation calls above the canopies of different forest sites (coniferous monoculture plantations and semi-natural mixed deciduous forests) in Germany and compared different forest types in terms of species richness, total bat activity, activity of the three bat species groups and species composition. Generalised linear models revealed that forest type and the amount of forest biotopes did not enhance bat activity. Ordination showed that species composition was not affected by forest type, location and connectivity. Mono-specific forest plantations can harbour a diverse bat fauna with high species activity and are, therefore, valuable bat habitats just as near-natural or semi-natural woodlands are. Environmental impact assessment and mitigation measures are vital in all forest types before and after planning for wind energy turbines. In particular, future planning and approval processes must consider the importance of mono-specific forest plantations for bat species protection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Woodlands are vital for many bat species by providing habitats, roosting sites and hunting grounds (e.g. Jantzen and Fenton 2013; Kalda et al. 2015; Charbonnier et al. 2016a). In general, the functional quality of woodlands is determined by a number of landscape and habitat parameters such as landscape connectivity and complexity (Boughey et al. 2011; Frey-Ehrenbold et al. 2013; Fuentes-Montemayor et al. 2013). It has been widely shown that anthropogenic impacts negatively influence the distribution of forest bat species by affecting roosting and foraging habitats (fragmentation, Lesiński et al. 2007; habitat loss, Crampton and Barclay 1998; logging, Fukui et al. 2011). Furthermore, negative changes in bat density and diversity are often related to wind farms or wind turbines, respectively (Kunz et al. 2007a, 2007b; Horn et al. 2008; Cryan and Barclay 2009; Rydell et al. 2010), which is of particular importance because many bat species are endangered at global, national and regional levels (Mickleburgh et al. 2002; Jones et al. 2009).
In Germany, renewable energies are forecasted to rise to about 40–45% of the gross electricity consumption by 2025 to fulfil climate protection goals and up to 80% by 2050, with wind energy being the most important energy source (Informationsportal Erneuerbare Energien 2020). Aside from open landscapes, woodlands are considered potential locations for wind power stations. The German Federal Agency of Nature Conservation (BFN 2011) estimates that about 14% of existing German woodlands are suitable sites. In contrast to near-natural or semi-natural woodlands, mono-specific forest plantations are lower quality habitats for bats, and decision-makers are recommending them for wind power stations (e.g. MUGV Brandenburg 2011). Decision-makers argue that mono-specific forest plantations—characterised by one tree species (pine in NE-Germany) (Klimo et al. 2000)—are ecologically less valuable, as they emaciate the soil, are particularly sensitive to windfall and pests and are less diverse in plant and animal species (Klimo et al. 2000; MUGV Brandenburg 2011). Therefore, wind energy development in mono-specific forest plantations is seen as ecologically sound and, thus, has become the focus of attention of wind power operators (BFN 2011). The general consensus is therefore that the expansion of wind turbines in these plantations should be promoted although landscape changes are associated with this (for instance cleared forests for wind turbine pads and access roads as, wind turbines above the remaining forest patches).
In contrast, near-natural or semi-natural woodlands, such as deciduous and mixed deciduous forests, are considered worth protecting and ecologically valuable habitats, and several studies prove their importance for bat conservation (Kaňuch and Krištín 2005; Kaňuch and Celuch 2007; Zahn et al. 2008; Kalda et al. 2015). This is especially true in terms of wind energy development and its negative effects on bats and bat habitats. It is known that higher collision rates may occur due to higher bat activities above the canopy level (Grunwald and Schäfer 2007; Brinkmann et al. 2011; Zahn et al. 2014). Furthermore, indirect impacts, such as habitat loss, can be expected through logging, as many bat species strongly depend on tree holes as roosting sites (Meschede and Heller 2000; Dietz et al. 2009).
To ensure environmentally sustainable energy production, the implications of wind power stations in mono-specific forest plantations versus near-natural or semi-natural woodlands should be assessed and evaluated. A number of studies highlighted the adverse effects of wind power stations for bats and bat habitats in woodlands (e.g. Brinkmann et al. 2011; Hötker et al. 2006; Aschoff et al. 2006, Kunz et al. 2007b), but most studies focused on near-natural or semi-natural woodlands while forest plantations have been nearly neglected in Europe (but see Kirkpatrick et al. 2017a). This is a drawback because the adverse effects of wind turbines may even be stronger through the attraction effects of wind turbines for bats (Cryan et al. 2014). We hypothesise that mono-specific forest plantations can be valuable bat habitats and that stand type is not necessarily a predictor of bat activity as long as appropriate features such as tree holes, food availability and beneficial habitat structure or connectivity, respectively, are provided at the landscape and habitat level (Charbonnier et al. 2016b).
Methods
Field methods
We recorded bat echolocation calls with the batcorder system (Batcorder—ecoObs, Nuremberg, Germany) above the canopies of 24 different forest sites in Germany (Appendices Fig. 3 and Table 3). All sites were in proposed wind farms in a landscape dominated by different forest types. Bat activity can be highly variable with the daily variation of activity patterns being as high as the variation between different sites (Mathews et al. 2016, pers. observation). There are several guidelines recommending minimum buffer zones of 200 m to habitats of importance to bats (Rodrigues et al. 2015; Kelm et al. 2014). Consequently, we defined a 200-m radius for each location around the recording device to assess the environmental variables using QGIS 3.4 (Quantum GIS Development Team 2018 - © GeoBasis-DE/LGB, dl-de/by-2-0). In this radius, we calculated the amount of the forest biotopes (in percent) within each site and evaluated the available forest type and assigned it as a coniferous plantation (consisted of a single pine species (Pinus sylvestris)) or mixed deciduous forest (if at least 50% of the forest cover belonged to this category) (Table 1). All forest patches showed a similar age structure for around 50 years (± 20 years). To assess the connectivity, we measured all linear landscape elements, e.g. forest edges, hedges, roads or streams. We measured the distance (in m) from the recording device to the nearest forest edge that marked a change in habitat type (roads or streams were not considered) or water body. We defined water bodies as those with standing water and a minimum size of 8 × 8 m.
At each site, we recorded calls for 57–183 entire nights (sunset to sunrise) between April and November from 2011 to 2018. To ensure comparability, all surveys were conducted on mild nights (> 10 °C ambient temperature), without rain and low wind speeds (visual assessment with ≤ small branches moving, which according to the Beaufort scale is a maximum speed of 3.4–5.4 m/s). We detected the bat passes above the canopy with a batcorder fixed in a weatherproof box that was attached on a pole protruding the treetops about 1.5 m. The data transmission was facilitated via a GSM module, and the power supply was provided by a solar panel. All batcorders recorded the full frequency spectrum between 16 and 150 kHz in real time with a sampling rate of 500 kHz. We used a threshold of 27 dB and a pre-trigger of 400 ms for the recordings.
We analysed the ultrasound recordings with the batIdent and bcAnalyze software (ecoObs, Nuremberg, Germany) to organise and identify species-specific echolocation calls. This software automatically measures the primary sound parameters of the recorded echolocation calls and calculates a probability value that a call is from a specific bat species. The software identifies bat echolocation calls from simultaneous recordings of up to three individuals per sequence (Runkel and Marckmann 2009). After the automatic analysis, we manually validated all echolocation calls for misidentifications and, when possible, identified additional calls following the criteria of species identification described in Hammer and Zahn (2009) and Skiba (2003). Where the recording quality allowed, we identified the following bats to species level: Barbastella barbastellus, Eptesicus serotinus, Myotis daubentonii, Myotis myotis, Myotis nattereri, Nyctalus leisleri, Nyctalus noctula, Pipistrellus nathusii, Pipistrellus pipistrellus, Pipistrellus pygmaeus. As some bat species are acoustically nearly indistinguishable, we combined the recordings of Myotis brandtii and Myotis mystacinus to the species group Mbra and the species Plecotus auritus and Plecotus austriacus to the species group Plecotus. Furthermore, we included all Myotis-like, Nyctalus-like, Pipistrellus-like and Plecotus-like calls in the categories Myotis sp., Nyctaloid, Pipistrelloid and Plecotus sp. For testing environmental effects on bat species composition and activity, we first classified all bat species following their collision risk at wind turbines as well as their foraging and echolocation behaviour to one of the three groups, Nyctaloid group (high collision risk—open space forager), Pipistrelloid group (medium collision risk—edge space forager) and Myotis/Plecotus group (low collision risk—narrow space forager). For detailed information about classifying bats in groups and guilds, see Denzinger and Schnitzler (2013). To describe bat activity, we counted the number of bat passes for each of the different species or groups. A ‘bat pass’ was defined as a sequence of at least two echolocation pulses of a passing bat (Thomas and West 1989). Therefore, we assume that the number of bat passes is highly correlated with bat activity. In addition to the already high reliability and precision of the batcorder system, we professionally calibrated all used microphones prior to their field use to warrant the same detectability under similar conditions. Consequently, all zero observations were regarded statistically as true absences of bats in a certain range.
Data analysis
The data analysis was done using the free software package R-3.4.4 (R Core Team 2019). To test, if environmental variables (explanatory variables: amount of forest patches, connectivity, distance to forest edge, distance to next water body, forest type) had a significant effect (alpha = P < 0.05) on total bat activity, the species group activity and species numbers as response variables, we performed generalised linear models (GLM). Furthermore, we used the number of recording days as an offset variable to correct for different recording intensities. To avoid multicollinearity, we only included environmental parameters with correlations of |r| < 0.7. To compensate for the detected overdispersion, the standard errors were corrected using a quasi-Poisson GLM (Crawley 2007; Zuur et al. 2009). The most appropriate models were stepwise (backward) determined by analysis of deviance (Zuur et al. 2009). The residual deviance was used as a goodness-of-fit measure by calculating the pseudo-R2(Dobson 2002). Direct comparisons of categorical variable ‘forest type’ were conducted with post hoc Tukey’s pairwise comparisons using R package LSMEANS.
In order to detect groupings of sample sites and the impact of environmental variables on species composition, bat frequencies were subjected to non-metric multidimensional scaling (NMDS) using VEGAN and MASS packages in R. For ordination, the frequencies of each species were square root transformed. NMDS is an iterative ordination method that places samples in a k-dimensional space using the ranked distances between them (McCune and Grace 2002). The ordination was based on the Bray-Curtis dissimilarity matrix. In search of a stable solution, a maximum of 100 random starts was used. We applied Kruskal’s stress formula multiplied by 100, as given in McCune and Grace (2002). After two trials, two convergent solutions were found for a three-dimensional model. Afterwards, environmental data was fitted onto the ordination. The significance of the explanatory power of each variable was evaluated with a Monte-Carlo randomisation test (10,000 permutations) (Manly 1997) using R function ‘envfit’.
Results
In all, we detected 10 bat species (Appendix Table 3). Most frequent were Nyctalus noctula, Pipistrellus pipistrellus, Pipistrellus pygmaeus and Pipistrellus nathusii that occurred at all study sites. Generalised linear models revealed that total bat activity (total bat passes) (Chi2 = 1.56, P = 0.20, GLM) and number of bat species (Chi2 = 0.65, P = 0.42, GLM) in coniferous plantations and mixed deciduous forests did not differ significantly (Fig. 1). Moreover, forest type did not significantly affect the activity level of the three species groups (Table 2), Myotis/Plecotus group (Chi2 = 0.48, P = 0.49, GLM), Nyctaloid group (Chi2 = 0.83, P = 0.36, GLM) and Pipistrelloid group (Chi2 = 2.82, P = 0.09, GLM). Bat activity level in general (Chi2 = 3.87, P = 0.05, GLM) and the activity level of the Nyctaloid (Chi2 = 3.72, P = 0.05, GLM) and Pipistrelloid groups (Chi2 = 3.07, P = 0.08, GLM) increased with closer distance to the next water body as a statistical trend. Also, as a statistical trend, the amount of forest patches negatively affected total bat activity (Chi2 = 3.40, P = 0.07, GLM) and Nyctaloid group activity (Chi2 = 3.34, P = 0.07, GLM) while Myotis/Plecotus species responded positively (Chi2 = 3.08, P = 0.08, GLM). All other environmental variables had no impact on the activity levels of the groups or on the number of encountered species.
Bat activity and number of bat species (mean and standard error of the mean) in coniferous plantations and mixed deciduous forest did not differ significantly between both forest types (bat activity: Chi2 = 1.56, P = 0.20, GLM; number of bat species: Chi2 = 0.65, P = 0.42, see Table 2). For bat activity, additional post hoc Tukey’s pairwise comparisons of categorical variable ‘forest type’ have been conducted: z.ratio = 1.23, P = 0.22
The NMDS based on the bat species activity level revealed no significant effects of the environmental variables (Fig. 2). None of the variables (connectivity, distance to forest edge, distance to next water body, forest or forest type) affected species groups (amount of forest patches: F = 0.1, R2 = 0.01, P = 0.96; connectivity: F = 0.4, R2 = 0.02, P = 0.73; distance to forest edge: F = 0.4, R2 = 0.02, P = 0.75; distance to next water body: F = 1.4, R2 = 0.06, P = 0.25; forest type: F = 2.1, R2 = 0.09, P = 0.10; permutational multivariate analysis of variance with 9999 permutations).
Ordination plot (NMDS, 2 dimensions, stress = 0.18) based on bat species activity (number of bat passes). Forest types are displayed as triangles (= coniferous plantations) and circles (= mixed deciduous forests). Environmental variable did not affect species distribution (amount of forest patches: F = 0.1, R2 = 0.01, P = 0.96; connectivity: F = 0.4, R2 = 0.02, P = 0.73; distance to forest edge: F = 0.4, R2 = 0.02, P = 0.75; distance to next water body: F = 1.4, R2 = 0.06, P = 0.25; forest type: F = 2.1, R2 = 0.09, P = 0.10; Permutational multivariate analysis of variance with 9999 permutations). Species abbreviations: B.bar = Barbastella barbastellus, E.ser = Eptesicus serotinus, M.bra = Myotis brandtii/mystacinus, M.dau = Myotis daubentonii, M.myo = Myotis myotis, M.nat = Myotis nattereri, N.lei = Nyctalus leisleri, N.noc = Nyctalus noctula, P.nat = Pipistrellus nathusii, P.pip = Pipistrellus pipistrellus, P.pyg = Pipistrellus pygmaeus, Pl.au = Plecotus auritus/austriacus
Discussion
Impact of forest type
It is well known that near-natural or semi-natural woodlands, such as deciduous and mixed deciduous forests, are ecologically valuable bat habitats; thus, they are considered worth protecting (e.g. Jantzen and Fenton 2013; Kalda et al. 2015; Charbonnier et al. 2016a, 2016b). However, we found no significant differences in bat densities in mixed deciduous forest and mono-specific forest plantations. Given that none of the bat groups responded to forest type, we encourage a change of thinking regarding the ecological value of mono-specific forest plantations for bats. Even if forest plantations are ecologically critical, they still provide a habitat with equally high bat activity as near-natural or semi-natural woodlands (Pereira et al. 2016; da Silva et al. 2019). Moderate silvicultural methods can also promote bat diversity (Kirkpatrick et al. 2017a), such as harvesting, which creates a patch mosaic with different tree densities (Perry et al. 2007; Blakey et al. 2016; Kirkpatrick et al. 2017b).
Kalda et al. (2015) found only a minor impact of forest type on bat diversity. They concluded that natural and anthropogenic woodlands could be seen as a single land-cover type regardless of the dominating tree species. In addition, Glendell and Vaughan (2002) found forest plantations to be suitable habitats for bats, as they showed that bats similarly forage in semi-natural and plantation woodlands. Bats depend on a variety of landscape and habitat properties, for example forest edge structures for commuting and foraging (Hein et al. 2009; Kelm et al. 2014; Morris et al. 2010; Jantzen and Fenton 2013), open patches (Loeb and O’Keefe 2006; Perry et al. 2007; Kirkpatrick et al. 2017b), diverse surrounding landscape compositions (Boughey et al. 2011; Frey-Ehrenbold et al. 2013; Heim et al. 2018), water bodies (Morris et al. 2009; Heim et al. 2018) and roosting sites (Kühnert et al. 2016; Coronado et al. 2017; Dietz et al. 2018). However, all these features potentially exist in all diverse, near-natural and mono-specific forest plantations (Cruz et al. 2016; Kirkpatrick et al. 2017a). According to Ruczyński et al. (2010) and Toth et al. (2015), woodland bats may change their roost site preferences and could, thus, occupy habitats previously considered less suitable. Runkel (2009) observed that bats generally use a wide range of forest habitats, but only a few specialists succeed in niche partitioning in forests with a low spacing between trees. Our results show that many open and edge space species, such as Pipistrellus pipistrellus, Pipistrellus nathusii, Eptesicus serotinus and Nyctalus noctula, also had high activity in coniferous forestry zones which adds to findings of Kirkpatrick et al. (2017b) for Sitka spruce (Picea sitchensis) plantations. All these species are negatively affected by wind energy development (Brinkmann et al. 2011) and at risk from wind turbines (Müller et al. 2013).
Differences in species composition naturally occur across different regions; however, other drivers—such as landscape composition, habitat structure and stand origin—are comparable (Kalda et al. 2015). In our study, species composition was not influenced by forest type, even though Pipistrellus species usually show a preference for open patches in forests, such as thinnings and windbreaks (Kirkpatrick et al. 2017b), which are habitat conditions that rather occur in mixed deciduous forests. However, Pipistrellus pipistrellus is a generalist species that can reach high densities in manifold landscapes (Davidson-Watts et al. 2006). Moreover, Myotis daubentonii can use open space landscapes (Kalda et al. 2015) but strongly depends on ponds and lakes as primary foraging habitats (Glendell and Vaughan 2002; Dietz et al. 2009). In general, Myotis species are known to select habitat structure over forest type. Thus, bats of the Myotis genus are able to negotiate cluttered woodland interiors (Glendell and Vaughan 2002).
Impact of spatial variables
There were no significant effects of forest type, but we found that distance to the next water body had a positive impact on the activity level of the Nyctaloid group, as well as on the overall bat activity. Water bodies play an important role as a bat habitat, as they are visited for drinking, offer an abundance of prey (Fukui et al. 2006) and are landmarks for bat orientation and navigation (Serra-Cobo et al. 2000).
Habitat connectivity is a very important driver for bat diversity and density, while fragmentation and isolation, in contrast, negatively affect bats (Gorresen and Willig 2004; Yates and Muzika 2006; Lesiński et al. 2007) especially in the presence of wind turbines (Roscioni et al. 2014). The amount of forest area did not affect our three functional bat groups. However, generally, the amount of forest enhances bat activity, which corroborates the great importance of woodlands (e.g. Jantzen and Fenton 2013; Kalda et al. 2015; Neece et al. 2018). In contrast, higher stocking levels and lesser distances between tree trunks can decrease the activity of certain species (Runkel 2009). This is especially true for Pipistrellus species and barbastelle bats (Barbastella barbastellus) that are active in semi-open airspace and are dependent on thinnings (Kaňuch and Krištín 2005; Dietz et al. 2009).
Impact of wind development in plantations
Harvesting operations in forests do not differ crucially from interventions in monoculture plantations. They are usually excluded from any BACI (before-after-control-impact design, Smith 2002) examination (Mortimer 2006) and nevertheless lead to fragmentation, depreciation or loss of habitats (Farneda et al. 2015). The impact of wind turbine construction in forests is no exception at first, as habitat alteration through linear clearings for the installation and maintenance may result in habitat alteration or roost loss as well (Meschede and Heller 2000). Still, the extent of interference in plantations varies greatly depending on the tree species planted. While the loss of roosting opportunities due to felling in Picea sitchensis plantations is limited (Kirkpatrick et al. 2017a), several bat species are affected by possible roost loss in Pinus nigra and Pinus sylvestris plantations (Mortimer 2006, pers. observation). The main difference between the impact of harvesting and wind turbine construction is the possible attraction effects for bats at wind turbines (Cryan et al. 2014). Habitat alteration, attracting lights or accumulated insect prey at wind turbines, may also foster an increased bat activity in forest plantations due to increased foraging opportunities especially for Nyctalus or Pipistrellus species (Ober et al. 2020; Kirkpatrick et al. 2017a). Installed wind turbines could act as an ecological trap in the newly emerged foraging areas (Tscharntke et al. 2012). Yet, a comparative study with a broad dataset of 193 nacelle height monitoring surveys showed that the overall activity at nacelle height is not necessarily higher at wind turbines in forest habitats compared to open habitats (Reers et al. 2017). Our results show a high bat activity over the canopy of monocultural plantations. Moreover, Reers et al. (2017) argue that bat activity also needs to be measured at the lowest rotor outreach. Thus, longer-term monitoring of possible impact zones should be considered for wind turbines with larger rotor diameter. Currently, the evaluation of negative effects of wind energy development in forests is limited because bat fatality searches are incredibly difficult; the heterogeneous environment of forest grounds hinders finding possible carcasses or carcasses may get caught in the surrounding trees (an umbrella effect). Snapshot acoustic monitoring or the limits of carcass searches may underestimate the collision risk for bats in forested areas.
Conclusions
Mono-specific forest plantations can be considered just as valuable bat habitats as near-natural or semi-natural woodlands. This is especially true when water bodies are existent in the immediate vicinity because these habitats have an overall positive effect on bat activity. While many studies use acoustic snapshots to compare bat activity or even conduct risk assessment during planning processes, our study strongly demonstrates the importance of long-term monitoring above the canopy to evaluate bat activity. All forest types potentially harbour a diverse bat fauna with high species activity. Therefore, wind turbine development in mono-specific forest plantations is far from ecologically sound. Our results suggest that environmental impact assessments and mitigation measures are vital in all forest types before and after planning for wind energy turbines. In this context, it is mandatory to include the entire bat fauna by also focussing on species hunting above the canopy level, because migration behaviour and species composition may differ among different forest strata. We are aware that industrial wind energy production is needed to meet the growing demand for energy but, if bat activity is high, we need to include mitigation measures for all habitat types to assure sustainable development.
References
Aschoff T, Holderied MW, Marckmann U, Runkel V (2006) Forstliche Maßnahmen zur Verbesserung von Jagdlebensräumen von Fledermäusen. Osnabrück, Deutsche Bundesstiftung Umwelt
BFN (Bundesamt für Naturschutz) (2011) Windkraft über Wald - Positionspapier des Bundesamtes für Naturschutz. Bonn, Bundesamt für Naturschutz
Blakey RV, Law BS, Kingsford RT, Stoklosa J, Tap P, Williamson K (2016) Bat communities respond positively to large-scale thinning of forest regrowth. J Appl Ecol 53:1694–1703
Boughey KL, Lake IR, Haysom KA, Dolman PM (2011) Effects of landscape-scale broadleaved woodland configuration and extent on roost location for six bat species across the UK. Biol Conserv 144:2300–2310
Brinkmann R, Niermann I, Behr O, Mages J, Reich M (2011) Kurzfassung des Forschungsvorhabens Methoden zur Untersuchung und Reduktion des Kollisionsrisikos von Fledermäusen an Onshore-Windenergieanlagen. Cuvillier, Göttingen
Charbonnier YM, Barbaro L, Barnagaud JY, Ampoorter E, Nezan J, Verheyen K, Jactel H (2016a) Bat and bird diversity along independent gradients of latitude and tree composition in European forests. Oecologia 182:529–537
Charbonnier Y, Gauzere P, van Halder I, Nezan J, Barnagaud JY, Jactel H, Barbaro L (2016b) Deciduous trees increase bat diversity at stand and landscape scales in mosaic pine plantations. Landsc Ecol 31:291–300
Coronado A, Flaquer C, Puig-Montserrat X, Barthe E, Mas M, Arrizabalaga A, Lopez-Baucells A (2017) The role of secondary trees in Mediterranean mature forests for the conservation of the forest-dwelling bat Myotis alcathoe. Are current logging guidelines appropriate? Hystrix 28:240–246
Crampton LH, Barclay RMR (1998) Selection of roosting and foraging habitat by bats in different-aged aspen mixedwood stands. Conserv Biol 12:1347–1358
Crawley MJ (2007) The R book. Wiley, Chichester
Cruz J, Sarmento P, Rydevik G, Rebelo H, White PCL (2016) Bats like vintage: managing exotic eucalypt plantations for bat conservation in a Mediterranean landscape. Anim Conserv 19:53–64
Cryan PM, Barclay RMR (2009) Causes of bat fatalities at wind turbines: hypotheses and predictions. J Mammal 90:1330–1340
Cryan PM, Gorresen PM, Hein CD, Schirmacher MR, Diehl RH, Huso MM, Hayman DTS, Fricker PD, Bonaccorso FJ, Johnson DH, Heist K, Dalton DC (2014) Behavior of bats at wind turbines. Proc Natl Acad Sci 111:5126–15131
da Silva LP, Heleno RH, Costa JM, Valente M, Mata VA, Gonçalves SC, da Silva AA, Alves J, Ramos JA (2019) Natural woodlands hold more diverse, abundant, and unique biota than novel anthropogenic forests: a multi-group assessment. Eur J For Res 138:461–472
Davidson-Watts I, Walls S, Jones G (2006) Differential habitat selection by Pipistrellus pipistrellus and Pipistrellus pygmaeus identifies distinct conservation needs for cryptic species of echolocating bats. Biol Conserv 133:118–127
Denzinger A, Schnitzler HU (2013) Bat guilds, a concept to classify the highly diverse foraging and echolocation behaviors of microchiropteran bats. Front Physiol 4:164
Dietz C, Nill D, von Helversen O (2009) Bats of Britain. In: Europe and Northwest Africa. A & C Black, London
Dietz M, Brombacher M, Erasmy M, Fenchuk V, Simon O (2018) Bat community and roost site selection of tree-dwelling bats in a well-preserved European lowland forest. Acta Chiropterol 20:117–127
Dobson AJ (2002) Introduction to generalized linear models, 2nd edn. Chapman & Hall, London
Farneda FZ, Rocha R, López-Baucells A, Groenenberg M, Silva I, Palmeirim JM, Bobrowiec PE, Meyer CF (2015) Trait-related responses to habitat fragmentation in Amazonian bats. J Appl Ecol 52:1381–1391
Frey-Ehrenbold A, Bontadina F, Arlettaz R, Obrist MK (2013) Landscape connectivity, habitat structure and activity of bat guilds in farmland-dominated matrices. J Appl Ecol 50:252–261
Fuentes-Montemayor E, Goulson D, Cavin L, Wallace JM, Park KJ (2013) Fragmented woodlands in agricultural landscapes: the influence of woodland character and landscape context on bats and their insect prey. Agric Ecosyst Environ 172:6–15
Fukui D, Murakami M, Nakano S, Aoi T (2006) Effect of emergent aquatic insects on bat foraging in a riparian forest. J Anim Ecol 75:1252–1258
Fukui D, Hirao T, Murakami M, Hirakawa H (2011) Effects of treefall gaps created by windthrow on bat assemblages in a temperate forest. For Ecol Manag 261:1546–1552
Glendell M, Vaughan N (2002) Foraging activity of bats in historic landscape parks in relation to habitat composition and park management. Anim Conserv 5:309–316
Gorresen PM, Willig MR (2004) Landscape responses of bats to habitat fragementation in Atlantic forest of Paraguay. J Mammal 85:688–697
Grunwald T, Schäfer F (2007) Aktivität von Fledermäusen im Rotorbereich von Windenergieanlagen an bestehenden WEA in Südwestdeutschland – Teil 2: Ergebnisse. Nyctalus 12:182–198
Hammer M, Zahn A (2009) Kriterien für die Wertung von Artnachweisen basierend auf Lautaufnahmen. Version, 1. Koordinationsstellen für Fledermausschutz, Patersdorf
Heim O, Lenski J, Schulze J, Jung K, Kramer-Schadt S, Eccard JA, Voigt CC (2018) The relevance of vegetation structures and small water bodies for bats foraging above farmland. Basic Appl Ecol 27:9–19
Hein CD, Castleberry SB, Miller KV (2009) Site-occupancy of bats in relation to forested corridors. For Ecol Manag 257:1200–1207
Horn JW, Arnett EB, Kunz TH (2008) Behavioral responses of bats to operating wind turbines. J Wildl Manag 72:123–132
Hötker H, Thomsen K-M, Jeromin H (2006) Impacts on biodiversity of exploitation of renewable energy sources: the example of birds and bats - facts, gaps in knowledge, demands for further research, and ornithological guidelines for the development of renewable energy exploitation. Michael-Otto-Institut, Bergenhusen
Informationsportal Erneuerbare Energien (2020) Das Erneuerbarer-Energien-Gesetz. Online at: https://www.erneuerbare-energien.de/EE/Redaktion/DE/Dossier/eeg.html?cms_docId=132292. Accessed 25 Aug 2020
Jantzen MK, Fenton MB (2013) The depth of edge influence among insectivorous bats at forest–field interfaces. Can J Zool 91:287–292
Jones G, Jacobs DS, Kunz TH, Willig MR, Racey PA (2009) Carpe noctem: the importance of bats as bioindicators. Endanger Species Res 8:93–115
Kalda R, Kalda O, Lōhmus K, Liira J (2015) Multi-scale ecology of woodland bat the role of species pool, landscape complexity and stand structure. Biodivers Conserv 24:337–353
Kaňuch P, Celuch M (2007) Bat assemblage of an old pastured oak woodland (Gavurky Protected Area, central Slovakia). Verspertilio 11:57–64
Kaňuch P, Krištín A (2005) Factors influencing bat assemblages in forest parks. Ekol Bratisl 24:45–56
Kelm DH, Lenski J, Kelm V, Toelch U, Dziock F (2014) Seasonal bat activity in relation to distance to hedgerows in an agricultural landscape in Central Europe and implications for wind energy development. Acta Chiropt 16:65–73
Kirkpatrick L, Maher SJ, Lopez Z, Lintott PR, Bailey SA, Dent D, Park KJ (2017a) Bat use of commercial coniferous plantations at multiple spatial scales: management and conservation implications. Biol Conserv 206:1–10
Kirkpatrick L, Oldfield IF, Park K (2017b) Responses of bats to clear fell harvesting in Sitka spruce plantations, and implications for wind turbine installation. For Ecol Manag 395:1–8
Klimo E, Hager H, Kulhavý J (eds) (2000) Spruce monocultures in Central Europe – problems and prospects. European Forest Institute Proceedings 33:1–208
Kühnert E, Schonbachler C, Arlettaz R, Christe P (2016) Roost selection and switching in two forest-dwelling bats: implications for forest management. Eur J Wildl Res 62:497–500
Kunz TH, Arnett EB, Erickson WP, Hoar AR, Johnson GD, Larkin RP, Strickland MD, Thresher RW, Tuttle MD (2007a) Ecological impacts of wind energy development on bats: questions, research needs, and hypotheses. Front Ecol Environ 5:315–324
Kunz TH et al (2007b) Assessing impacts of wind-energy development on nocturnally active birds and bats: a guidance document. J Wildl Manag 71:2449–2486
Lesiński G, Kowalski M, Wojtowicz B, Gulatowska J, Lisowska A (2007) Bats on forest islands of different size in an agricultural landscape. Folia Zool 56:153–161
Loeb SC, O’Keefe JM (2006) Habitat use by Forest bats in South Carolina in relation to local, stand, and landscape characteristics. J Wildl Manag 70:1210–1218
Manly BFJ (1997) Randomization, bootstrap and Monte Carlo methods in biology. Chapman & Hall, London
Mathews F, Richardson S, Lintott P, Hosken D (2016) Understanding the risk of European protected species (bats) at onshore wind turbine sites to inform risk management. Report by University of Exeter. Report for Renewable UK. Report for UK Department of Energy and Climate Change (DECC)
McCune B, Grace JB (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach
Meschede A, Heller K-G (2000) Ökologie und Schutz von Fledermäusen in Wäldern. Schriftenreihe für Landschaftspflege und Naturschutz 66:1–374
Mickleburgh S, Hutson A, Racey P (2002) A review of the global conservation status of bats. Oryx 36:18–34
Morris AD, Miller DA, Kalcounis-Rueppell MC (2010) Use of forest edges by bats in a managed pine forest landscape. J Wildl Manag 74:26–34
Morris AD, Vonhof MJ, Miller DA, Kalcounis-Rueppell MC (2009) Myotis septentrionalis Trouessart (northern long-eared bat) records from the North Carolina coastal plain. Southeast Nat 8:355–362
Mortimer G (2006) Foraging, roosting and survival of natterer’s bats, Myotis nattereri, in a commercial coniferous plantation. Unpublished PhD thesis. University of St Andrews
MUGV Brandenburg (Ministry for environment, health and consumer protection of Brandenburg) (2011) Decree on wind energy of 1.1.2011 concerning the designation of areas suitable for wind energy, § 8 Abs. 7 — Landuse Planning Act (ROG) Brandenburg, Germany, 2011. Potsdam: Ministry for environment, health and consumer protection of Brandenburg
Müller J, Brandl R, Buchner J, Pretzsch H, Seifert S, Strätz C, Veith M, Fenton B (2013) From ground to above canopy-bat activity in mature forests is driven by vegetation density and height. For Ecol Manag 306:179–184
Neece BD, Loeb SC, Jachowski DS (2018) Variation in regional and landscape effects on occupancy of temperate bats in the southeastern US. PLoS One 13:e0206857
Ober HK, Jones GM, Gottlieb IGW, Johnson SA, Smith L, Brosi B, Fletcher RJ (2020) Bat community response to intensification of biomass production for bioenergy across the southeastern United States. Ecol Appl 30:e02155
Perry RW, Thill RE, Leslie DM (2007) Selection of roosting habitat by forest bats in a diverse forested landscape. For Ecol Manag 238:156–166
Pereira MJR, Peste F, Paula A, Pereira P, Bernardino J, Vieira J, Bastos C, Mascarenhas M, Costa H, Fonseca C (2016) Managing coniferous production forests towards bat conservation. Wildl Res 43:80–92
R Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. URL http://www.R-project.org/. Accessed 01 Dec 2019
Reers H, Hartmann S, Hurst J, Brinkmann R (2017) Bat activity at nacelle height over forest. In: Köppel J (eds) Wind Energy and Wildlife Interactions –Presentations from the CWW 2015. Springer, Cham, pp 79–98
Rodrigues L, Bach L, Dubourg-Savage M, Karapandža B, Kovač D, Kervyn T, Dekker J, Kepel A, Bach P, Collins J, Harbusch C, Park K, Micevski B, Mindermann J (2015) Guidelines for consideration of bats in wind farm projects – Revision 2014. EUROBATS Publication Series No. 6. UNEP/EUROBATS Secretariat Bonn, Germany, p 133
Roscioni F, Rebelo H, Russo D, Carranza ML, Di Febbraro M, Loy A (2014) A modelling approach to infer the effects of wind farms on landscape connectivity for bats. Landsc Ecol 29:891–903
Ruczyński I, Nicholls B, MacLeod CD, Racey PA (2010) Selection of roosting habitats by Nyctalus noctula and Nyctalus leisleri in Białowieża forest-adaptive response to forest management? For Ecol Manag 259:1633–1641
Runkel V (2009) Mikrohabitatnutzung syntoper Waldfledermäuse - Ein Vergleich der genutzten Strukturen in anthropogen geformten Waldbiotopen Mitteleuropas. PhD thesis, University of Erlangen-Nürnberg
Runkel V, Marckmann U (2009) Schuster P. Manual. ecoobs GbR, Nürnberg
Rydell J, Bach L, Dubourg-Savage M-J, Green M, Rodrigues L, Hedenström A (2010) Bat mortality at wind turbines in northwestern Europe. Acta Chiropt 12:261–274
Serra-Cobo J, Lopez-Roig M, Marqucs-Bonet T, Lahuerta E (2000) Rivers as possible landmarks in the orientation flight of Miniopterus schreibersii. Acta Theriol 45:347–352
Skiba R (2003) Europäische Fledermäuse; Kennzeichen, Echoortung und Detektoranwendung, 1st edn. Westarp, Hohenwarsleben
Smith EP (2002) BACI design. In: El-Shaarawi AH, Piegorsch WW (eds) Encyclopedia of environmetrics. Wiley, Chichester, pp 141–148
Thomas DW, West SD (1989) Sampling methods for bats. In: Ruggiero LF, Carey AB (eds) Wildlife-habitat relationships: sampling procedures for Pacific Northwest vertebrates. U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station, Portland, pp 1–20
Toth CA, Cummings G, Dennis TE, Parsons S (2015) Adoption of alternative habitats by a threatened, “obligate” forest-dwelling bat in a fragmented landscape. J Mammal 96:927–937
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batary P, Bengtsson J, Clough Y, Crist TO, Dormann CF, Ewers RM, Fründ J, Holt RD, Holzschuh A, Klein AM, Kleijn D, Kremen C, Landis DA, Laurance W, Lindenmayer D, Scherber C, Sodhi N, Steffan-Dewenter I, Thies C, van der Putten WH, Westphal C (2012) Landscape moderation of biodiversity patterns and processes – eight hypotheses. Biol Rev 87:661–685
Yates MD, Muzika RM (2006) Effect of forest structure and fragmentation on site occupancy of bat species in Missouri Ozark forests. J Wildl Manag 70:1238–1248
Zahn A, Lustig A, Hammer M (2014) Potenzielle Auswirkungen von Windenergieanlagen auf Fledermauspopulationen. Anliegen Natur 36:21–35
Zahn A, Gelhaus M, Zahner V (2008) Die Fledermausaktivität in unterschiedlichen Waldtypen, an Gewässern und im Offenland – eine Untersuchung auf der Herreninsel im Chiemsee (Bayern). Allgemeine Forst- und Jagdzeitschrift 179:173–177
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, Berlin
Acknowledgements
We would like to thank Fred Albespy, Bela Catherin Bruhn, Charlotte Foisl, Michael Schlockermann, Jan Skroblin and Lena Strauss for providing assistance with fieldwork. Many thanks to Tanja Straka for giving feedback on the manuscript and Monika Egerer for linguistic revision.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Road Ecology
Guest Editor: Marcello D’Amico
Appendices
Appendix 1
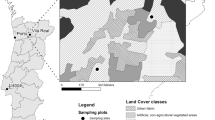
Location of the study region in Germany (a) and location of study plots (dots = coniferous plantations and triangles = deciduous mixed forests) (b). Inlet c shows an example of a study site together with the landscape configuration. The coordinates can be taken from Appendix Table 3
Appendix 2
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Buchholz, S., Kelm, V. & Ghanem, S.J. Mono-specific forest plantations are valuable bat habitats: implications for wind energy development. Eur J Wildl Res 67, 1 (2021). https://doi.org/10.1007/s10344-020-01440-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-020-01440-8